AT AN GLANCE
- The current diabetic retinopathy staging system relies on decades-old technology and does not harness the capabilities of modern retinal imaging.
- A new staging system based solely on OCT/OCT angiography overcomes many of the current system’s limitations.
- Barriers to adoption include the learning curve for OCT angiography interpretation and the cost of implementation.
Diabetic retinopathy (DR) remains the leading cause of blindness among adults of working age in developed countries, and the prevalence of DR continues to increase.1,2 This massive disease burden necessitates the development and implementation of effective and efficient strategies for screening, staging, and managing DR.
For more than 50 years, DR has been classified based on vascular abnormalities visible with conventional fundus photography.3 Although widely used, the current system relies on decades-old technology (ie, montaged 30° color fundus photographs) and does not harness the capabilities of modern retinal imaging. For this reason, current DR staging has many limitations, including the following:
- a limited field of view compared with modern photography that allows up to 200° in a single frame,
- a lack of angiography to define the extent of retinal ischemia or differentiate intraretinal microvascular abnormalities (IRMA) from neovascularization (NV), and
- no cross-sectional imaging to identify structural changes such as diabetic macular edema (DME) or areas of traction.4
Moreover, traditional DR staging is limited to clinically apparent abnormalities of the retinal vasculature—it does not address structural changes in the inner retina, such as retinal nerve fiber layer/ganglion cell layer thinning, vascular loss in the choriocapillaris/choroid, or vitreoretinal interface abnormalities such as tractional retinal detachment.
To account for advances in our understanding of DR, multiple people have called for the development of new DR staging systems.5-9 There is at least one large, multidisciplinary working group attempting to create one, although this group is still in preliminary discussions.8,10
A NEW WAY TO SEE
Over the past 2 decades, OCT has become an ubiquitous technology that enables visualization of retinal changes such as DME and atrophy. OCT angiography (OCTA) is a more recent advance that images both retinal structures and vascularity well beyond the ability of conventional fundus photography. We recently proposed a new staging system for DR based solely on OCT/OCTA scans (Figure 1).4 Our system leverages the current DR staging system but overcomes many of the limitations by visualizing the entire posterior pole with a single scan and by incorporating both angiographic and structural information. An OCTA-based system has a few other advantages, including an ability to:
- image the choroid and any tractional retinal detachment (TRD),
- differentiate between active versus quiescent proliferative DR (PDR), and
- register scans for direct comparison between visits.
This system encompasses all the clinically useful information needed for staging and managing DR.
The proposed OCTA-based system consists of six stages4:
No DR: This stage is defined as a normal widefield (WF) OCTA of the posterior pole.
Subclinical DR: This stage is defined as small areas of decreased vascular density without distinct vascular abnormalities (ie, microaneurysms, IRMA, or NV). With the current DR staging methods, eyes with subclinical DR are mistakenly labelled as normal, whereas OCTA clearly reveals signs of early disease.11
Nonproliferative DR (NPDR): The new staging system compresses mild and moderate NPDR into a single category that features microaneurysms and/or larger areas of capillary ischemia.
Severe NPDR: This is an important clinical threshold for clinicians to recognize because of the risk of progression to NV and its associated complications.
In the OCTA-based system, severe NPDR is replaced with a new category called preproliferative DR. This stage is defined by OCTA evidence of IRMA but not NV. Because IRMAs have been shown to be the vascular precursor to NV,12 IRMAs are the defining feature of the preproliferative stage.
