
First described by Jaffe in 1967, vitreomacular traction (VMT) is a disease entity in which incomplete posterior vitreous detachment (PVD) results in tractional forces on the retina that can lead to morphologic changes and functional vision deficits.1 It is estimated that VMT affects 1% to 2% of the population, although the rate is believed to be much higher among individuals with diabetic eye disease.2 VMT can cause decreased or distorted vision and is a risk factor for the development of a full thickness macular hole.
AT A GLANCE
• VMT can affect vision and result in a full thickness macular hole.
• In roughly two-thirds of cases, untreated VMT results in loss of visual acuity.
• Of patients treated with the highest dose of ALG-1001 in a phase 2 study, 65% had VMT release.
• ALG-1001’s dual mechanism of action may prove beneficial in other disease processes, such as DR, DME, and neovascular AMD.
Surgery is effective, but it is not without potential adverse events such as cataract formation, intraocular pressure changes, macular hole development, bleeding, infection, macular atrophy, retinal breaks, retinal detachment, infection, and damage to the retinal pigment epithelium.3-8 Risk of adverse events aside, the patient’s need for time off for the procedure, positioning requirements, and the inconvenience of follow-up visits are also factors to consider.
Natural history studies suggest that untreated VMT results in loss of visual acuity in about two-thirds of cases, with spontaneous resolution occurring in only about one in 10 cases.9 Thus, this condition has spurred recent interest in the concept of pharmacologic vitreolysis. This article takes a brief look at some nonsurgical treatment options, specifically with the synthetic integrin antagonist ALG-1001 (Luminate, Allegro Ophthalmics).
INVESTIGATING TREATMENT OPTIONS
Ocriplasmin and VMT
In phase 3 clinical trials, ocriplasmin intravitreal injection 2.5 mg/mL (Jetrea, ThromboGenics), which is approved by the US Food and Drug Administration for pharmacologic vitreolysis, demonstrated on optical coherence tomography (OCT) 26% efficacy in causing pharmacologic release of VMT by day 28 after a single intravitreal injection.10 Of note, use of the drug was associated with acute, mainly transient side effects including loss of vision, changes on electroretinography (ERG), photopsias, color vision changes, and outer retinal changes on OCT. In the phase 3 trials of ocriplasmin, vitreous floaters (16.8%) and photopsias (11.8%) were relatively common, and blurred vision and visual impairment occurred in 8.6% and 5.4% of patients, respectively. Postmarketing surveillance revealed dyschromatopsia in 0.4% of patients, compared with 1.7% of patients in the clinical trial program.10 Changes on ERG were present in half of patients with dyschromatopsia. In most cases, these symptoms resolved after 1 to 6 months.
ALG-1001 and VMT
ALG-1001 is primarily an antiangiogenic agent that blocks integrin receptors on vascular endothelial cells that mediate a number of angiogenic processes, including endothelial cell migration, proliferation, differentiation, and maturation. While anti-VEGF agents mainly affect leakage and temporarily abate neovascularization, ALG-1001’s activity is directed at the underlying causative effect. For purposes of this discussion, a secondary effect of ALG-1001, as a vitreolytic agent, is detailed on the following pages.
In a double-masked, placebo-controlled, randomized, multicenter phase 2 study in the United States and Europe, at least 100 individuals with VMT with or without macular hole were enrolled and randomly assigned to one of four treatment groups—2.0-, 2.5-, or 3.2-mg ALG-1001, or balanced salt solution intravitreal injection—and followed for 3 months. The primary endpoint was resolution of VMT by OCT by day 90. Images were assessed by a central reading center in a masked fashion.
