Several important organ functions in the human body regulate themselves in order to preserve life. For example, body temperature is regulated within 2 degrees Celsius before organ systems begin to malfunction.1 In a similar way, intraocular pressure (IOP) is also regulated within a narrow range, and if exceeded, can result in morbid sequelae. Acute intraoperative ocular hypotony can result in suprachoroidal hemorrhage, especially if the initial IOP is elevated.2 Ocular hypertension can also have its consequences. Data from the cataract surgery literature indicate that postoperative and intraoperative elevated IOP can adversely affect visual outcomes in the form of post-cataract-extraction anterior ischemic optic neuropathy (PCE-AION), which develops within a few hours or days after the extraction,3 or postoperative nonarteritic anterior ischemic optic neuropathy (NAION), characterized by sudden, painless, mostly irreversible, and generally nonprogressive visual loss.4 IOP elevations of 45 mm Hg or higher have been documented to be associated with NAION.5 A study by Findl determined that in healthy subjects, choroidal blood flow decreases when IOP is increased by as little as 10 mm Hg, and significantly with an increase of 20 mm Hg. Reduced blood flow velocity was also noted in the central retinal artery with elevated IOP.6 Nevertheless, the ocular elements that ultimately suffer the consequences leading to compromised vision occur in the posterior segment, despite the abnormal intraoperative IOP occurring within the anterior segment. As opposed to the healthy subjects in the study by Findl, the effect of reduced perfusion from elevated IOP may have a further detrimental effect in unhealthy subjects who typically undergo vitreoretinal surgery and could experience compromised perfusion. Any factor that reduces circulation, such as elevated intraoperative IOP, can jeopardize vision.
Intraoperative IOP Management Over Time
Retinal surgeons, for the most part, are knowledgeable about avoiding the extremes of IOP during surgery. There has been significant progress since the days of Machemer, who used gravity to infuse the eye with saline during vitrectomy. Some of today’s modern surgical vitrectomy consoles utilize a version of Vented Gas-forced Infusion (VGFI) in which an increased pressure, as well as a rapid decrease of infusion pressure in the buffered saline solution (BSS) bottle, is possible via a surgeon-controlled foot pedal.7 In 2014, Stalmans highlighted features of the EVA System (DORC International, Zuidland, The Netherlands), including Automatic Infusion Compensation (AIC), which compensates for the volume mismatch typically experienced when aspirating with the vitrector in vacuum mode at relatively high vacuum levels. The infusion pressure setting elevates by generating higher air pressure in the infusion bottle as the aspiration of the vitrector increases to prevent collapse; the bottle is vented back to baseline to prevent a rise in IOP when cutter aspiration stops.8 VGFI-based systems are a significant improvement, but they are not perfect. Studies in human eyes have shown that IOP during vitrectomy using a gravity flow or a vented gas-forced infusion method ranged from 0-120 mm Hg.9
There has been an evolution of control systems over time that optimize operational efficiency, such as fuel delivery for a gasoline internal combustion engine. Prior to the use of fuel injection in modern vehicles, the air-fuel mixture delivered to the cylinders was regulated by a carburetor. Although this system worked fairly well for over 100 years, modern fuel injection systems use mass airflow and oxygen sensor data to provide information to a computer that sends duty-cycle information to fuel injectors to provide optimized engine efficiency and performance. This closed-loop operation uses feedback to continuously deliver optimal combustion efficiency. In a similar manner, the CONSTELLATION Vision System (Alcon LLC, Fort Worth, TX; USA, Figure 1) has a closed-loop pressure control system that can maintain constant IOP during vitrectomy within ±1-2 mm Hg regardless of aspiration flow rates.10
