Polypoidal choroidal vasculopathy (PCV) is characterized by nodular dilatations at the terminus of a vascular network. These dilatations appear as orange nodules on fundus examination, but they are best seen on indocyanine green angiography (ICGA). Patients with PCV may present with hemorrhage and/or exudation in the macula, similar to those with wet AMD.
DIAGNOSIS
ICGA is widely considered the standard for differentiating PCV from typical wet AMD. However, ICGA provides limited depth resolution and is invasive. Several groups have evaluated the possibility of using OCT to differentiate PCV from typical wet AMD (Figure).1-6 This information is of clinical significance because the management of PCV varies from that of AMD and may involve laser and photodynamic therapy (PDT), in addition to anti-VEGF therapy.
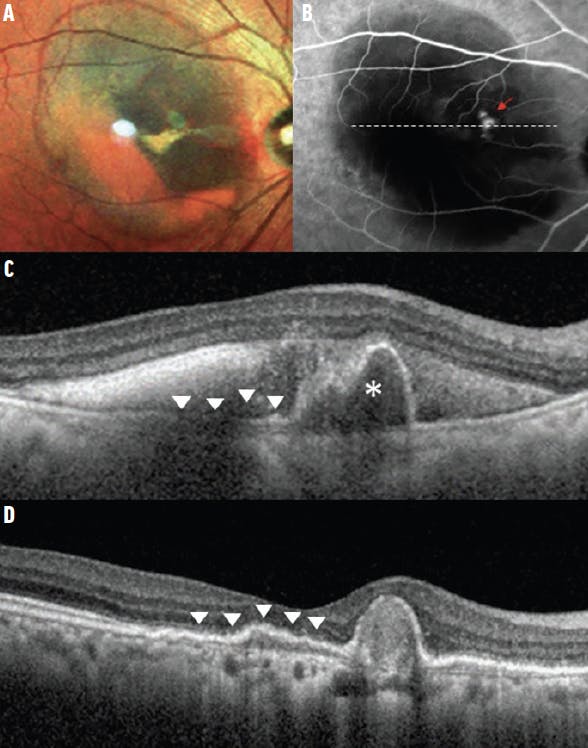
Figure. PCV at presentation (A-C) and following treatment (D). Serosanguinous maculopathy with evidence of subretinal hemorrhage is evident in the multicolor image (A). In the corresponding ICGA, focal hyperfluorescent nodules correspond to the polypoidal lesions (red arrow, B). The branching neovascular network cannot be discerned on ICGA due to masking from overlying hemorrhage. The cross-sectional OCT (corresponding to the dashed white line on the ICGA) transects the polypoidal lesion (C). A sharp-peaked PED with a sub-RPE ring-like structure (asterisk) corresponds to the focal hyperfluorescent nodule on ICGA. The PED is notched and multilocular. A double-layer sign (arrowheads) can also be seen, which corresponds to where the branching neovascular network resides (C and D). SRF is present and is accompanied by subretinal blood that appears as hyperreflective material. After three monthly anti-VEGF injections, the hemorrhage and SRF have resolved (D). The content of the PED becomes densely hyperreflective and the sub-RPE ring-like structure is not discernible, suggesting the polypoidal lesion is no longer perfused. Note that the double-layer sign remains.
Several features related to the alteration of the retinal pigment epithelium (RPE) contour have been associated with PCV (Table). More recently, the presence of a thickened choroid and lack of drusen have been suggested as useful clues to differentiate PCV from typical wet AMD. The Asia-Pacific Ocular Imaging Society's (APOIS) PCV workgroup reported a diagnostic set comprising three major criteria: sub-RPE ring-like lesion, sharp-peaked pigment epithelial detachment (PED), and complex RPE elevation on enface OCT.2 Using these criteria, treatment-naïve eyes with PCV can be differentiated from wet AMD with 75% sensitivity and 91% specificity.2
MANAGEMENT
Although a combination of anti-VEGF therapy and PDT can achieve high rates of closure of polypoidal lesions based on angiographic assessment, clinical trials suggest that anti-VEGF monotherapy is also efficacious in achieving visual improvement, and that PDT may be reserved for the subset of patients who show suboptimal response to anti-VEGF therapy.1,7,8 Therefore, the ability to differentiate PCV from typical wet AMD may be even more important in eyes that develop persistent fluid following anti-VEGF therapy than in eyes with fluid at initial presentation.
The APOIS PCV workgroup evaluated non-ICGA imaging features in eyes with suboptimal response to anti-VEGF loading and reported that PCV eyes can be differentiated from typical wet AMD based on a combination of the following three criteria: sub-RPE ring-like lesion, sharp-peaked PED, and the presence of an orange nodule, with 65% sensitivity and 82% specificity.2 Treatment strategies to consider in eyes that meet these criteria (indicating PCV) include switching the anti-VEGF agent and adding PDT.2
During the retreatment phase, recurrent disease activity may arise from the polypoidal lesion(s), the branching neovascular network, or a combination of both. Even after resolution of subretinal fluid (SRF) and/or intraretinal fluid (IRF), polypoidal lesions may still be present. These polypoidal lesions may be considered inactive, but they may continue to pose a threat to vision long-term due to the risk of recurrence and hemorrhage, especially if anti-VEGF therapy is not maintained. Therefore, while SRF and IRF are the main imaging criteria used for retreatment decisions for typical wet AMD, evaluating the status of polypoidal lesions remains important.
