

As the prevalence of diabetes continues to rise, diabetic retinopathy (DR) has become increasingly common.1 The gold standard for diagnosis of DR is a dilated fundoscopic examination by an ophthalmologist. In addition to slit-lamp biomicroscopy, several imaging techniques can be of use in evaluating various manifestations of the disease. Essential imaging techniques ophthalmologists should be familiar with include color fundus photography, fluorescein angiography (FA), B-scan ultrasonography, and optical coherence tomography (OCT). This article reviews these techniques and their roles in examining patients with DR.
AT A GLANCE
• Fundus photography is useful for monitoring changes in degree of DR over time.
• FA can be used to confirm the diagnosis of neovascularization in PDR and to evaluate DME.
• On OCT, retinal fluid in DR manifests as dark spaces or cysts; hard exudates are hyperreflective.
COLOR FUNDUS PHOTOGRAPHY
Color fundus photography is a useful tool for managing diabetic eye disease. Color photos can be used to document DR, and many clinicians find photography useful for counseling patients and explaining pathology to them. In addition, fundus photography can be used to monitor changes in the degree of DR over time. Types of fundus photography include standard, widefield, and stereoscopic.
Standard Fundus Photography
Standard macular fundus photography captures 30˚ of the posterior pole of the eye, including the macula and the optic nerve (Figure 1).2 Standard fundus cameras can collect seven fundus fields, which can be combined to create a montage showing a 75° field of view. Benefits of standard field color photography include ease of use, wide availability, and reproducibility.
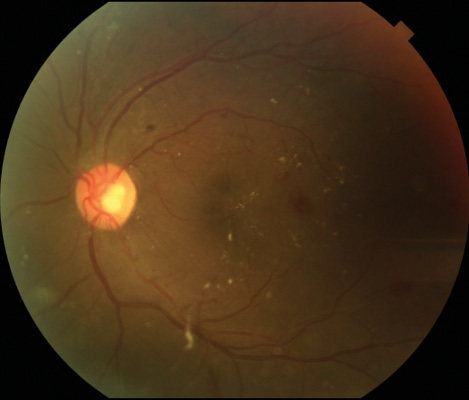
Figure 1. Standard 30 ̊ photo of an eye with DR.
One limitation of fundus photography is that it cannot confirm clinically significant macular edema (CSME). This is an examination finding diagnosed by fundus biomicroscopy with one of three criteria: hard exudates within 500 µm of the fovea with associated retinal thickening; retinal thickening within 500 µm of the fovea; or an area of retina thickening more than 1500 µm in diameter that is less than 1500 µm from the fovea.3 Other limitations of standard fundus photography include decreased quality of imaging in the presence of media opacity, lack of fine retinal detail, and poor visualization of the peripheral retina.
Widefield Fundus Photography
Widefield fundus photography is a newer method of fundus photography that can capture up to a 200° field of view, even through an undilated pupil, allowing viewing of more than 80% of the total retinal surface area.4 There are limitations to this technology as well, including distortion of images due to the spherical nature of the globe, eyelash artifacts, false color representation of fundus findings, and high equipment cost.
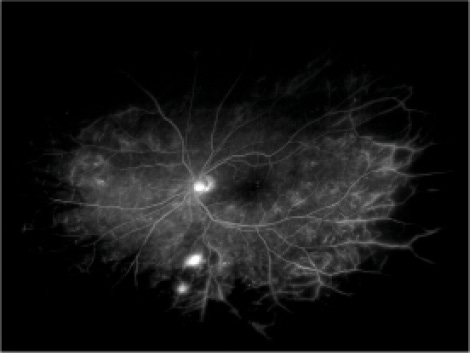
Figure 2. Widefield FA showing significant nonperfusion in the periphery.
Stereoscopic Fundus Photography
Stereoscopic photography is another form of fundus photography that is less often utilized in clinical practice. This modality involves taking two pictures of the fundus and displaying them so that the viewer can fuse the two images, allowing 3-D re-creation of the fundus.
