

One of the earliest papers describing optical coherence tomography (OCT) touted the technology as providing “information on the contour and thickness of important retinal structures that can be the basis for new clinical diagnostic procedures.”1 Fast forward to the present, and OCT has revolutionized our understanding, diagnosis, and management of retinal disorders, and it is now the most commonly ordered diagnostic test in medicine.2,3
Although OCT has been readily adopted in the clinical setting, its transition to the OR has met various roadblocks, including lack of integrative solutions, lack of OCT-friendly instrumentation, lack of efficient software analysis systems, and lack of optimal surgeon feedback platforms. The move to portable handheld systems created a major impetus for the adoption of this technology in the surgical setting.4-7 This article reviews some of the uses for intraoperative OCT.
AT A GLANCE
• The introduction of portable OCT systems made it possible to employ these technologies in the OR.
• Intraoperative OCT provides real-time feedback to surgeons to help guide surgical maneuvers.
• During membrane peeling, intraoperative OCT can provide vital guidance.
OCT IN THE OR
Until recently, the two most common systems described for intraoperative use are the handheld Envisu Spectral Domain Ophthalmic Imaging System (SDOIS; Bioptigen) and the stand-mounted iVue (Optovue). Although both systems enable portability, they carry a significant surgeon learning curve, which diminishes their stability and the reproducibility of scan location. This weakness led to the development of portable scan heads tethered to the operating microscope, which enhanced surgeon control of the system via the operating microscope joystick and allowed greater stability (Figure 1).8 Studies involving microscope-mounted OCT systems showed that imaging can be obtained efficiently and with minimal delay in surgical procedures.8 However, more extensive integration, including true microscope integration allowing real-time intraoperative OCT, is needed to ensure widespread adoption.
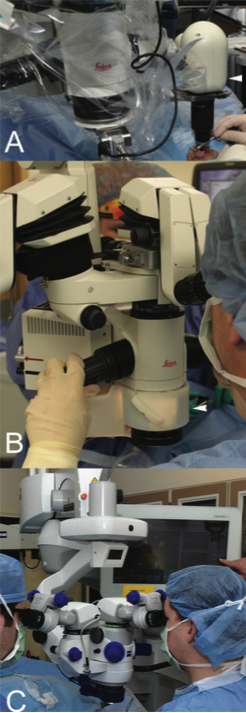
Figure 1. Examples of intraoperative OCT systems: microscope-mounted EnVisu system (arrowhead, A); Cole Eye Institute’s microscope-integrated intraoperative OCT prototype (arrowhead, B); Carl Zeiss Meditec’s Rescan 700 microscope-integrated intraoperative OCT system (C).
INTRAOPERATIVE OCT: WHAT’S THE USE?
Real-Time Feedback
Real-time imaging provides feedback to surgeons to help guide surgical maneuvers via visualization of tissue-instrument interactions. Research groups initially developed novel prototypes and created microscope-integrated OCT systems (Figure 1).9-12 Each of these systems provided a novel approach to microscope-integrated OCT as an add-on system to a conventional microscope. More recently, there have been significant advances in integrative technology, and more seamless commercial systems have been developed that do not alter the overall form of the microscope head (Figure 1).13,14 Each of these systems represents a significant step toward the adoption of OCT into the surgical setting, allowing true real-time intraoperative OCT with visualization of surgical motion.9,15-17
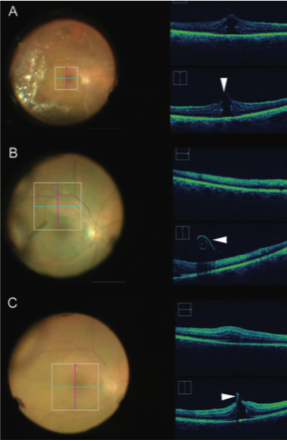
Figure 2. Image-guided feedback with intraoperative OCT during macular hole repair. Location of macular hole (arrowhead) with associated vitreomacular traction identified using pre-peel intraoperative OCT (A). After initiation of ILM peeling, a membrane edge with curled configuration consistent with ILM is easily visualized (arrowhead, B). After complete membrane peel, hole configuration has changed, and minimal residual ILM is visualized at the hole edge (arrowhead, C).
How Am I Peeling?
Early research indicates that intraoperative OCT may have great value during vitreoretinal surgical procedures, particularly those involving the vitreoretinal interface (eg, surgery for macular hole, epiretinal membrane, or vitreomacular traction). The crux of surgical management of these vitreoretinal interface disorders is membrane peeling.
