

Decreased blood flow to the retina is the underlying pathophysiology in a number of major retinal diseases, including diabetic retinopathy, retinal vascular occlusive disease, and retinopathy of prematurity. This tissue ischemia causes a cascade of cellular events, most notable among these the overproduction of VEGF, which causes neovascularization and increased vessel permeability. Current treatments for these diseases include laser, vitrectomy, and intravitreal anti-VEGF injections. Although these are all effective, they do not address the underlying pathophysiology: decreased tissue oxygen.
AT A GLANCE
• Retinal ischemia is the underlying pathophysiology in diabetic retinopathy, retinal vascular occlusive disease, and retinopathy of prematurity.
• The OxyCell implant contains oxygen-producing cyanobacteria and uses encapsulated cell technology to deliver oxygen intraocularly.
• If it is shown to be safe and effective, the OxyCell implant may have applications in retinal ischemic conditions.
TARGETING RETINAL ISCHEMIA
Encapsulated cell technology (ECT) was first reported by Vincenzo Bisceglie in 1934 as a solution to overcoming immunologic rejection to nonself cells, tissues, or organisms.1 This treatment mechanism holds promise for a variety of human diseases including diabetes, hemophilia, and Parkinson disease.2-4
Clinical studies involving the implantation of a ciliary neurotrophic factor ECT (NT-501, Neurotech) for the treatment of retinal degenerative conditions are ongoing. This ECT implant contains genetically modified human cells within a semipermeable membrane, and these cells produce a factor to prevent retinal cell death.5,6 In general, ECT allows bidirectional mass transport, which permits passage of nutrients and oxygen to the cells but prevents immune cells and antibodies from destroying the cells inside the membrane.7
OXYCELL DEVICE
OxyCell is a novel implant that was designed and tested at the University of Colorado Eye Center. It uses ECT to deliver oxygen intraocularly. OxyCell is an encapsulated intraocular bioimplant that contains oxygen-producing cyanobacteria inside a semipermeable dialysis membrane. These nonpathologic, single-celled organisms originate from the Synechococcus strain, which is ubiquitous in oceans and freshwater environments all over the world. For more than 4 billion years, cyanobacteria have been performing a single metabolic feat: using sunlight to convert carbon dioxide and water into oxygen and simple sugars. The intraocular environment provides a similarly hospitable milieu. Encased in a microdialysis membrane, the organisms can produce oxygen when stimulated by ambient light.
DEVELOPMENT
In designing the OxyCell device, it was advantageous to have a large surface-to-volume ratio and a sustained, continuous release. An initial prototype was constructed out of polyvinyl chloride tubing with a nanofiltration membrane (NF90) secured with cyanoacrylate adhesive. After several iterations, it is now a compact bundle of microdialysis fibers (Figure).
In vitro testing showed a pure Synechococcus cell culture to have an elevated partial pressure oxygen reading. When a vial containing Synechococcus sp. cells was exposed to light and temperature, there was a statistically significant increase in the oxygen content compared with baseline. There was no statistically significant change from baseline in a sterile saline control group. Testing with the Synechococcus sp. in the implant demonstrated an increase in oxygen concentration in a 4-mL saline bath.
Ex vivo testing in freshly enucleated porcine eyes showed an increase in vitreous oxygen concentration for the first 4 days after implantation. The oxygen concentration increase was sustained for the entire 15-day study period. This finding indicated that the cyanobacteria continued to produce oxygen inside the implanted eye over a period of time and that oxygen levels in the eye were significantly higher than in eyes that did not contain a bioimplant.
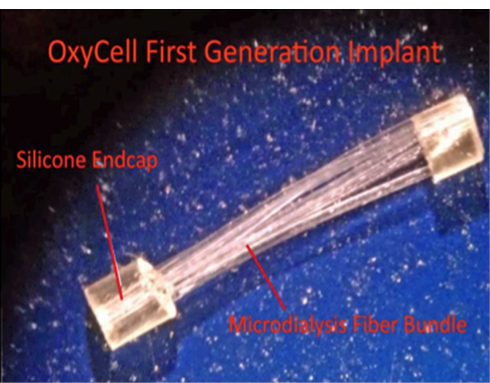
Figure. First-generation OxyCell implant.
IN VIVO STUDIES
Initial in vivo studies have provided a proof of concept to demonstrate the feasibility of oxygen delivery with the intravitreal implant device. In a rabbit model, a central retinal artery occlusion (CRAO) was induced by raising intraocular pressure to 90 mm Hg. Animals were randomly assigned to receive no implant, an empty implant, or an implant with Synechococcus sp. cells. Intraocular oxygen measurements were taken before and after the procedure at various time points for 15 days. Sham implant animals had a hollow polysulfone fiber membrane tube implanted for a total of 30 minutes. After the animals were euthanized, microscopic analysis of retinal thickness was performed.
