Protocol T: 2-Year Data Released
All three anti-VEGF agents evaluated in the Diabetic Retinopathy Clinical Research Network Protocol T trial were effective in treating diabetic macular edema (DME) at 2 years, according to a study update published in Ophthalmology.1 Some significant differences between the evaluated anti-VEGF agents seen at the 1-year point were no longer present at year 2.
In patients with baseline visual acuity of 20/50 or worse, aflibercept (Eylea, Regeneron) treatment showed superior visual acuity improvement compared with bevacizumab (Avastin, Genentech), but the superiority of aflibercept over ranibizumab (Lucentis, Genentech) noted at the 1-year time point of the study was no longer seen; no difference in visual acuity result was observed between the ranibizumab and bevacizumab treatment arms for patients with baseline visual acuity of 20/50 or worse. In patients with baseline visual acuity of 20/32 to 20/40, all three anti-VEGF agents resulted in similar visual acuity outcomes.
Researchers randomly assigned 660 patients with DME to treatment with 2.0 mg aflibercept, 1.25 mg compounded bevacizumab, or 0.3 mg ranibizumab. Participants received laser therapy if DME persisted and did not improve beyond 6 months. During year 1 of the study, patient visits occurred every 4 weeks, and the interval was extended up to every 4 months thereafter if visual acuity and macular thickness were stable.
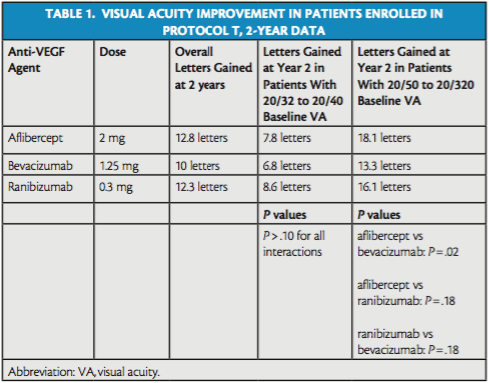
Overall, 2-year mean visual acuity letter score improved by 12.8 letters in the aflibercept arm, 10.0 letters in the bevacizumab arm, and 12.3 letters in the ranibizumab arm (Table 1). In patients with baseline visual acuity of 20/32 to 20/40, mean improvement at 2 years was 7.8 letters in the aflibercept group, 6.8 letters in the bevacizumab group, and 8.6 letters in the ranibizumab group (P > .10 for pairwise comparisons). In patients with baseline visual acuity of 20/50 to 20/320, mean improvement at 2 years was 18.1 letters in the aflibercept arm, 13.3 letters in the bevacizumab arm, and 16.1 letters in the ranibizumab arm (aflibercept vs. bevacizumab, P = .02; aflibercept vs. ranibizumab, P = .18; ranibizumab vs. bevacizumab, P = .18).
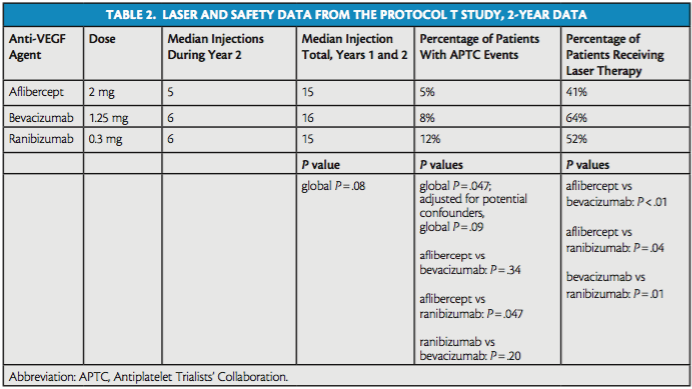
Focal or grid laser was administered in 41%, 64%, and 52% of patients in the aflibercept, bevacizumab, and ranibizumab groups, respectively (aflibercept vs. bevacizumab, P < .01; aflibercept vs. ranibizumab, P = .04; bevacizumab vs. ranibizumab, P = .01; Table 2).
The study authors also assessed safety considerations, including the median number of injections required in each arm during year 2 and during the entire trial and the number of Antiplatelet Trialists’ Collaboration (APTC) events in each arm (Table 2).
The median number of injections did not differ significantly among groups during year 2 nor during the entire trial. The median number of injections during year 2 of the study was five in the aflibercept group, six in the bevacizumab group, and six in the ranibizumab group; during the entire course of the study, the median number of injections in those groups, respectively, was 15, 16, and 15.
The researchers found that APTC events occurred at a rate of 5% in the aflibercept arm, 8% in the bevacizumab arm, and 12% in the ranibizumab arm. There was a significant difference in this measure between the aflibercept and ranibizumab arms (P = .047), but the difference was not significant between the aflibercept and bevacizumab arms (P = .34) or the ranibizumab and bevacizumab arms (P = .20). The study authors noted that these data on APTC events had not been demonstrated consistently in previously reported clinical trials, and that the higher rate of APTC events in the ranibizumab arm warranted continued evaluation in future trials
1. Wells JA, Glassman AR, Ayala A, et al. Aflibercept, bevacizumab, or ranibizumab for diabetic macular edema: two-year results from the comparative effectiveness randomized clinical trial [published online ahead of print February 27, 2016]. Ophthalmology.
Biomarker for Uveal Melanoma Metastasis Identified
Class 1 uveal melanomas with high levels of PRAME mRNA were more likely to metastasize than those with low levels of PRAME mRNA, according to a press release detailing a retrospective study slated to be published in Clinical Cancer Research.1
Uveal melanomas are classified as class 1 or class 2 tumors, and class 1 tumors have a much lower chance of metastasis than class 2 tumors. However, about 10% of class 1 uveal melanomas metastasize.
Researchers in this study performed a genome-wide analysis of mRNA isolated from 13 uveal melanomas, five of which had metastasized, and found that the metastatic tumors highly overexpressed PRAME mRNA. Further quantitative analysis showed similar results. In that analysis, seven of seven class 1 tumors that metastasized overexpressed PRAME mRNA, and 16 of 19 class 1 tumors that did not metastasize expressed minimal levels of PRAME mRNA.
The estimated 5-year rate of metastasis in class 1 tumors with low PRAME mRNA expression was 0%; in class 1 tumors with high PRAME mRNA expression, the estimated rate was 38%.
“The data imply that patients with class 1 uveal melanomas with increased PRAME expression should be managed differently than patients with class 1 uveal melanomas without PRAME expression,” J. William Harbour, MD, said in the press release. “They should be monitored more closely for metastatic disease, and they should be considered for clinical trials of adjuvant therapy.” n
1. New biomarker identifies uveal melanoma patients at high risk for metastasis [press release]. American Association for Cancer Research; March 1, 2016.









_1773249222.png?auto=compress,format&w=75)










