
A total of 1.7 million new cases of diabetes were diagnosed in 2012, and, as of 2014, 29.1 million people, or 9.3% of the U.S. population, had diabetes.1 Diabetic retinopathy occurs when the small blood vessels that feed the retina are damaged. Although patients with early stages of diabetic retinopathy are typically asymptomatic, their visual acuity is often affected as the disease becomes more severe. Diabetic macular edema (DME) can also cause a decrease in visual acuity and can occur at any level of retinopathy.
AT A GLANCE
• A post hoc analysis of the ranibizumab arms of DRCR.net Protocol I indicates that long-term response to anti-VEGF therapy in patients with DME can be predicted after three injections.
• Patients who do not receive appropriate therapy in a timely fashion may not respond as well as those who do.
• If patients are not likely to gain visual acuity in the long term, injecting an anti-VEGF agent every 4 weeks may not be sustainable in a real-life setting.
VEGF levels are high in the retina and vitreous of eyes with diabetic retinopathy. Focal/grid photocoagulation has been the standard of care for DME for the past 25 years, but anti-VEGF therapy represents a useful therapeutic modality that targets one of the major underlying features of DME: permeability. Although studies have reported promising results in patients with DME treated with anti-VEGF injections, the ability to tell which patients will respond well to the treatment and which will not has evaded retina specialists. This article reviews the findings of the Early Anti-VEGF Response and Long-term Efficacy (EARLY Analysis) of the Diabetic Retinopathy Clinical Research Network (DRCR.net) Protocol I study, which suggests such a prediction may be reliably made after the first three anti-VEGF injections.
PROTOCOL I STUDY DESIGN
The DRCR.net Protocol I study was a randomized trial evaluating ranibizumab (Lucentis, Genentech) plus prompt or deferred laser or triamcinolone plus prompt laser for DME.2 The 3-year, phase 3 multicenter trial, which included 854 eyes (691 patients) with visual acuity of 20/32 to 20/320 and DME involving the fovea, aimed to evaluate the efficacy and safety of 0.5-mg intravitreal ranibizumab plus prompt or deferred laser, or 4-mg intravitreal triamcinolone plus prompt laser, in comparison with sham plus prompt laser for treatment of DME. The primary outcome was change in visual acuity from baseline to 1 year (intent to treat analysis).
The study authors found that intravitreal ranibizumab plus prompt or deferred laser was more effective through at least 1 year compared with prompt laser alone for the treatment of DME involving the central macula.
POST HOC ANALYSIS
Soon after the completion of the DRCR.net Protocol I study, several colleagues and I conducted a post hoc analysis of the ranibizumab arms from the Protocol I 3-year data, looking specifically at the 375 eyes with DME that received anti-VEGF injections.3
Can Response to Anti-VEGF Therapy Be Predicted?
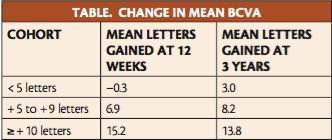
We questioned whether we could predict which patients, in the long term, were going to respond well to anti-VEGF monotherapy based on their status after three injections, at 12 weeks. To answer this question, we analyzed the dataset and stratified the database into three cohorts: those who responded well (ie, gained ≥ 10 letters of visual acuity after three injections); those who did not respond well
(ie, gained < 5 letters of visual acuity after three injections); and those whose responses fell in between (ie, gained between 5 and 9 letters of visual acuity after three injections). The unadjusted differences in mean BCVA change from baseline in these three groups, respectively, were + 15.2, -0.3, and + 6.9 at 12 weeks, and + 13.8, + 3.0, and + 8.2 at 3 years (Table). After multivariate adjustment, significant correlation remained between BCVA gain at week 12 and at years 1 and 3 (P < .001).
In other words, we found that, if we followed these patients on an ongoing basis, their responses were maintained not only for 1 year, but for 3 years (Table). Therefore, if a patient responded well after three injections, he or she would continue to do well for 3 years. Conversely, patients who did not respond well after three injections would not respond well after 3 years, despite following the strict Protocol I regimen of injections every 4 weeks.
