

Diabetic vitreous hemorrhage (VH) secondary to proliferative diabetic retinopathy (PDR) is one of the main causes of severe vision loss in patients with diabetes. Since Machemer performed the first pars plana vitrectomy (PPV) for VH in 1970, monumental improvements in technology have made the procedure more routine.1 Despite these major advances, maintaining hemostasis is often the most challenging aspect of complex vitrectomy for complications of PDR.
AT A GLANCE
• Maintaining hemostasis is a challenging aspect of complex vitrectomy for complications of PDR.
• Standard bimanual intraoperative cauterization techniques take time to exchange instruments, add to equipment costs, increase the number of incisions, and prolong surgical setup time.
• A novel technique described here uses the endodiathermy probe to both clear and treat active bleeding during diabetic vitrectomy cases, minimizing issues inherent in a bimanual approach.
Diabetic patients are frequently coagulopathic, and, even with preoperative anti-VEGF therapy, persistent bleeding can result in poor outcomes by inhibiting visualization during membrane peeling or by limiting visual acuity postoperatively. Persistent visually significant VH (Figure 1) has been reported after one-third of cases, and it can necessitate repeating the procedure in up to 13% of cases.2 Risk factors for postoperative day 1 hemorrhage include incomplete preoperative panretinal photocoagulation, postoperative hypotony, young patient age, and phakic status.2
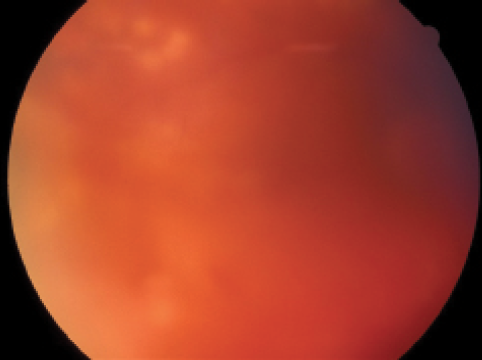
Figure 1. Postoperative VH after PPV.
A NEW APPROACH
In standard intraoperative cauterization techniques, two steps are required: (1) aspiration or reflux with the vitrector or extrusion cannula to clear the hemorrhage, followed by (2) contact with an endodiathermy probe or direct endolaser photocoagulation. The latter is a useful approach for small bleeds, but it can be challenging to achieve effective cautery in larger vessels. The process of exchanging instruments allows time for additional bleeding, which may block access to neovascular membranes, prolong the duration of tamponade pressure, increase total operative time, and cause surgeon frustration. Bimanual techniques, aided by a chandelier-style light source, add to equipment costs, increase the number of incisions, and prolong surgical setup time.
These problems can be minimized with a novel single-instrument technique. This method uses the endodiathermy probe to both clear and treat active bleeding, transforming the device into a multifunctional tool during diabetic vitrectomy cases. The steps of the procedure are as follows:
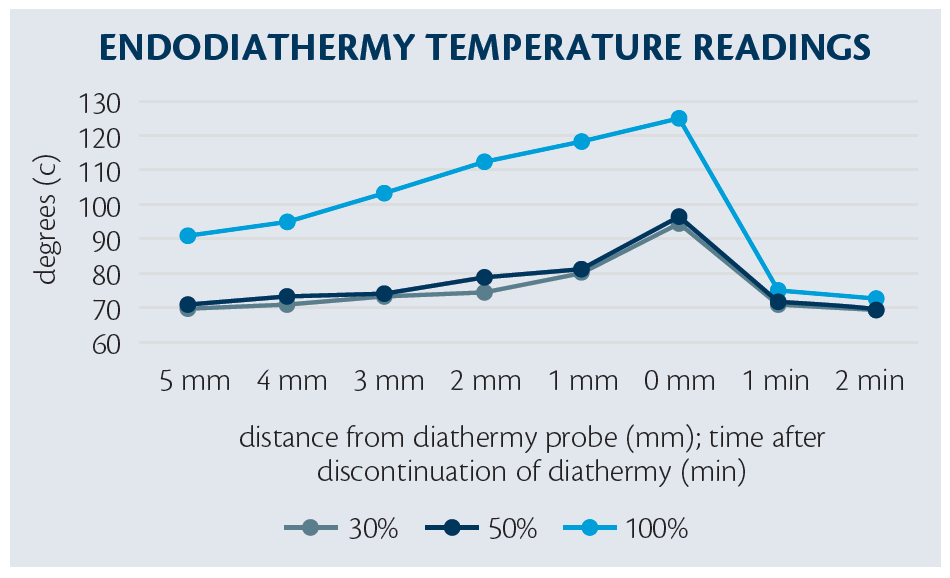
Figure 2. To achieve temperatures high enough for vaporization, diathermy power must be maximized.
Step 1. Increase diathermy power to the maximum level, approximately three times higher than standard settings (eg, 100% on the Constellation Vision System [Alcon]). Total output is under proportional control by the surgeon, and low power levels can still be achieved through less depression of the footpedal.
Step 2. Identify vessels with active hemorrhage and, if possible, relieve them of traction.
Step 3. If necessary, increase tamponade pressure (eg, to 60 mm Hg).