Membrane peeling is a delicate procedure with variable outcomes that demands excellent visualization of transparent tissues. Use of intraoperative OCT during membrane peeling has been shown to provide crucial feedback, which helps with intraoperative surgical decision-making. Studies have shown that in 13% to 22% of cases, surgeons using intraoperative OCT identified residual membranes that required membrane peeling that would otherwise not have been identified.8,13,14 In addition, intraoperative OCT has also revealed both macro- and microarchitectural changes in the retina during membrane peeling resulting from direct instrument-tissue interactions. Commonly reported changes seen following membrane peeling include expansion of the distance between the ellipsoid zone (EZ) and the retinal pigment epithelium and alterations of the inner retinal surface.18-20
Similarly, intraoperative OCT has demonstrated that internal limiting membrane (ILM) peeling during macular hole surgery can cause changes in the geometry of the hole and in the outer retinal architecture (Figure 2). These changes may in turn have implications for anatomic success, defined as primary closure of the macular hole.21 In addition to the geometric changes that occur during ILM peeling, alterations in the EZ have been linked with the speed of anatomic normalization.22
Move Over, Vitreoretinal Dyes
Another important aspect of vitreoretinal interface surgeries is the use of vital dyes and other visualization aids such as such as indocyanine green, triamcinolone acetonide, and other steroid suspensions. Studies have suggested that intraoperative OCT–guided membrane peeling reduces the need for these dyes during vitreoretinal interface surgeries, thereby reducing surgical time and the risks to the retina from use of these dyes.14,23
Repairing RRDs
Another area of vitreoretinal surgery that may benefit from use of intraoperative OCT is the repair of rhegmatogenous retinal detachments (RRDs). Rates of retinal reattachment following surgery are high, and vision recovery tends to be limited. Small studies with intraoperative OCT have demonstrated anatomic changes in the fovea that may help to explain this discrepancy.
For example, perfluoro-n-octane (PFO) infusion during RRD repair appears to induce generalized blunting of the EZ junction and alterations in the foveal architecture, which have been associated with visual recovery in the postoperative period.24 In addition, subretinal fluid is frequently identified after PFO tamponade (Figure 3).8,14,24 These findings highlight yet another potential utility of intraoperative OCT: prognostication of vision outcomes following vitreoretinal interventions.
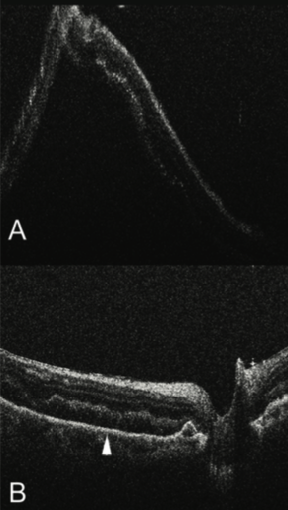
Figure 3. Intraoperative OCT during RRD repair showing bullous detached retina (A). Following perfluorocarbon liquid tamponade, the retina has significantly flattened, but persistent subretinal fluid remains (arrowhead, B).
A PROMISING PLACE IN THE OR
Although numerous studies have highlighted the impact of intraoperative OCT use on surgical decision-making, work remains to be done in exploring whether patient outcomes improve as a result of the adoption of these technologies. The overall surgeon feedback platform also needs further development and refinement, including enhancements to OCT-compatible instrumentation, software platforms, and the integration of OCT data through optimized heads-up feedback. n
1. Huang D, Swanson EA, Lin CP, et al. Optical coherence tomography. Science. 1991;254(5035):1178-1181.
2. Chen TC, Cense B, Pierce MD, et al. Spectral domain optical coherence tomography: ultra-high speed, ultra-high resolution ophthalmic imaging. Arch Ophthalmol. 2005;123(12):1715-1720.
3. Hee MR, Izatt JA, Swanson EA, et al. Optical coherence tomography of the human retina. Arch Ophthalmol. 1995;113(3):325-332.
4. Scott AW, Farsiu S, Enyedi LB, et al. Imaging the infant retina with a hand-held spectral-domain optical coherence tomography device. Am J Ophthalmol. 2009;147(2):364-373.
5. Chavala SH, Farsiu S, Maldonado R, et al. Insights into advanced retinopathy of prematurity using handheld spectral domain optical coherence tomography imaging. Ophthalmology. 2009;116(12):2448-2456.
6. Dayani PN, Maldonado R, Farsiu S, Toth CA. Intraoperative use of handheld spectral domain optical coherence tomography imaging in macular surgery. Retina. 2009;29(10):1457-1468.
7. Radhakrishnan S, Rollins AM, Roth JE, et al. Real-time optical coherence tomography of the anterior segment at 1310 nm. Arch Ophthalmol. 2001;119(8):1179-1185.
