

AT A GLANCE
• VEGF plays a substantial role in the pathophysiology of DR.
• Serial intravitreal anti-VEGF injections have become the standard of care in patients with center-involved DME.
• Evidence exists that serial intravitreal anti-VEGF injections can prevent PDR onset and may have durable effects.
Although laser strategies are still used in managing diabetic eye disease, the paradigm is shifting toward use of serial intravitreal anti-VEGF injections, particularly for the treatment of patients with DME. As a result, retina specialists are now equipped with targeted therapies that have substantially improved their ability to combat blinding disease. This article explores the rationale in using anti-VEGF therapy and highlights recent insights gained for managing patients with diabetic eye disease.
THE VEGF CONNECTION TO DIABETIC EYE DISEASE
Microvascular damage caused by chronic hyperglycemia due to diabetes can lead to DR. Increased disease duration and worsening glycemic control represent major risk factors for initial development of DR and subsequent progression through advancing stages.4 Therefore, optimization of systemic well-being is fundamental in managing DR. The development of ocular neovascularization separates nonproliferative stages from proliferative DR (PDR).
Exact mechanisms of DR development are unknown, but structural and biochemical abnormalities are implicated. Loss of pericytes and endothelial cells are histopathological features of DR. These factors, which lead to breakdown of the blood-retina barrier along with vascular hyperpermeability of microaneurysms, is believed to contribute to the development of DME. Although many cytokines are implicated in the pathophysiology of DR, VEGF has a substantial role. In fact, VEGF was first called vascular permeability factor due to its property of increasing the permeability of blood vessels.5,6 VEGF is induced by hypoxia and is elevated in the vitreous of eyes undergoing vitrectomy for PDR.7,8 Anti-VEGF injections into the vitreous humor can induce regression of neovascularization and reduce vascular permeability, which are both beneficial effects for patients with DR.
ANTI-VEGF DRUGS VS. DIABETIC EYE DISEASE
The anti-VEGF molecules that have been extensively tested intravitreally for treatment of DR and DME include pegaptanib (Macugen, Bausch + Lomb); bevacizumab (Avastin, Genentech); ranibizumab (Lucentis, Genentech); and aflibercept (Eylea, Regeneron). At this time, bevacizumab is not approved by the US Food and Drug Administration for intravitreal indications; however, it is commonly used off label in this manner. Although inhibition of VEGF is common among these drugs, their pharmacologic profiles and differences in cost can influence clinical decision-making. Pharmaceutical charges vary by region, but, on average, 1.25 mg of bevacizumab costs approximately $70, 0.3 mg of ranibizumab costs around $1200, and 2.0 mg of aflibercept costs nearly $2000.
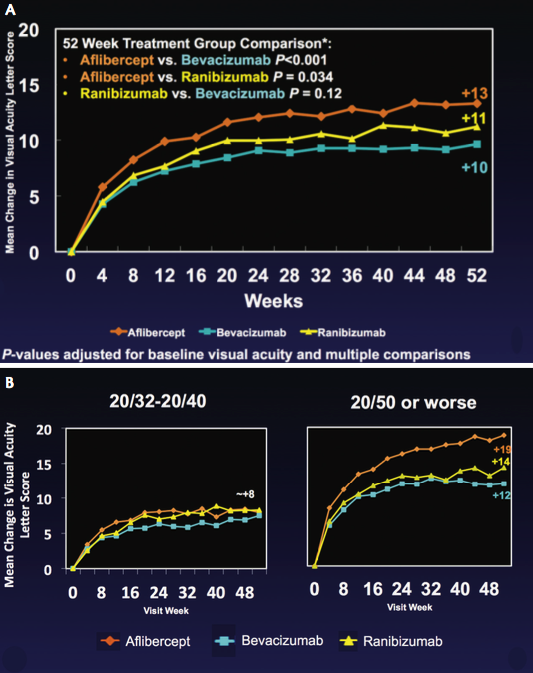
Figure. Mean change in visual acuity letter score. One-year results from all DRCR.net Protocol T (all participants; A). One-year baseline BCVA results from DRCR.net Protocol T subgroup analysis (B). Graphs courtesy of DRCR.net.