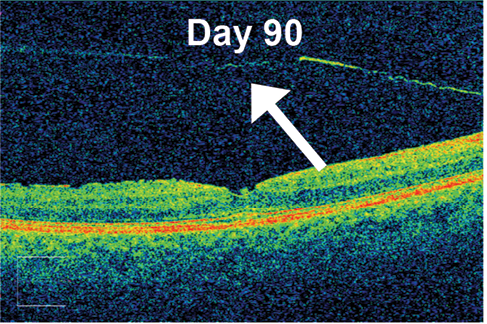
Results from this study suggest an alternative approach to pharmacologic vitreolysis (unpublished data). Although further data analysis is ongoing, roughly 65% of patients in the highest dose group in this dose-ranging study achieved the study’s primary endpoint of complete VMT release on OCT by day 90. Importantly, ALG-1001 was well tolerated, and no significant safety issues were seen. Injections of ALG-1001 can be repeated, and in this study up to three monthly injections were allowed. In patients in the 3.2-mg group (the highest dose tested), an average of 1.6 doses were required to achieve VMT release (Figure).
Overall, 30% of patients treated across all ALG-1001 doses had VMT release, compared with 9.7% in the placebo control group (P = .0129). Release rates were 23% and 21% for the 2.0- and 2.5-mg groups, respectively. Almost half of patients who achieved release (46%) did so after the first injection, while 27% required a second injection and another 27% required the full per-protocol three injections. The average number of injections was 1.6, 2.0, 1.8, and 1.75 in the 3.2 mg, 2.5 mg, 2.0 mg, and control groups, respectively.
There was a significant decrease in central macular thickness in all ALG-1001–treated groups compared with baseline and also when compared with the control group, which may be an indication of ALG-1001’s antiangiogenic effect.
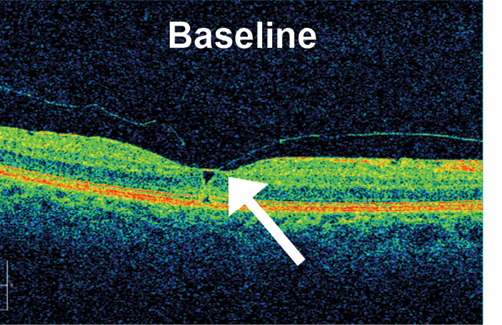
Figure. Example of VMT release in a patient at baseline (TOP) and after 90 days of follow-up after an injection of ALG-1001 3.2 mg (BOTTOM). Note the full separation of the posterior hyaloid membrane from the underlying retinal interface.
ALG-1001: DR, AMD, and DME
An additional study being conducted with ALG-1001 bears mentioning. A high rate of PVD was noted in early phase studies of this compound in patients with diabetic macular edema (DME). The integrin antagonist should block the angiogenic integrin receptors on vascular endothelial cells, thus preventing the development of neovascularization and progression to proliferative DR, while ALG-1001’s vitreolytic properties should be complementary in resolving the pathology that allows vascular growth factors to accumulate in the vitreous with consequent new blood vessel formation in the retina. Thus, the drug’s sponsor is enrolling 100 individuals with nonproliferative diabetic retinopathy (DR) without PVD to investigate ALG-1001’s ability to induce PVD and to protect against progression to proliferative DR.
The antiangiogenic properties of ALG-1001 were also examined in a phase 1b/2a study in patients (n = 15) with end-stage DME. Many of the patients were no longer responding to anti-VEGF therapy, but, after 60 days of active treatment with ALG-1001 followed by 3 months of observation off treatment, mean BCVA had improved by 9.1 letters, and in eight patients it improved by 3 or more lines. There was a corresponding mean 31% reduction in central macular thickness, from a mean 516 µm at baseline to a mean 359 µm at day 120 (60 days off treatment) and 387 µm at day 180 (90 days off treatment). No patient lost vision in the study.
ALG-1001’s dual mechanism of action would seem to have theoretical benefit in other disease processes involving an antiangiogenic component, such as DR and neovascular age-related macular degeneration (AMD). In the latter, posterior hyaloid traction may limit the effectiveness of anti-VEGF therapy. In such patients, ALG-1001 would seem to be an ideal treatment because it would address both choroidal neovascularization and VMT with one (or repeated) injections.
The phase 2b DEL MAR study has completed enrolling patients with DME for a dose-ranging study of ALG-1001. The rationale for the study follows from a phase 1 proof-of-concept study demonstrating up to 3 months of durability with ALG-1001 as monotherapy.