PDR: This stage is defined by OCTA evidence of NV (Figure 2).13 Unlike fluorescein angiography, OCTA captures vascular flow in a binary fashion (either present or absent) and lacks the ability to evaluate dynamic transit of dye and leakage over time. However, OCTA’s ability to correlate vascular information with structural details facilitates the differentiation of NV from IRMA based on the location of the lesion (preretinal vs intraretinal, respectively). Moreover, OCTA can distinguish active versus quiescent NV based on the caliber and density of vessels within the neovascular complex.13
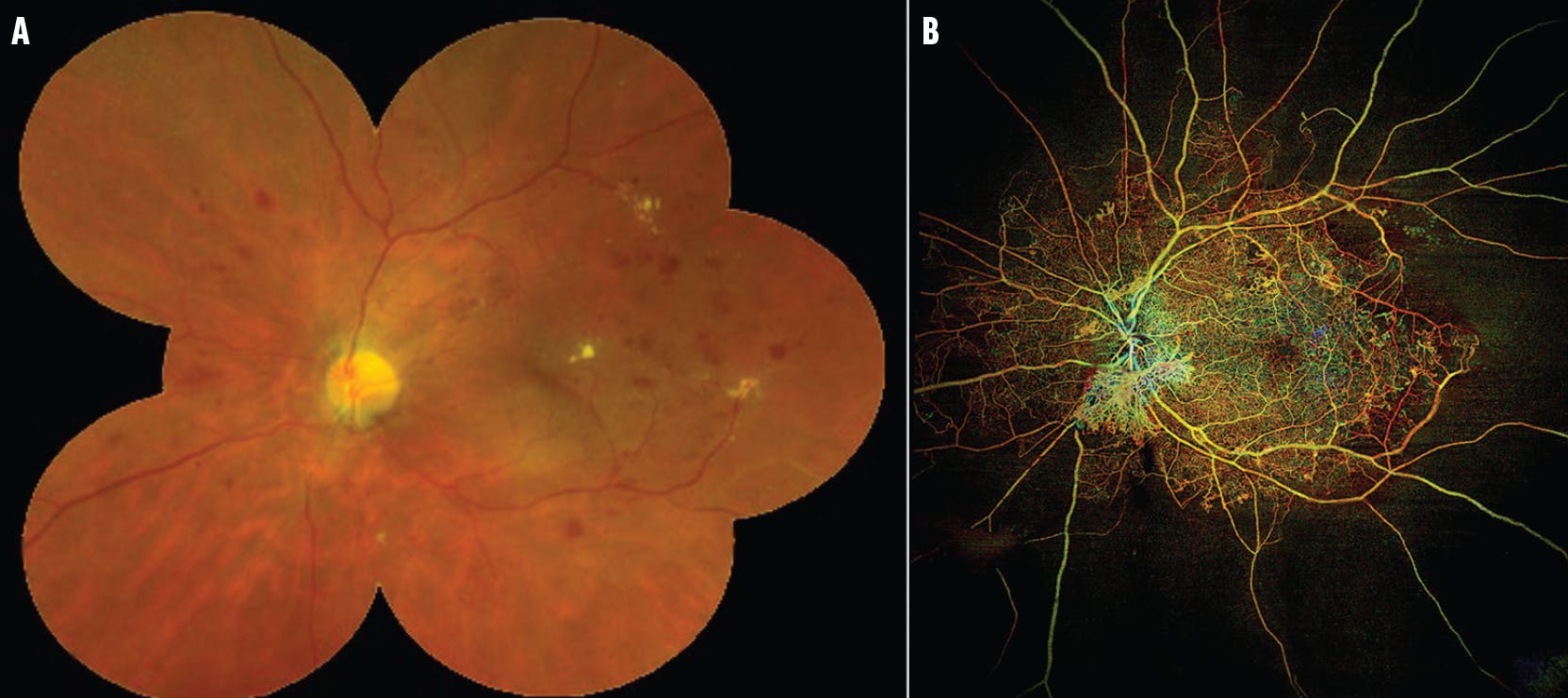
Figure 2. An eye with PDR imaged with fundus photography (A) and WF OCTA (B). The ultra-widefield photograph is cropped to reflect the field of view with current DR staging protocols. The WF OCTA image captures a larger field of view and reveals retinal ischemia and vascular abnormalities such as NV of the disc.
TRD: The final stage is defined by RD with traction from active or quiescent NV or fibrosis.14 TRD substages are defined based on location: extramacular, macular (but not foveal), or foveal detachment. DME remains an off-axis parameter that is imaged and potentially quantifiable with every OCTA scan, unlike in the current staging system.
In contrast to current DR staging, this OCTA-based system intentionally does not include intraretinal hemorrhages, which are readily apparent on fundus photography but less distinguishable on OCTA and typically not visually significant. Because hemorrhages are secondary to vascular compromise, eyes with hemorrhages always have other vascular features of DR on OCTA. Moreover, the lack of hemorrhages can deceive a clinician into undergrading diabetic eyes. Specifically, some eyes with a featureless fundus with few or no hemorrhages may have severe ischemia and, often, NV.
As such, our system was designed to focus on other, more clinically relevant lesions, such as IRMA, NV, and DME. The goal of a staging system should be to guide the clinician on how to help the patient see better, not to make the fundus look better.