CONSTELLATION has a noninvasive flow sensor that uses ultrasound wave differential to detect the actual flow velocity that can, then, derive flowrate and other operational parameters.11 This approach allows the IOP to be constantly controlled throughout the various operating conditions experienced during a case. Sugiura et al also concluded that CONSTELLATION IOP Control can attenuate IOP fluctuations during vitrectomy maneuvers with no significant difference in IOP fluctuations between gauge sizes.12
Intraoperative IOP Management — Surgical Console Performance
In 2020, Shinkai et al first reported on the variability of IOP during ex-vivo vitrectomy in Porcine eyes between the EVA AIC and the CONSTELLATION Vision System IOP Control. The experimental findings demonstrated that during core vitrectomy for the 25G and 27G cutters, there was less variability with the CONSTELLATION Vision System when compared to the EVA in vacuum mode. However, the least variability was with the 27G EVA in Flow Mode.13 Although this study measured IOP variability, the time duration of IOP outside of the set range was not measured. To further evaluate intraoperative IOP performance, data from a video presentation at the 2023 American Society of Retina Specialists meeting in Seattle described an experiment undertaken to evaluate the IOP variability of these same two systems during simulated vitrectomy and described the time during which each system spent outside of the set range. The study was performed at vacuum setting of 250 mm Hg, 450 mm Hg and 650 mm Hg for both the 25- and 27- gauge TDC cutters (DORC International, Zuidland, The Netherlands) at 16,000 cuts per minute with the AIC settings that allowing IOP to be at 30 mm Hg during the aspiration of pure BSS. The CONSTELLATION Vision System was also tested using the same vacuum settings with the 25- and 27- gauge HYPERVIT cutters (Alcon LLC, Fort Worth, TX; USA) at 20,000 cuts per minute with IOP Control ON set to 30 mm Hg.14
The average IOP prior to aspiration, average IOP during the vitreous removal, and average IOP after full vitreous removal were determined and analyzed; statistical analysis was performed using ANOVA with a P-value set to 0.05.14 The general appearance of the IOP pressure tracing for the 25G probes is shown on Figure 2. For CONSTELLATION, there is little IOP variation regardless of the engaged medium (vitreous or BSS) or the applied vacuum; the IOP ranged from 30.16 ± 0.17 to 31.55 ± 1.38 mm Hg. However, the EVA AIC shows a greater IOP variation dependent on both the applied vacuum as well as with the engaged medium, with IOP ranging from 30.43 ± 0.15 to 44.62 ± 1.24 mm Hg.14 Figure 3 also shows that CONSTELLATION IOP Control also spends a longer duration of the time (from 63% to 93%) in the target range (27.5-32.5 mm Hg) than the EVA AIC (from 16% to 23%).14

Figure 3. Comparison of the IOP distributions during 25-gauge vitrectomy using CONSTELLATION IOP Control (left) versus the EVA AIC (right). Blue rectangle demarcates the desired IOP range.14
Similarly, for the 27G probes, the general appearance of the IOP tracing is shown on Figure 4. For CONSTELLATION, little variation is seen regardless of the engaged medium or the applied vacuum, with IOP ranging from 30.19 ± 0.33 to 31.32 ± 1.06 mm Hg. However, the EVA AIC shows a greater IOP variation, ranging from 30.34 ±0.21 mm Hg to 38.18±0.76 mm Hg, dependent on both the engaged medium as well as with the applied vacuum.14 Furthermore, Figure 5 shows that CONSTELLATION IOP Control also spends a longer duration of the time (from 69% to 93%) in the target range (27.5-32.5 mm Hg) than the EVA AIC (from 13% to 22%).14
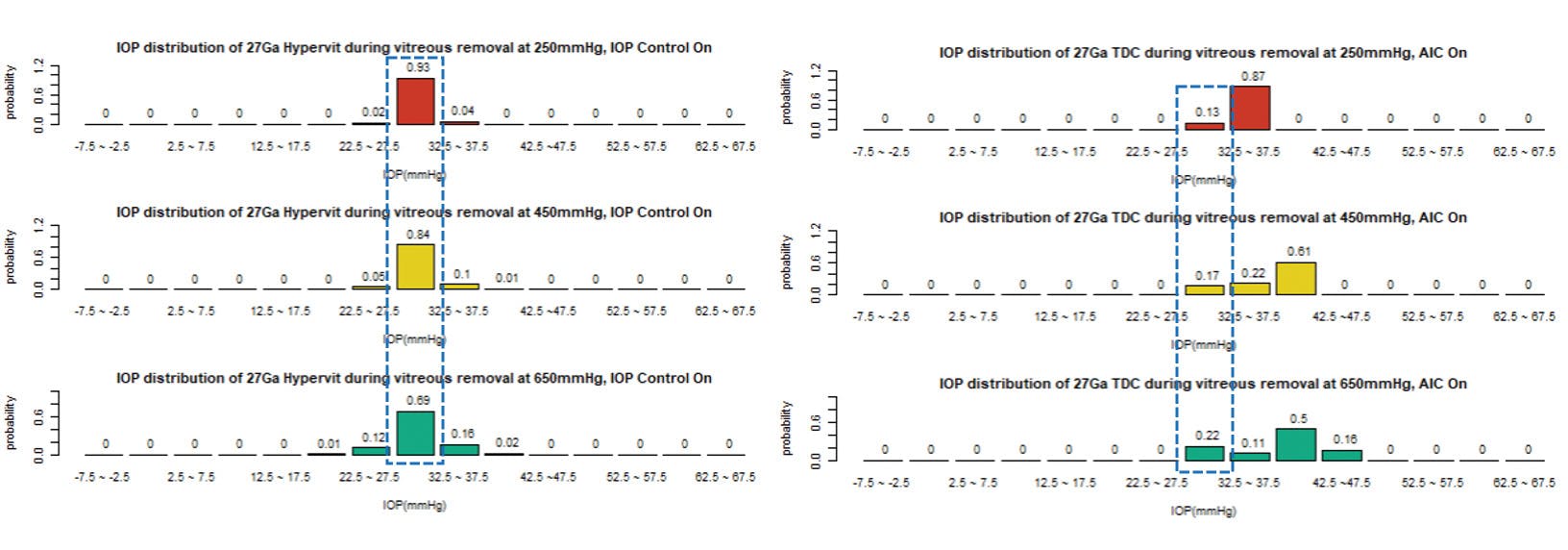