CONCLUSION
The phase 2 data suggest strong efficacy and safety for the treatment of VMT with ALG-1001 and provide rationale for further study. There may be additional implications of the positive findings. A number of observational studies have reported that eyes of patients with neovascular AMD have a higher propensity for an attached hyaloid compared with eyes of those without wet AMD.11,12 It is unknown at this point whether inducing vitreomacular release will reduce the need for anti-VEGF injections in these patients or reduce the possibility of neovascular complications among patients with dry AMD; however, it stands to reason that, if ALG-1001 is approved as an antiangiogenic agent, its secondary effect of inducing a PVD may be helpful in patients over the long run.
Both the AMD and DME investigational programs for ALG-1001 are studying the antiangiogenic properties of the drug, with posterior hyaloid release listed as a secondary endpoint. In concept, it would appear that the drug’s dual mechanism of action could have a synergistic effect in these retinal disease states. However, early analyses of these studies also indicate that ALG-1001 is effective irrespective of traction release.
Finally, if the results of the phase 2 study of ALG-1001 for VMT are confirmed in subsequent studies, ALG-1001 could represent an alternative approach to pharmacologic vitreolysis. n
1. Jaffe NS. Vitreous traction at the posterior pole of the fundus due to alterations in the vitreous posterior. Trans Am Acad Ophthalmol Otolaryngol. 1967;71(4):642-652.
2. Jackson TL, Nicod E, Simpson A, et al. Symptomatic vitreomacular adhesion. Retina. 2013;33(8):1503-1511.
3. Johnson MW. Tractional cystoid macular edema: a subtle variant of the vitreomacular traction syndrome. Am J Ophthalmol. 2005;140(2):184-192.
4. McDonald HR, Johnson RN, Schatz H. Surgical results in the vitreomacular traction syndrome. Ophthalmology. 1994;101(8):1397-1402.
5. Witkin AJ, Patron ME, Castro LC, et al. Anatomic and visual outcomes of vitrectomy for vitreomacular traction syndrome. Ophthalmic Surg Lasers Imaging. 2010;41(4):425-431.
6. Smiddy WE, Michels RG, Glaser BM, deBustros S. Vitrectomy for macular traction caused by incomplete vitreous separation. Arch Ophthalmol. 1988;106(5):624-628.
7. Bottós J, Elizalde J, Arevalo JF, et al. Vitreomacular traction syndrome. J Ophthalmic Vis Res. 2012;7(2):148-161.
8. Yamada N, Kishi S. Tomographic features and surgical outcomes of vitreomacular traction syndrome. Am J Ophthalmol. 2005;139(1):112-117.
9. Hikichi T, Yoshida A, Trempe CL. Course of vitreomacular traction syndrome. Am J Ophthalmol. 1995;119(1):55-61.
10. Stalmans P, Benz MS, Gandorfer A, et al; MIVI-TRUST Study Group. Enzymatic vitreolysis with ocriplasmin for vitreomacular traction and macular holes. N Engl J Med. 2012; 367(7):606-615.
11. Ondes F, Yilmaz G, Acar MA, et al. Role of the vitreous in age-related macular degeneration. Jpn J Ophthalmol. 2000;44(1):91-93.
12. Weber-Krause B, Eckardt C. Incidence of posterior vitreous detachment in the elderly. Ophthalmologe. 1997;94(9):619-623.
Peter K. Kaiser, MD
• the Chaney Family Endowed Chair in Ophthalmology Research; a professor of ophthalmology at the Cleveland Clinic Lerner College of Medicine; a staff surgeon in the vitreoretinal department at the Cole Eye Institute, Cleveland Clinic; and the founding director of the Digital Optical Coherence Tomography Reading Center (DOCTR) at the Cole Eye Institute
• member of the Retina Today editorial board
• financial interest: consultant for Alcon, Bayer, Regeneron,Novartis, Genentech, Kanghong, Neurotech, Ophthotech, Ohr Pharmaceuticals, Aerpio, Allegro, Bioeq, ThromboGenics, and Formycon
• pkkaiser@gmail.com









_1773249222.png?auto=compress,format&w=75)