These post hoc exploratory findings suggest that after three injections we can predict how a patient will respond in the long term if we continue to treat him or her with anti-VEGF monotherapy. For a patient who may not respond well, therefore, we may want to consider an alternative mode of treatment. We analyzed these data in every statistical variation possible and found that the results remained consistent and were statistically significant and reproducible with sensitivity testing.
Incidentally, we also looked at responses after one and two injections of ranibizumab, and we could see the same predictability even after one injection. However, we chose to focus on three injections in this analysis because we wanted to have a statistically robust sample, and we also felt that administering three injections is consistent with what is done in the real-world clinical setting.
Persistence May Pay Off in Some Cases
We took a closer look at patients who did not respond well initially (gained < 5 letters of visual acuity after three injections) but continued to receive injections. Approximately 28% of these patients responded well (gained ≥ 10 letters of visual acuity) by 1 year. This was also true at 3 years.
This means that, if one continues to administer injections to a patient who is responding poorly, in two-thirds of cases that patient will not gain a lot of vision, even with injections every 4 weeks. This has implications for counseling patients. It may be difficult to convince a patient who has not responded well initially to continue coming in every 4 weeks for a 1-in-3 chance of improvement at 3 years. This regimen is a heavy treatment burden that may simply not be sustainable for most patients.
CLINICAL RELEVANCE
With the data from this post hoc analysis, we can predict with confidence how patients are going to respond after three anti-VEGF injections. We can also determine early whether to continue treatment or to consider alternative modalities.
There is a price to be paid for not selecting the appropriate therapy in a timely fashion. We can extrapolate from studies such as RIDE and RISE and others in which therapies were switched in a patient population.4 In these trials, patients who were initially given sham treatment and then switched to anti-VEGF therapy after 2 years never achieved a response as good as patients who were started on the active therapy from the beginning.
Our analysis was also consistent with what we think may be happening physiologically in this patient population. It is believed that DME evolves from primarily a permeability-driven disease, which can respond to anti-VEGF monotherapy, to primarily an inflammation-driven disease, which may not respond as well to anti-VEGF monotherapy.
The problem is, phenotypically, patients who fall in either group look the same. Until we develop a biomarker or some type of genetic analysis, patients' response to three injections of anti-VEGF therapy may be the best method to determine which phase of this process they are in.
Additional analysis is needed to confirm these findings, but information from the EARLY Analysis may help to shape treatment patterns for patients with DME. n
1. National Diabetes Fact Sheet: National Estimates and General Information on Diabetes and Prediabetes in the United States, 2014. http://www.cdc.gov/diabetes/data/statistics/2014statisticsreport.html. Accessed February 15, 2016.
2. Elman MJ, Aiello LP, Beck RW, et al; Diabetic Retinopathy Clinical Research Network. Randomized trial evaluating ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular edema. Ophthalmology. 2010;117(6):1064-1077.
3. Dugel PU. Long-term response to anti-VEGF therapy for diabetic macular edema can be predicted after three injections: an analysis of Protocol I data. Paper presented at: The American Academy of Ophthalmology Annual Meeting; Nov 13-17, 2015; Las Vegas, NV.
4. Nguyen QD, Brown DM, Marcus DM, et al. Ranibizumab for diabetic macular edema: results from 2 phase III randomized trials: RISE and RIDE. Ophthalmology. 2012;119(4):789-801.
Pravin U. Dugel, MD
• managing partner of Retinal Consultants of Arizona in Phoenix; a clinical professor of ophthalmology, USC Eye Institute, Keck School of Medicine at the University of Southern California in Los Angeles; founding member of the Spectra Eye Institute in Sun City, Ariz.
• member of the Retina Today editorial advisory board
• financial interest: consultant for Allergan, Alcon, Novartis, Roche, and Genentech
• pdugel@gmail.com









_1773249222.png?auto=compress,format&w=75)