Figure 5. Comparison of the IOP distributions during 27-gauge vitrectomy using the CONSTELLATION IOP Control (left) versus the EVA AIC (right). Blue rectangle demarcates the desired IOP range.14
Clinical Experience
When retinal surgeons perform a vitrectomy, it is paramount that the IOP remain steady and consistent. Large fluctuations in IOP can cause retinal incarceration in the mouth of the cutter while performing a close vitreous shave with mobile retina in retinal detachment cases. Large fluctuations in IOP can also cause decreased vascular perfusion to the optic nerve, most notably when injecting a stain, such as tissue blue, triamcinolone acetonide or ICG during chemovitrectomy prior to a membrane peel. Finally, large fluctuations in IOP can cause a rare, but detrimental suprachoroidal hemorrhage if low pressure ensues while converting from BSS to air in an Air-Fluid exchange maneuver. Regardless of where the anomalous pressure originates (anterior or posterior segment), the retina will be subjected to the consequences of IOP instability. Having confidence that the IOP is continuously monitored and adjusted with CONSTELLATION IOP Control, regardless of the operative conditions, instills confidence that this important intraoperative parameter will not need my attention during the case.
Summary
Evidence regarding the extremes of low or high IOP indicates that visual loss can occur, especially in those with compromised retinal circulation. Not all IOP control systems operate equally; the closed-loop technology of IOP Control that has existed on the CONSTELLATION Vision System since 2008 rapidly adapts to changing intraoperative conditions and can provide confidence to the surgeon regarding IOP maintenance within a tight desired range regardless of probe gauge size or aspiration vacuum — this allows the surgeon to focus completely on the surgical procedure at hand.
CONSTELLATION® System with PUREPOINT® Laser Brief Statement
Caution: Federal law restricts this device to sale by, or on the order of, a physician.
Indications for Use: The CONSTELLATION® Vision System is an ophthalmic microsurgical system that is indicated for both anterior segment (i.e., phacoemulsification and removal of cataracts) and posterior segment (i.e., vitreoretinal) ophthalmic surgery.
The ULTRAVIT® Vitrectomy Probe is indicated for vitreous cutting and aspiration, membrane cutting and aspiration, dissection of tissue and lens removal. The valved entry system is indicated for scleral incision, canulae for posterior instrument access and venting of valved cannulae. The infusion cannula is indicated for posterior segment infusion of liquid or gas.
The PUREPOINT® Laser is indicated for use in photocoagulation of both anterior and posterior segments of the eye including:
• Retinal photocoagulation, panretinal photocoagulation and intravitreal endophotocoagulation of vascular and structural abnormalities of the retina and choroid including: Proliferative and nonproliferative retinopathy (including diabetic); choroidal neovascularization secondary to age-related macular degeneration; retinal tears and detachments; macular edema, retinopathy of prematurity; choroidal neovascularization; leaking microaneurysms.
• Iridotomy/Iridectomy for treatment of chronic/primary open angle glaucoma, acute angle closure glaucoma and refractory glaucoma.
• Trabeculoplasty for treatment of chronic/primary open angle glaucoma and refractory glaucoma.
• And other laser treatments including: internal sclerostomy; lattice degeneration; central and branch retinal vein occlusion; suturelysis; vascular and pigment skin lesions.
The FlexTip* laser probe is intended to be used with ALCON® 532nm laser systems.
Contraindications:
• Patients with a condition that prevents visualization of target tissue (cloudy cornea, or extreme haze of the aqueous humor of the anterior chamber of vitreous humor) are poor candidates for LIO delivered laser treatments.
• The infusion cannula is contraindicated for use of oil infusion.