8. Ehlers JP, Dupps WJ, Kaiser PK, et al. The prospective intraoperative and perioperative ophthalmic imaging with optical coherence tomography (PIONEER) study: 2-year results. Am J Ophthalmol. 2014;158(5):999-1007.
9. Binder S, Falkner-Radler CI, Hauger C, et al. Feasibility of intrasurgical spectral-domain optical coherence tomography. Retina. 2011;31(7):1332-1336.
10. Tao YK, Ehlers JP, Toth CA, Izatt JA. Intraoperative spectral domain optical coherence tomography for vitreoretinal surgery. Opt Lett. 2010;35(20):3315-3317.
11. Ehlers JP, Srivastava SK, Feiler D, et al. Integrative advances for OCT-guided ophthalmic surgery and intraoperative OCT: microscope integration, surgical instrumentation, and heads-up display surgeon feedback. PloS One. 2014;9(8):e105224.
12. Ehlers JP, Tao YK, Farsiu S, et al. Integration of a spectral domain optical coherence tomography system into a surgical microscope for intraoperative imaging. Invest Ophthalmol Vis Sci. 2011;52(6):3153-3159.
13. Ehlers JP, Kaiser PK, Singh RP, Srivastava SK. Feasability and utility of microscope-integrated intraoperative OCT for vitreoretinal surgery: the DISCOVER study 1-year results. Paper presented at: American Society of Retina Specialists; July 11-14, 2015; Vienna, Austria.
14. Ehlers JP, Goshe J, Dupps WJ, et al. Determination of feasibility and utility of microscope-integrated optical coherence tomography during ophthalmic surgery: The DISCOVER study RESCAN results. JAMA Ophthalmol. 2015;133(10):1124-1132.
15. Ehlers JP, Tao YK, Farsiu S, et al. Visualization of real-time intraoperative maneuvers with a microscope-mounted spectral domain optical coherence tomography system. Retina. 2013;33(1):232-236.
16. Hahn P, Migacz J, O’Connell R, et al. Unprocessed real-time imaging of vitreoretinal surgical maneuvers using a microscope-integrated spectral-domain optical coherence tomography system. Graefes Arch Clin Exp Ophthalmol. 2013;251(1):213-220.
17. Hahn P, Migacz J, O’Connell R, et al. Preclinical evaluation and intraoperative human retinal imaging with a high-resolution microscope-integrated spectral domain optical coherence tomography device. Retina. 2013;33(7):1328-1337.
18. Ray R, Baranano DE, Fortun JA, et al. Intraoperative microscope-mounted spectral domain optical coherence tomography for evaluation of retinal anatomy during macular surgery. Ophthalmology. 2011;118(11):2212-2217.
19. Ehlers JP, Han J, Petkovsek D, et al. Membrane peeling-induced retinal alterations on intraoperative OCT in vitreomacular interface disorders from the PIONEER study. Invest Ophthalmol Vis Sci. 2015;56(12):7324-7330.
20. Nam DH, Desouza PJ, Hahn P, et al. Intraoperative spectral domain optical coherence tomography imaging after internal limiting membrane peeling in idiopathic epiretinal membrane with connecting strands. Retina. 2015;35(8):1622-1630.
21. Ehlers JP, Xu D, Kaiser PK, et al. Intrasurgical dynamics of macular hole surgery: an assessment of surgery-induced ultrastructural alterations with intraoperative optical coherence tomography. Retina. 2014;34(2):213-221.
22. Ehlers JP, Itoh Y, Xu L, et al. Factors associated with persistent subfoveal fluid and complete macular hole closure in the PIONEER study. Invest Ophthalmol Vis Sci. 2014;56(2):1141-1146.
23. Falkner-Radler CI, Glittenberg C, Gabriel M, Binder S. Intrasurgical microscope-integrated spectral domain optical coherence tomography-assisted membrane peeling. Retina. 2015;35(10):2100-2106.
24. Ehlers JP, Ohr MP, Kaiser PK, Srivastava SK. Novel microarchitectural dynamics in rhegmatogenous retinal detachments identified with intraoperative optical coherence tomography. Retina. 2013;33(7):1428-1434.
Justis P. Ehlers, MD
• assistant professor of ophthalmology at the Cole Eye Institute at the Cleveland Clinic in Cleveland, Ohio
• financial interest: consultant to Bioptigen, Leica, Alimera, Alcon, ThromboGenics, and Zeiss; intellectual property licensed to Bioptigen and Synergetics
• ehlersj1@yahoo.com
Mehnaz Khan, MD, MS
• vitreoretinal surgery fellow at the Cole Eye Institute at the Cleveland Clinic in Cleveland, Ohio
• financial interest: none acknowledged









_1773249222.png?auto=compress,format&w=75)