A potential benefit of stereoscopic photography is in helping to distinguish between neovascularization elsewhere, which appears elevated, and intraretinal microvascular abnormalities, which are typically flat.5 However, this technology is more time-consuming than standard photography and has not been shown to significantly improve clinical care. A comparative study of monoscopic and stereoscopic fundus photography found no difference in the ability of a trained ophthalmologist to evaluate the severity of DR using ETDRS criteria.6
FLUORESCEIN ANGIOGRAPHY
FA is another imaging modality useful in patients with DR. This technology was introduced to clinical ophthalmology by Gass et al in 1967.7 FA is useful in delineating the retinal vasculature, which is the part of the retina primarily affected by DR. In patients with diabetes, FA can show microaneurysms that can be difficult to identify clinically. They manifest as punctate areas of hyperfluoresence. Areas of nonperfusion manifest as patchy areas of hypofluoresence. Nonperfusion appears on FA as an enlarged foveal avascular zone; it can lead to macular ischemia and can be an occult cause of visual loss not apparent on clinical exam.
FA also can reveal abnormal blood vessels such as intraretinal microvascular abnormalities or neovascularization in the eye. This imaging method takes place over a period of
5 to 10 minutes, during which leakage of the unbound fluorescein molecules from incompetent retinal vessels can be seen. Diabetic macular edema (DME) can be evaluated using FA by looking for leakage of dye in the macular region. FA can also be used to confirm the diagnosis of neovascularization in proliferative DR (PDR).
Like fundus photography, FA is not part of the ETDRS criteria for determining whether a patient has CSME. Its usefulness for determining treatment parameters for focal laser in macular edema is also unclear.8 FA is an invasive test and can have side effects related to dye administration. It should be noted that fluorescein dye is not iodine-based, so patients with iodine allergy can receive it. The most common complications associated with fluorescein are transient nausea or vomiting in about 3% of patients.9 Patients can have allergic reactions ranging from urticaria to severe anaphylaxis, although the latter is extremely rare.10-12 Most physicians avoid performing FA in pregnant patients, despite absence of any definitive risks.13
Widefield FA
FA also comes with a widefield option (Figure 2). Advantages of this technology over traditional FA include the ability to detect peripheral neovascularization and improved ability to visualize the extent of peripheral retinal nonperfusion.14-16 It has been hypothesized that, in patients with proliferative DR or DME, VEGF production from peripheral areas of ischemia may cause a large amount of disease burden.17 By targeting panretinal photocoagulation (PRP) to these ischemic areas, VEGF may be decreased, which may help diminish the complications of DR in the eye.18,19 There is no conclusive evidence from large randomized studies to confirm whether targeted PRP based on widefield FA imaging can help improve visual outcomes.
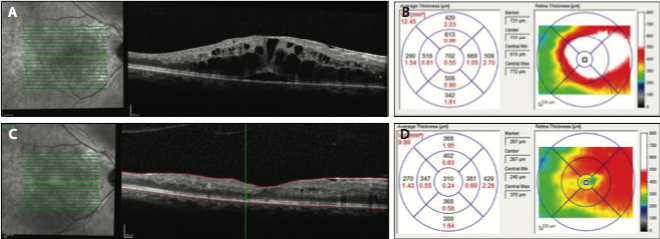
Figure 3. OCT in an eye with DME showing significant intraretinal and subretinal fluid (A). The corresponding OCT map shows increased retinal thickening (B). One month later, following an anti-VEGF injection, there was decreased intraretinal fluid and resolution of the subretinal fluid (C), as well as significant improvement in the thickness on the OCT map (D).
B-SCAN ULTRASONOGRAPHY
B-scan ultrasonography is a useful imaging modality in some patients with DR. High-frequency ultrasound waves are sent from a transducer to ocular tissue and then relayed back to the probe. Based on the time and amplitude of the sound wave that returns, the signal is summed to form a 2-D image of the eye. Higher density tissue returns a higher amplitude (more echogenic) signal that appears as a denser white color on the recreated image.20
This technology is most useful when there is a poor view of the posterior pole. It can be used to evaluate for retinal detachments in PDR when a media opacity such as a cataract or vitreous hemorrhage obscures the retina.21 In a diabetic patient with clear media, this imaging modality is not generally useful clinically.