Complications:
Corneal burns, inflammation, loss of best-corrected visual acuity, loss of visual field and transient elevations in intraocular pressure can occur as a result of ophthalmic laser treatment. Unintentional retinal burns can occur if excessive treatment beam power or duration is used.
Warnings and Precautions:
• The disposables used in conjunction with ALCON® instrument products constitute a complete surgical system.
• Use of disposables and handpieces other than those manufactured by Alcon may affect system performance and create potential hazards.
• Attach only Alcon supplied consumables to console and cassette luer fittings. Do not connect consumables to the patient’s intravenous connections.
• Mismatch of consumable components and use of settings not specifically adjusted for a particular combination of consumable components may create a patient hazard.
• Vitreous traction has been known to create retinal tears and retinal detachments.
• The closed loop system of the CONSTELLATION® Vision System that adjusts IOP cannot replace the standard of care in judging IOP intraoperatively. If the surgeon believes that the IOP is not responding to the system settings and is dangerously high or low, this may represent a system failure. Note: To ensure proper IOP
• Compensation calibration, place infusion tubing and infusion cannula on a sterile draped tray at mid-cassette level during the priming cycle.
• Leaking sclerotomy may lead to post-operative hypotony.
• Back scattered radiation is of low intensity and is not harmful when viewed through a protective filter. All personnel in the treatment room must wear protective eyewear, OD4 or above at 532nm, when the system is in Standby/Ready mode as well as during treatment. The doctor protection filter is an OD greater than 4 at 532nm.
Attention: Please refer to the CONSTELLATION® Vision System Operators Manual for a complete listing of indications, warnings, and precautions.
*Trademarks are property of their respective owners.
© 2023 Alcon Inc. 07/23 US-CON-2300010
1. Moran DS, Mendal L. Core temperature measurement: methods and current insights Sports Med. 2002;32(14):879-85.
2. Tabandeh H, Flynn HW Jr. Suprachoroidal hemorrhage during pars plana vitrectomy Curr Opin Ophthalmol. 2001 Jun;12(3):179-85.
3. Hayreh SS. Anterior ischemic optic neuropathy. IV. Occurrence after cataract extraction Arch Ophthalmol. 1980 Aug;98(8):1410-6.
4. Fontes BM, Jung LS, Soriano ES, Chicani CF. Nonarteritic anterior ischemic optic neuropathy after uneventful phacoemulsification: case report. Arq Bras Oftalmol. 2007 May-Jun;70(3):544-6.
5. McCulley TJ, Lam BL, Feuer WJ. Nonarteritic anterior ischemic optic neuropathy and surgery of the anterior segment: temporal relationship analysis. Am J Ophthalmol. 2003 Dec;136(6):1171-2.
6. Findl O, Strenn K, Wolzt M, Menapace R, Vass C, Eichler HG, Schmetterer L. Effects of changes in intraocular pressure on human ocular haemodynamics. Curr Eye Res. 1997 Oct;16(10):1024-9.
7. Charles S. Fluidics and cutter dynamics Dev Ophthalmol. 2014;54:31-37.
8. Stalmans P. Enhancing visual acuitv Dev Ophthalmol. 2014-54:23.30.
9. Moorhead LC, Gardner TW, Lambert HM, O’Malley RE, Willis AW, Meharg LS, Moorhead WD. Dynamic intraocular pressure measurements during vitrectomy. Arch Ophthalmol. 2005 Nov;123(11):1514-23.
10. Riemann CD, Buboltz DC. Prevention of Intraoperative Hypotony During Vitreoretinal Surgery: An Instrumental Comparison. Poster, American Society of Retina Specialists, 2010; Vancouver.
11. Comaratta M, Hariprasad SM, Reddy R. The Evolution of Vitreoretinal Surgery Platforms. Ophthalmic Surg Lasers Imaging Retina. 2017 Jul 1;48(7):532-538.
12. Sugiura Y, Okamoto F, Okamoto Y, Hiraoka T, Oshika T. Intraocular pressure fluctuation during microincision vitrectomy with CONSTELLATION vision system. Am J Ophthalmol. 2013 Nov;156(5):941-947.
13. Shinkai Y, Yoneda K, Sotozono C. Ex vivo Comparison of Intraocular Pressure Fluctuation during Pars Plana Vitrectomy Performed Using 25- and 27-Gauge Systems. Ophthalmic Res. 2020 (Online).
14. Charles, M. et al. “Intraocular Pressure (IOP) control in Vitreoretinal surgical systems”, Film presented at American Society of Retina Specialists (ASRS) Annual Meeting; Seattle, WA, USA. July 31, 2023.