Step 4. Using the endodiathermy probe, hover above the hemorrhage and fully activate cautery; the vaporization effect will create a bubble stream (Video 1) that will expose the naked vessel.
WATCH IT NOW
Video 1. SINGLE-INSTRUMENT CAUTERIZATION TECHNIQUE: STEP 4
After increasing diathermy power to the maximum level (Step 1), identifying vessels with active hemorrhage (Step 2), and increasing tamponade pressure, if necessary (Step 3), hover the endodiathermy probe above the hemorrhage and fully activate cautery. A bubble stream created by the vaporization effect will expose the naked vessel.
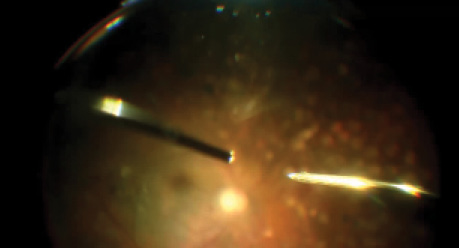
Step 5. Reduce diathermy power by decreasing footpedal depression and approach the bleeding site, making gentle contact to achieve cauterization (Video 2). Repeat as needed at additional locations.
Step 6. Decrease intraocular pressure (IOP) to normal operating levels to verify hemostasis. At the conclusion of the case, consider decreasing to a more physiologic IOP, such as 12 mm Hg, to help identify any additional areas of bleeding that could persist postoperatively if not treated (Video 3).
Step 7. Apply endolaser around the area of cauterization to seal any possible retinal microbreak.
TWO TOOLS IN ONE
We investigated the thermal properties of noncontact endodiathermy with in vitro simulations in balanced salt solution.3 We determined that, in order to achieve temperatures high enough for vaporization, the diathermy power must be maximized (Figure 2). At these high levels, caution should be used, and contact with tissue should be avoided. By proportionally reducing the power to approximately 30% of maximum, safe cauterization can be achieved. With this approach, therefore, the diathermy probe can serve two functions, depending on power level.
WATCH IT NOW
Video 2. SINGLE-INSTRUMENT CAUTERIZATION TECHNIQUE: STEP 5
Next, reduce diathermy power and approach the bleeding site, making gentle contact to achieve cauterization.
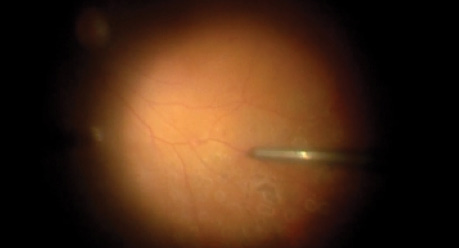
Using the minimum amount of diathermy necessary can help avoid collateral damage to the retina, which can result in atrophic holes, and to large vessels, which can result in vascular occlusions and ischemia. Placing laser barricade around the area of cautery will ensure that potential retinal breaks do not progress. It should be noted that vaporization will clear active, fresh hemorrhage. Clotted blood is less likely to be displaced by this technique.
Standard IOPs of 25 mm Hg to 30 mm Hg are above the normal physiologic range. Such artificial circumstances can mask bleeding vessels, and low IOP after the conclusion of surgery can precipitate hemorrhage from these sites. Reducing the infusion pressure at the end of the case helps to ensure that all potential sources of VH are identified for appropriate treatment with the above technique.
WATCH IT NOW
CONCLUSION
Performing PPV for complications of PDR requires considerable surgical skill. Even with small-gauge instruments, meticulous dissection, and adjuvant anti-VEGF therapy, intraoperative and postoperative bleeding are challenges that can result in unfavorable outcomes. Using the diathermy probe as a multifunctional tool can help decrease surgical time and tilt the battle of hemostasis in your favor (Video 4).
1. Machemer R, Buettner H, Norton EW, Parel JM. Vitrectomy: a pars plana approach. Trans Am Acad Ophthalmol Otolaryngol. 1971;75(4):813-820.
2. Khuthaila MK, Hsu J, Chiang A, et al. Postoperative vitreous hemorrhage after diabetic 23-gauge pars plana vitrectomy. Am J Ophthalmol. 2013;155(4)757-763.
3. Pitcher JD 3rd, Gupta OP. Hi-power, non-contact endodiathermy to clear vitreous hemorrhage from neovascularization prior to cautery in vitrectomy for proliferative diabetic retinopathy: a novel technique. Paper presented at: Vail Vitrectomy 2016; February 20-23, 2016; Vail, CO.
Omesh P. Gupta, MD, MBA
• assistant professor of ophthalmology at Thomas Jefferson University Hospital, an attending surgeon at Wills Eye Hospital, and a partner at Mid Atlantic Retina, all in Philadelphia, Pa.
• financial interest: none acknowledged
• ogupta@midatlanticretina.com
John D. Pitcher III, MD
• retina specialist and partner, Eye Associates of New Mexico; assistant clinical professor of ophthalmology at The University of New Mexico, both in Albuquerque, N.M.
• financial interest: none acknowledged
• jdpitcher@eyenm.com









_1773249222.png?auto=compress,format&w=75)