OPTICAL COHERENCE TOMOGRAPHY
This technology works by emitting light and detecting what is reflected from the retina. Creation of an image from light reflections in OCT is similar to the way an image is created from sound reflections in B-scan ultrasonography. Because light rays are too fast to detect directly, a technique called low-coherence interferometry is employed to allow indirect measurement of the reflected light rays. Since its advent, OCT technology has advanced through a number of software and hardware improvements to become one of the most useful technologies to examine the retina in numerous eye diseases.22-25
In patients with diabetes, OCT is clinically utilized most frequently in those with DME. This objective test has software that is capable of automatically measuring retinal thickness in multiple areas of the macula. The measurements can then be compared to large normative databases. The OCT scan can also be repeated at follow-up visits with accuracy, which is useful for assessing response to treatment over time (Figure 3).26,27
In patients with DME, OCT can show whether the macular edema is center-involving or non–center-involving. This is an important distinction in the age of anti-VEGF therapy, as these agents have frequently been studied specifically in patients with center-involving macular edema. It is important to be familiar with common features of DR on OCT. Retinal fluid, which can either be intraretinal or subretinal, manifests as dark spaces or cysts. Hard exudates, most commonly found in the outer plexiform layer, are hyperreflective on OCT. OCT can also detect areas of subclinical macular edema. In addition, OCT can show loss of photoreceptors, which can help account for poor visual acuity in the absence of macular edema or other retinal pathology.28
Advantages of OCT include the short time needed for patient cooperation, its noncontact and noninvasive nature, and the detail and reproducibility with which it can image the retina. There are also limitations to OCT technology. It is not a substitute for slit-lamp biomicroscopy, nor is it part of the ETDRS criteria for determining CSME. It is also currently unknown at what threshold patients should be treated if they have good visual acuity and retinal swelling that is subclinical but seen on OCT. Additionally, it is unclear whether OCT maps can be useful in defining areas to be treated with focal laser.
CONCLUSION
The imaging modalities described in this article play vital roles in the evaluation and management of DR. Color fundus photography is important for documenting disease and monitoring changes in retinopathy severity over time. FA can show retinal ischemia and leakage from incompetent vessels in macular edema and PDR. B-scan ultrasonography is essential when media opacity is obscuring retinal examination. OCT is a valuable tool to assist with the diagnosis and management of DME. As these technologies continue to improve, their roles in the management of patients with DR are becoming increasingly important. n
1. American Academy of Ophthalmology Retina/Vitreous Panel. Preferred Practice Pattern Guidelines. Diabetic Retinopathy. San Francisco, CA: American Academy of Ophthalmology; 2014. www.aao.org/preferred-practice-pattern/diabetic-retinopathy-ppp--2014. Accessed February 1, 2016.
2. Saine PJ, Tyler ME. Fundus photography overview. In: Ophthalmic Photography: Retinal Photography, Angiography, and Electronic Imaging. 2nd ed. New York, NY: Butterworth-Heinemann; 2001.
3. Early Treatment Diabetic Retinopathy Study Research Group. Treatment techniques and clinical guidelines for photocoagulation of diabetic macular edema. Early Treatment Diabetic Retinopathy Study report number 2. Ophthalmology. 1987;94(7):761-774.
4. Witmer MT, Kiss S. The clinical utility of ultra-wide-field imaging. Rev Ophth. 2012;19(3):60-64.
5. Tyler ME. Stereo fundus photography: principles and technique. J Ophthalmic Photogr. 1996:18(2):68-89.
6. Li HK, Hubbard LD, Danis RP, et al. Monoscopic versus stereoscopic retinal photography for grading diabetic retinopathy severity. Invest Ophthalmol Vis Sci. 2010;51(6):3184-3192.
7. Gass JDM, Sever, RJ, Sparks D, Goren J. A combined technique of fluorescein fundoscopy and angiography of the eye. Arch Ophthalmol. 1967;78(4):455-461.
8. Early Treatment Diabetic Retinopathy Study Research Group. Photocoagulation for diabetic macular edema. Early Treatment Diabetic Retinopathy Study report number 1. Arch Ophthalmol. 1985;103(12):1796-1806.
9. Kwiterovich KA, Maguire MG, Murphy RP, et al. Frequency of adverse systemic reactions after fluorescein angiography. Ophthalmology. 1991;98(2):1139-1142.
10. The diagnosis and management of anaphylaxis. Joint Task Force on Practice Parameters, American Academy of Allergy, Asthma and Immunology, American College of Allergy, Asthma and Immunology, and the Joint Council of Allergy, Asthma and Immunology. J Allergy Clin Immunol. 1998;101(6 Pt 2):S465-528.