DME
DME is a leading cause of vision loss in people with DR. In the early 1980s, the ETDRS study defined the role of laser photocoagulation in reducing the risk of vision loss from swelling in the macula. However, substantial gains were rarely seen.9 Within the past 5 years, pivotal clinical trials have established the superior outcomes of serial intravitreal anti-VEGF injections compared with focal or grid laser photocoagulation, independent of the agent utilized. Specifically, the BOLT trial and the Diabetic Retinopathy Clinical Research Network (DRCR.net) Protocol T study provide the best evidence for bevacizumab.10-11 The RISE and RIDE studies demonstrated the benefit of ranibizumab, and VIVID and VISTA evaluated aflibercept.12,13 As a result of these trials, serial intravitreal anti-VEGF injections have become the standard of care for individuals with center-involved DME and visual compromise.
With multiple drug options and substantial cost differences, the DRCR.net Protocol T study was developed to directly compare serial intravitreal injections of bevacizumab, ranibizumab, and aflibercept in 660 subjects with center-involved DME over 1 year. Treatment algorithms dictated therapy as determined by visual acuity and central retinal thickness on optical coherence tomography (OCT). Application of focal or grid laser was permitted beyond week 24 if DME was not improving. All three anti-VEGF agents demonstrated improvements in visual acuity (Figure, A). According to a prespecified subgroup analysis, individuals with worse initial visual acuity (≤ ETDRS 20/50) had a statistically significantly better response to the use of aflibercept in terms of visual acuity, OCT thickness, and frequency of laser application (Figure, B). This benefit of aflibercept was also clinically meaningful, with an increased likelihood of gaining 3 lines compared with bevacizumab or ranibizumab.11 Two-year outcomes will be examined to determine whether this effect is persistent and to help guide future clinical care.
PDR
With neovascularization comes increased risk of visual acuity loss from complications such as vitreous hemorrhage and tractional retinal detachment (TRD). Fifty percent of individuals with high-risk characteristics experience severe visual acuity loss within 5 years of diagnosis with high-risk PDR.1 PRP permanently reduces that risk, but approximately 5% of eyes with PDR develop severe vision loss after PRP, thus necessitating vitrectomy.14 Furthermore, systematic ablation of the peripheral retina can induce complications such as pupillary and accommodative abnormalities, choroidal and retinal detachment, vision loss from macular edema or misplaced burn, and damage to peripheral or night vision. Historically, the benefits of laser have outweighed the risks in the most advanced stages of diabetic damage (ie, high-risk PDR).
Recent evidence suggests that serial intravitreal anti-VEGF injections can prevent PDR onset, and repeated use may have durable effects. Eyes in the DRCR.net Protocol I study receiving repeated ranibizumab injections had a lower likelihood of requiring PRP or developing vitreous hemorrhage than eyes that received steroid injections or either focal or grid laser.15 The risk of DR worsening is less likely and the chance of DR improvement is more likely in eyes receiving anti-VEGF injections than in eyes receiving laser.16
Eyes that have vitreous hemorrhage that precludes adequate laser have traditionally had two options: wait for the hemorrhage to clear and attempt more laser, or perform vitrectomy. The timing of clinic visits to apply laser is logistically challenging, and the application of PRP with concurrent vitreous hemorrhage can be technically difficult. The DRCR.net Protocol N study examined the utility of intravitreal ranibizumab to facilitate clearance of vitreous hemorrhage and permit PRP laser compared with intravitreal saline. The 1-year results demonstrated low rates of vitrectomy in both arms (38% overall; 35% in the ranibizumab arm vs. 41% in the saline arm; P > .05) with modest improvement in PRP completion rates (55% ranibizumab vs. 42% saline; P = .04). Recurrent vitreous hemorrhage was noted in 6% of eyes with ranibizumab versus 17% with saline (P = .01). No difference was observed in mean visual acuity outcomes.17,18 As a result, there is limited evidence supporting the use of anti-VEGF injections for nonclearing vitreous hemorrhages.