The oxygen concentration 30 minutes after reperfusion showed a significant increase in vitreous oxygenation in the active implant group, almost three times the oxygen content of the sham group and 14 times the oxygen content in the control group. There were no differences in concentration at days 3, 7, 10, and 15 of follow-up.
These initial studies indicate that the prototype is successful at producing oxygen in the vitreous cavity and suggest that it may prove useful for diseases such as diabetic retinopathy in which the retina receives inadequate oxygenation.
CONCLUSION
The retina has the highest rate of oxygen consumption in the body at 13 mL/100 g/min, making it sensitive to any variation in the oxygen supply.8 Current treatment options for CRAO vary widely, with no standard of care and generally poor outcomes. In CRAO, the inner retinal layers normally served by the inner circulation lose viability, and loss of vision results.8
The use of an external source of oxygen to improve retinal oxygenation during and after exposure to an ischemic injury has been the aim of studies for many decades, with hyperbaric oxygen being the mechanism most extensively studied.9 Nevertheless, the systemic complications associated with sustained delivery of hyperbaric oxygen and its technical difficulties have limited its widespread use in clinical practice and prevented randomized controlled trials.10
The cyanobacteria-containing bioimplant is a novel approach to oxygen delivery directly to the vitreous cavity. We successfully demonstrated that oxygen can be produced in vitro and expressed in high quantities by a bioimplant made of a semipermeable membrane and Synechococcus sp. cells activated by light exposure. In addition, we demonstrated the technique for placing the bioimplant in an ex vivo setting with a resulting increase in vitreous cavity oxygen concentration. While the true biocompatibility of the membrane material has yet to be established, if it is shown to be safe and effective, the bioimplant has several promising investigational and clinical applications for retinal ischemic conditions. n
1. Bisceglie V. Uber die antineoplastiche Immunitat. Zeitschrift fur Krebsforschung. 1934;40(1):122-140.
2. Omer A, Duvivier-kali V, Fernandes J, et al. Long-term normoglycemia in rats receiving transplants with encapsulated islets. Transplantation. 2005;79(1):52-58.
3. Brauker J, Frost GH, Dwarki V, et al. Sustained expression of high levels of human factor IX from human cells implanted within an immunoisolation device into athymic rodents. Hum Gene Ther. 1998;9(6):879-888.
4. Kishima H, Poyot T, Bloch J, et al. Encapsulated GDNF-producing C2C12 cells for Parkinson’s disease: a pre-clinical study in chronic MPTP-treated baboons. Neurobiol Dis. 2004;16(2):428-439.
5. Colton CK. Oxygen supply to encapsulated therapeutic cells. Adv Drug Deliv Rev. 2014;67-68:93-110.
6. Calafiore R, Basta G. Clinical application of microencapsulated islets: actual prospectives on progress and challenges. Adv Drug Deliv Rev. 2014;67-68:84-92.
7. Waga J, Ehinger B. Passage of drugs through different intraocular microdialysis membranes. Graefes Arch Clin Exp Ophthalmol. 1995;233(1):31-37.
8. Murphy-Lavoie H, Butler F, Hagan C. Central retinal artery occlusion treated with oxygen: a literature review and treatment algorithm. Undersea Hyperb Med. 2012;39(5):943-953.
9. Abdallah W, Ameri H, Barron E, et al. Vitreal oxygenation in retinal ischemia reperfusion. Invest Ophthalmol Vis Sci. 2011;52(2):1035-1042.
10. Lee HC, Niu KC, Chen SH, et al. Hyperbaric oxygen therapy in clinical application. A report of a 12-year experience. Zhonghua Yi Xue Za Zhi (Taipei). 1989;43(5):307-316.
Logan Christensen, MD
• ophthalmology resident at the University of Colorado Eye Center in Aurora, Colo.
• financial interest: none acknowledged
Jeffrey L. Olson, MD
• associate professor of vitreoretinal diseases and surgery in the department of ophthalmology at the University of Colorado School of Medicine, Anschutz Medical Campus, in Aurora, Colo.
• financial interest: patent for OxyCell through the Regents of the University of Colorado; research support from Genentech; consultant for MicroSurgical Technology and Galaxy Ophthalmics
• jeffrey.olson@ucdenver.edu









_1773249222.png?auto=compress,format&w=75)