Without ICGA, several OCT-based features (densely hyperreflective content of PED, absence of SRF, and absence of a sub-RPE ring) can be helpful in assessing whether polypoidal lesions have closed. In eyes with residual perfused polypoidal lesions, a treat-and-extend approach may be preferable to a prn approach. The addition of an occlusive therapy element, such as PDT or focal laser for extrafoveal polypoidal lesions, remains an important part of the treatment armamentarium for patients who are reluctant to adhere to long-term anti-VEGF therapy or in whom disease from the polypoidal lesions activity (particularly hemorrhage and increasing PEDs) is not adequately controlled.
CHOROIDAL BACKGROUND
Pachychoroid phenotype refers to eyes with a congested choroid, which typically appears as increased thickness and exhibits dilated vascular lumen in the Hallers vessels, as well as choroidal hyperpermeability. While some eyes with pachychoroid do not exhibit any complications (uncomplicated pachychoroid), several conditions reside on the pachychoroid spectrum. These include non-neovascular complications, such as pachychoroid pigment epitheliopathy, central serous chorioretinopathy, and peripapillary pachychoroid syndrome, and neovascular complications, such as pachychoroid neovasculopathy (PNV) and PCV. Eyes with PNV and PCV typically lack a background of soft drusen. Therefore, pachychoroid has been suggested as an alternative pathogenic mechanism for macular neovascularization; in addition, PCV may evolve from PNV.8,9 However, more longitudinal data are necessary to support this theory.
Some investigators have suggested that eyes with pachychoroid background may respond differently to therapy.10 For example, eyes with a thick choroid may respond less favorably to anti-VEGF monotherapy, while eyes with choroidal hyperpermeability may respond to PDT.10 However, these reports are limited to small clinical series. In a post-hoc analysis of the EVEREST study, which compared ranibizumab (Lucentis, Genentech/Roche) with or without PDT, subfoveal choroidal thickness was not found to significantly influence visual outcomes.11
Less commonly, PCV may also develop over an area affected by choroidal nevus or at the edge of a posterior staphyloma. Alteration in choroidal blood flow and hemodynamics may also play a role in these eyes, but these eyes should be differentiated from those with pachychoroid background.
CLINICAL IMPACT
Recent research on PCV has led to important clinically relevant lessons that reduce our reliance on ICGA. Although anti-VEGF monotherapy can be effective in most eyes with PCV, differentiating this lesion subtype from typical wet AMD remains important for planning long-term treatment strategies that may involve laser and PDT. In addition to SRF and IRF, which are commonly used for assessing disease activity, PED-related imaging features may provide additional information regarding polypoidal lesion status during monitoring visits. Finally, research on choroidal features suggests that pachychoroid may be a novel pathogenic mechanism in eyes that lack drusen. Further studies are necessary to understand the long-term evolution of pachychoroid spectrum disorders.
1. Cheung CMG, Lai TYY, Ruamviboonsuk P, et al. Polypoidal choroidal vasculopathy: definition, pathogenesis, diagnosis, and management. Ophthalmology. 2018;125(5):708-724.
2. Cheung CMG, Lai TTY, Teo K, et al. Polypoidal choroidal vasculopathy: consensus nomenclature and non-ICGA diagnostic criteria from the Asia-Pacific Ocular Imaging Society (APOIS) PCV Workgroup. Ophthalmology. 2021;128(3):443-452.
3. De Salvo G, Vaz-Pereira S, Keane PA, Tufail A, Liew G. Sensitivity and specificity of spectral-domain optical coherence tomography in detecting idiopathic polypoidal choroidal vasculopathy. Am J Ophthalmol. 2014;158(6):1228-1238e1.
4. Liu R, Li J, Li Z, et al. Distinguishing polypoidal choroidal vasculopathy from typical neovascular age-related macular degeneration based on spectral domain optical coherence tomography. Retina. 2016;36(4):778-786.
5. Chaikitmongkol V, Kong J, Khunsongkiet P, et al. Sensitivity and specificity of potential diagnostic features detected using fundus photography, optical coherence tomography, and fluorescein angiography for polypoidal choroidal vasculopathy. JAMA Ophthalmol. 2019;137(6):661-667.
6. de Carlo TE, Kokame GT, Kaneko KN, Lian R, Lai JC, Wee R. Sensitivity and specificity of detecting polypoidal choroidal vasculopathy with en face optical coherence tomography and optical coherence tomography angiography. Retina. 2019;39(7):1343-1352.
7. Koh A, Lai TYY, Takahashi K, et al. Efficacy and safety of ranibizumab with or without verteporfin photodynamic therapy for polypoidal choroidal vasculopathy: a randomized clinical trial. JAMA Ophthalmol. 2017;135(11):1206-1213.
8. Lee WK, Iida T, Ogura Y, et al. Efficacy and safety of intravitreal aflibercept for polypoidal choroidal vasculopathy in the PLANET Study: a randomized clinical trial. JAMA Ophthalmol. 2018;136(7):786-793.
9. Cheung CMG, Lee WK, Koizumi H, Dansingani K, Lai TYY, Freund KB. Pachychoroid disease. Eye (Lond). 2019;33(1):14-33.
10. Chung SE, Kang SW, Lee JH, Kim YT. Choroidal thickness in polypoidal choroidal vasculopathy and exudative age-related macular degeneration. Ophthalmology. 2011;118(5):840-845.
11. Cheung CMG, Tan CS, Patalauskaite R, Margaron P, Lai TYY. Ranibizumab with or without verteporfin photodynamic therapy for polypoidal choroidal vasculopathy: predictors of visual and anatomical response in the EVEREST II study. Retina. 2021;41(2):387-392.













_1773249222.png?auto=compress,format&w=75)