Due to the risk of worsening macular edema with PRP laser treatment, several studies have examined the use of anti-VEGF injections at the time of PRP application. In patients with concurrent DME who have PRP applied, intravitreal anti-VEGF injections given alongside PRP laser demonstrate superior visual and structural outcomes when compared with PRP alone or with PRP and focal or grid laser.19-22
Because anti-VEGF injections inhibit neovascularization and patients with PDR undergoing PRP have better outcomes when laser is combined with anti-VEGF injections, could the need for PRP be completely omitted with serial anti-VEGF injections? The recently published DRCR.net Protocol S study prospectively examined outcomes comparing PRP treatment with intravitreal ranibizumab for eyes with PDR.23 Nearly 400 eyes were randomized, and primary outcomes were measured at 2 years. Intravitreal ranibizumab was administered up to every 4 weeks in the ranibizumab group and was permitted as needed for DME in the PRP group (53% of eyes). Eyes that received ranibizumab by study algorithm demonstrated visual acuity outcomes that were noninferior to PRP, had reduced OCT thickness, and had less need for vitrectomy surgery.23 Whether the results of this trial can be replicated in the real world remains to be seen. Protocol S gives clinicians security in implementing initial anti-VEGF treatment for DME when concurrent PDR is present. At this time, the effect of discontinuing anti-VEGF therapy is unknown, but 60% of patients with PDR in the RISE and RIDE extension study did not have recurrence.24 The clinical application of this study’s findings will be variable and largely dictated by the adequacy of follow-up and relationship between provider and patient. The 5-year results and cost-effectiveness analysis will shape practice patterns as individual physicians determine what is best in their practice settings.
Uncomplicated DR
If features of advanced DR can be reduced with intravitreal anti-VEGF therapy, could there be rationale for using it at an earlier stage? Investigators using serial anti-VEGF injections for DME observed improvements in features of DR beyond regression of neovascularization. Analysis of the RISE and RIDE phase 3 clinical trials using ranibizumab for DME demonstrated a significant reduction in progression of retinopathy in subjects receiving monthly treatment over 2 years compared with eyes treated with laser alone.12,16 Much interest exists in preventing advanced stages of DR with anti-VEGF injections, and several clinical trials aimed at examining the robustness of this effect are anticipated in the near future. A careful balance optimizing timing of therapy to offset risks of injections and costs of drugs will be necessary.
Surgical Adjunct
Vitrectomy is considered for eyes with complications of DR such as TRD and nonclearing vitreous hemorrhage. PRP laser should always be applied to attached retina preoperatively when available due to the high likelihood of improvement with time and the benefit of retinopexy.17,18 Complications of surgery include early and delayed recurrent vitreous hemorrhage due to persistent ocular neovascularization. Several studies have examined the role of perioperative anti-VEGF injections on surgical outcomes. Preoperative bevacizumab has been associated with decreased duration of surgery, fewer retinal breaks, less intraoperative bleeding, less endodiathermy application, and a lower likelihood of early postoperative recurrent vitreous hemorrhage.25-32
CONCLUSION
Anti-VEGF treatment is a truly revolutionary breakthrough for managing complications of DR. The pendulum has already swung away from focal or grid laser, a standard of care for DME that had persisted for decades, in favor of intravitreal anti-VEGF injections. Furthermore, these injections provide superior outcomes when used to facilitate clearance of vitreous hemorrhage, when combined with PRP, and before vitrectomy. We may be on the cusp of a similar paradigm shift for treatment of PDR or uncomplicated DR. The temporary effect of intravitreal injections, combined with the unreliable nature of patient follow-up, mandates that caution be exercised in interpreting results of Protocol S. At this time, PRP should be considered standard of care due to its durable benefit, especially in circumstances in which patient compliance may be poor or financial limitations exist. Long-term data and real-world experience will fine-tune our ability to optimize the translation of these groundbreaking developments into clinical practice. n
1. Photocoagulation treatment of proliferative diabetic retinopathy: the second report of diabetic retinopathy findings. Ophthalmology. 1978;85(1):82-106.
2. Photocoagulation treatment of proliferative diabetic retinopathy: relationship of adverse treatment effects to retinopathy severity. Diabetic Retinopathy Study report no. 5. Dev Ophthalmol. 1981;2:248-261.