HURDLES
An OCTA-based staging system for DR faces several potential barriers to adoption in the clinic and for clinical research purposes. For one, there is a learning curve for OCTA interpretation that can intimidate some technicians and clinicians. This curve is not steep for DR, however, because research shows that trainees and retina specialists can identify PDR on WF OCTA images with similar accuracy as for fluorescein angiograms.15
Another barrier is that the instruments required for WF OCTA are not universally available and are costly (on par with an ultra-widefield fundus camera), and current insurers do not reimburse OCTA interpretation separately from OCT. Finally, the large data files of WF OCTA present challenges in the amount of data storage required and the ease of real-time, in-clinic evaluation. Further technological improvements should help overcome these limitations.
TIME TO MODERNIZE
A new system that uses the advantages of WF OCTA can modernize the DR staging system to include all structural and vascular aspects of DR. For example, WF OCTA enables detection of all clinically relevant features of DR and builds upon current DR staging methods while overcoming their primary limitations.
The new OCTA-based system can be used for staging DR in routine clinical practice and can be standardized for research studies. Incorporation in future clinical trials is necessary to demonstrate the efficacy of this new staging system for preventing vision loss from DR.
1. Zhang X, Saaddine JB, Chou CF, et al. Prevalence of diabetic retinopathy in the United States, 2005-2008. JAMA. 2010;304(6):649-656.
2. Kempen JH, O’Colmain BJ, Leske MC, et al. The prevalence of diabetic retinopathy among adults in the United States. Arch Ophthalmol. 2004;122(4):552-563.
3. Early Treatment Diabetic Retinopathy Study Research Group. Grading diabetic retinopathy from stereoscopic color fundus photographs–an extension of the modified Airlie House classification. ETDRS report number 10. Ophthalmology. 1991;98(5 suppl):786-806.
4. Russell JF, Han IC. Toward a new staging system for diabetic retinopathy using wide field swept-source optical coherence tomography angiography. Curr Diab Rep. 2021;21(9):28.
5. Abramoff MD, Fort PE, Han IC, Jayasundera KT, Sohn EH, Gardner TW. Approach for a clinically useful comprehensive classification of vascular and neural aspects of diabetic retinal disease. Invest Ophthalmol Vis Sci. 2018;59(1):519-527.
6. Jampol LM, Tadayoni R, Ip M. Need for a new classification of diabetic retinopathy. Retina. 2021;41(3):459-460.
7. Solomon SD, Goldberg MF. ETDRS grading of diabetic retinopathy: still the gold standard? Ophthalmic Res. 2019;62(4):190-195.
8. Sun JK, Aiello LP, Abramoff MD, et al. Updating the staging system for diabetic retinal disease. Ophthalmology. 2021;128(4):490-493.
9. Sohn EH, Han IC, Abramoff MD. Diabetic retinal neurodegeneration-should we redefine retinopathy from diabetes? JAMA Ophthalmol. 2019;137(10):1132-1133.
10. Sun J. Updating the diabetic retinal disease staging system. Presented at: Angiogenesis, Exudation, and Degeneration; February 12, 2022; virtual meeting.
11. Cao D, Yang D, Huang Z, et al. Optical coherence tomography angiography discerns preclinical diabetic retinopathy in eyes of patients with type 2 diabetes without clinical diabetic retinopathy. Acta Diabetol. 2018;55(5):469-477.
12. Russell JF, Shi Y, Scott NL, Gregori G, Rosenfeld PJ. Longitudinal angiographic evidence that intraretinal microvascular abnormalities can evolve into neovascularization. Ophthalmol Retina. 2020;4(12):1146-1150.
13. Russell JF, Shi Y, Hinkle JW, et al. Longitudinal wide-field swept-source OCT angiography of neovascularization in proliferative diabetic retinopathy after panretinal photocoagulation. Ophthalmol Retina. 2019;3(4):350-361.
14. Russell JF, Scott NL, Townsend JH, et al. Wide-field swept-source optical coherence tomography angiography of diabetic tractional retinal detachments before and after surgical repair. Retina. 2021;41(8):1587-1596.
15. Al-Khersan H, Russell JF, Lazzarini TA, et al. Comparison between graders in detection of diabetic neovascularization with swept source optical coherence tomography angiography and fluorescein angiography. Am J Ophthalmol. 2021.;224:292-300.













_1773249222.png?auto=compress,format&w=75)