11. Fineschi V, Monasterolo G, Rosi R, Turillazzi E. Fatal anaphylactic shock during a fluorescein angiography. Forensic Sci Int. 1999;100 (1–2):137-142.
12. Hitosugi M, Omura K, Yokoyama T, et al. An autopsy case of fatal anaphylactic shock following fluorescein angiography: a case report. Med Sci Law. 2004;44(3):264-265.
13. Berkow JW, Flower RW, Orth DH, Kelley JS. Fluorescein and indocyanine green angiography: technique and interpretation. Monograph 5. San Francisco, CA: American Academy of Ophthalmology; 1997.
14. Ciardella A, Brown D. Wide field imaging. In: Agarwal A, ed. Fundus Fluorescein and Indocyanine Green Angiography: A Textbook and Atlas. Thorofare, NJ: Slack; 2007:79-83.
15. Wessel MM, Nair N, Aaker G, et al. Peripheral retinal ischaemia, as evaluated by ultra-widefield fluorescein angiography, is associated with diabetic macular oedema. Br J Ophthalmol. 2012;96(5):694-698.
16. Friberg TR, Gupta A, Yu J, et al. Ultrawide angle fluorescein angiographic imaging: a comparison to conventional digital acquisition systems. Ophthalmic Surg Lasers Imaging. 2008;39(4):304-311.
17. Oliver SCN, Schwartz SD. Ultra-widefield fluorescein angiography. In: Fernando Arevalo J, ed. Retinal Angiography and Optical Coherence Tomography. New York, NY: Springer Science and Business Media LLC; 2009:407-417.
18. Reddy S, Hu A, Schwartz SD. Ultra wide field fluorescein angiography guided targeted retinal photocoagulation (TRP). Semin Ophthalmol. 2009;24(1):9-14.
19. Muqit MM, Marcellino GR, Henson DB, et al. Optos-guided pattern scan laser (Pascal)-targeted retinal photocoagulation in proliferative diabetic retinopathy. Acta Ophthalmol. 2013;91(3):251-258.
20. Coleman DJ, Silverman RH, Lizzi FL, Rondeau MJ. Ultrasonography of the eye and orbit. 2nd ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2005.
21. Mcleod D, Restori, M. Ultrasonic examination in severe diabetic eye disease. Brit J Ophthalmol. 1979;63(8):533-538.
22. Huang D, Swanson EA, Lin CP, et al. Optical coherence tomography. Science. 1991;254(5035):1178-1181.
23. Gabriele ML, Wollstein G, Ishikawa H, et al. Optical coherence tomography: history, current status, and laboratory work. Invest Ophthalmol Vis Sci. 2011;52(5):2425-2436.
24. Drexler W, Morgner U, Ghanta RK, et al. Ultrahigh-resolution ophthalmic optical coherence tomography. Nat Med. 2001;7(4):502-507.
25. Wojtkowski M, Bajraszewski T, Gorczynska I, et al. Ophthalmic imaging by spectral optical coherence tomography. Am J Ophthalmol. 2004;138(3):412-419.
26. Cunha-Vaz J, Coscas G. Diagnosis of macular edema. Ophthalmologica. 2010;224 Suppl 1:2-7.
27. Sikorski BL, Malukiewicz G, Stafiej J, et al. The diagnostic function of OCT in diabetic maculopathy. Mediators Inflamm. 2013;2013:434560.
28. Diabetic Retinopathy Clinical Research Network. Vitrectomy outcomes in eyes with diabetic macular edema and vitreomacular traction. Ophthalmology. 2010;117(6):1087-1093.e3.
Margaret A. Greven, MD
• clinical instructor of ophthalmology at Byers Eye Institute, department of ophthalmology, Stanford University School of Medicine, in Palo Alto, Calif.
• financial interest: none acknowledged
• margaret.greven@gmail.com
Peter Karth, MD, MBA
• assistant clinical professor at Byers Eye Institute, department of ophthalmology, vitreoretinal section, Stanford University School of Medicine, and co-director of Stanford ophthalmic innovation fellowship, in Palo Alto, Calif.
• financial interest: none acknowledged
• peterkarth@gmail.com









_1773249222.png?auto=compress,format&w=75)