3. Photocoagulation treatment of proliferative diabetic retinopathy. Clinical application of Diabetic Retinopathy Study (DRS) findings, DRS report number 8. The Diabetic Retinopathy Study Research Group. Ophthalmology. 1981;88(7):583-600.
4. Davis MD, Fisher MR, Gangon RE, et al. Risk factors for high-risk proliferative diabetic retinopathy and severe vision loss: Early Treatment Diabetic Retinopathy Study Report #18. Invest Ophthalmol Vis Sci. 1998;39(2):233-252.
5. Brown LF, Detmar M, Claffey K, et al. Vascular permeability factor/vascular endothelial growth factor: a multifunctional angiogenic cytokine. In: Goldberg ID, Rosen EM, eds. Regulation of Angiogenesis. Basel, Switzerland: Birkauser Verlag; 1997:233-269.
6. Matthews MK, Merges C, McLeod DS, et al. Vascular endothelial growth factor and vascular permeability changes in human diabetic retinopathy. Invest Ophthalmol Vis Sci. 1997;38(13):2729-2741.
7. Schweiki D, Itin A, Soffer D, Keshet E. Vascular endothelial growth factor induced by hypoxia may mediate hypoxia-initiated angiogenesis. Nature. 1992;359(6398):843-845.
8. Adamis AP, Miller JW, Bernal MT, et al. Increased vascular endothelial growth factor levels in the vitreous of eyes with proliferative diabetic retinopathy. Am J Ophthalmol. 1994;118(4):445-450.
9. Photocoagulation for diabetic macular edema. Early Treatment Diabetic Retinopathy Study report number 1. Early Treatment Diabetic Retinopathy Study research group. Arch Ophthalmol. 1985; 103(12):1796-1806.
10. Rajendram R, Fraser-Bell S, Kaines A, et al. A 2-year prospective randomized controlled trial of intravitreal bevacizumab or laser therapy (BOLT) in the management of diabetic macular edema: 24-month data: report 3. Arch Ophthalmol. 2012;130(8):972-979.
11. Wells JA, Glassman AR, Ayala AR, et al; Diabetic Retinopathy Clinical Research Network. Aflibercept, bevacizumab, or ranibizumab for diabetic macular edema. N Engl J Med. 2015; 372(13):1193-1203.
12. Brown DM, Nguyen QD, Marcus DM, et al. Long-term outcomes of ranibizumab therapy for diabetic macular edema: the 36-month results from two phase III trials: RISE and RIDE. Ophthalmology. 2013;120(10):2013-2022.
13. Korobelnik JF, Do DV, Schmidt-Erfurth U, et al. Intravitreal aflibercept for diabetic macular edema. Ophthalmology. 2014;121(11):2247-2254.
14. Flynn HJ, Chew E, Simons B, et al. Pars plana vitrectomy in the early treatment diabetic retinopathy study: ETDRS report number 17. Ophthalmology. 1992;99(9):1351-1357.
15. Bressler SB, Qin H, Melia M, et al; for the Diabetic Retinopathy Clinical Research Network. Exploratory analysis of effect of intravitreal ranibizumab or triamcinolone on worsening of diabetic retinopathy in a randomized clinical trial. JAMA Ophthalmol. 2013; 131(8):1033-1040.
16. Ip MS, Domalpally A, Hopkins JJ, et al. Long-term effects of ranibizumab on diabetic retinopathy severity and progression. Arch Ophthalmol. 2012;130(9):1145-1152.
17. Diabetic retinopathy clinical research network. Randomized clinical trial evaluating intravitreal ranibizumab or saline for vitreous hemorrhage from proliferative diabetic retinopathy. JAMA Ophthalmol. 2013;131(3):283-293.
18. Bhavsar AR, Torres K, Bressler NM, et al; for the Diabetic Retinopathy Clinical Research Network. Evaluation of results 1 year following use of short-term ranibizumab for vitreous hemorrhage due to proliferative diabetic retinopathy. JAMA Ophthalmol. 2014;132(7):889-890.
19. Filho JA, Messias A, Almeida FP, et al. Panretinal photocoagulation (PRP) versus PRP plus intravitreal ranibizumab for high-risk proliferative diabetic retinopathy. Acta Ophthalmol. 2011;89(7):e567-e572.
20. Googe J, Brucker AJ, Bressler NM, et al. Randomized trial evaluating short-term effects of intravitreal ranibizumab or triamcinolone acetonide on macular edema after focal/grid laser for diabetic macular edema in eyes also receiving panretinal photocoagulation. Retina. 2011;31(6):1009-1027.
21. Cho WB, Oh SB, Moon JW, Kim HC. Panretinal photocoagulation combined with intravitreal bevacizumab in high-risk proliferative diabetic retinopathy. Retina. 2009;29(4):516-522.
22. Mirshahi A, Roohipoor R, Lashay A, et al. Bevacizumab augmented retinal laser photocoagulation in proliferative diabetic retinopathy: a randomized double-masked clinical trial. Eur J Ophthalmol. 2008;18(2):263-269.
23. Diabetic Retinopathy Clinical Research Network. Panretinal photocoagulation vs intravitreous ranibizumab for proliferative diabetic retinopathy: a randomized trial. JAMA. 2015; 314(20):2137-2146.
24. Ho AC, Zhang J, Ehrlich JS. Ranibizumab for diabetic macular edema: long-term open label extension of the phase III RIDE and RISE trials. Paper presented at: Association for Research in Vision and Ophthalmology Annual Meeting; May 3-8, 2014; Orlando, FL.
25. Rizzo S, Genovesi-Ebert F, Di Bartolo E, et al. Injection of intravitreal bevacizumab (Avastin) as a preoperative adjunct before vitrectomy surgery in the treatment of severe proliferative diabetic retinopathy (PDR). Graefes Arch Clin Exp Ophthalmol. 2008;246(6):837-842.
26. Modarres M, Nazari H, Falavarjani KG, et al. Intravitreal injection of bevacizumab before vitrectomy for proliferative diabetic retinopathy. Eur J Ophthalmol. 2009;19(5):848-852.
27. Hernandez-Da Mota SE, Nunez-Solorio SM. Experience with intravitreal bevacizumab as a preoperative adjunct in 23-G vitrectomy for advanced proliferative diabetic retinopathy. Eur J Ophthalmol. 2010;20:1047-1052.
28. di Lauro R, De Ruggiero P, di Lauro MT, Romano MR. Intravitreal bevacizumab for surgical treatment of severe proliferative diabetic reitnopathy. Graefes Arch Clin Exp Ophthalmol. 2010;248(6):785-791.
29. El-Batamy AM. Intravitreal bevacizumab as an adjunctive therapy before diabetic vitrectomy. Clin Ophthalmol. 2008;2(4):709-716.
30. Faravash MS, Majidi AR, Roohipoor R, Ghassemi F. Preoperative injection of intravitreal bevacizumab in dense diabetic vitreous hemorrhage. Retina. 2011;31(7):1254-1260.
31. Ahmadieh H, Shoeibi N, Entezari M, Monshizadeh R. Intravitreal bevacizumab for prevention of early postvitrectomy hemorrhage in diabetic patients: a randomized clinical trial. Ophthalmology. 2009;116(10):1943-1948.
32. Ahn J, Woo SJ, Chung H, Park KH. The effect of adjunctive intravitreal bevacizumab for preventing postvitrectomy hemorrhage in proliferative diabetic retinopathy. Ophthalmology. 2011;118(11):2218-2226.
Andrew M. Hendrick, MD
• assistant professor of ophthalmology, vitreoretinal surgery and diseases, Emory Eye Center, in Atlanta, Ga.
• financial interest: none acknowledged
• ahendrick@emory.edu
Michael S. Ip, MD
• co-director of the Fundus Photograph Reading Center in the Department of Ophthalmology and Visual Sciences at the University of Madison-Wisconsin in Madison, Wisc.
• member of the Retina Today editorial advisory board
• financial interest: none acknowledged
• msip@wisc.edu









_1773249222.png?auto=compress,format&w=75)










