AT A GLANCE
- In proliferative diabetic retinopathy (PDR), when the balance between VEGF and the profibrotic connective tissue growth factor reaches a specific threshold ratio, an angiofibrotic switch occurs, which results in scarring and eventual blindness.
- The traditional indication for vitrectomy in the setting of PDR is waiting until relevant vision loss has occurred.
- Early vitrectomy, performed before the angiofibrotic switch occurs, is an important and viable treatment option to prevent vision-threatening complications in eyes with PDR.
Proliferative diabetic retinopathy (PDR) continues to be a major contributor to global blindness, as it may lead to tractional retinal detachment (TRD) and vitreous hemorrhage. While advanced treatment approaches, such as panretinal photocoagulation (PRP) and anti-VEGF therapy, decrease the likelihood of vision loss, a significant portion of eyes—approximately one-third—may still experience long-term complications related to PDR.1 Therefore, standard treatment with PRP or anti-VEGF therapy appears insufficient to prevent vision loss in specific high-risk eyes. Here, we discuss the value of early vitrectomy for the treatment of PDR before vision-threatening complications occur (Figures 1-3).
ANGIOFIBROTIC SWITCH
Vision loss from PDR stems from the growth of new blood vessels and the formation of scar tissue in the vitreous cavity. In PDR, there is a response, similar to wound healing, that involves neovascularization with infiltration of inflammatory cells and the formation of myofibroblasts. This advances to fibrovascular contraction, leading to complications such as hemorrhage, TRD, and subsequent vision loss. Various growth factors are involved, including VEGF-A, transforming growth factor-ß, hepatocyte growth factor, platelet-derived growth factor, and the profibrotic connective tissue growth factor (CTGF).
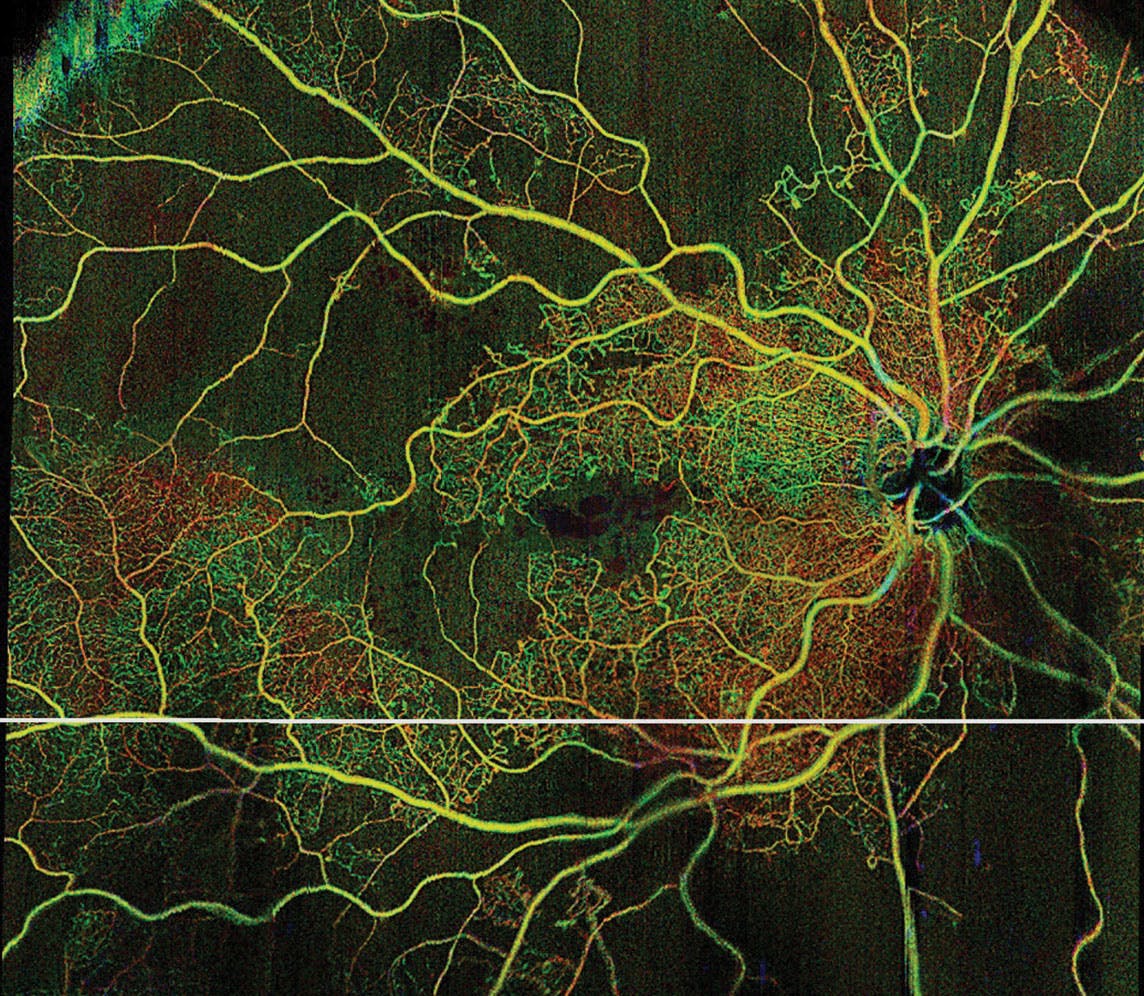
Figure 1. Widefield swept-source OCT angiography of a 56-year-old PDR patient with an indication for early vitrectomy.
VEGF is recognized as the principal factor driving angiogenesis and, among other elements, increases the expression of CTGF in different cell types within the newly developed neovascular membranes. Kuiper et al suggested that elevated CTGF levels, which have been found in the vitreous of PDR patients with active neovascularization, inactivate VEGF by diminishing production. When the balance between these two factors reaches a specific threshold ratio, the transition to angiofibrotic conditions, or an angiofibrotic switch, occurs with excessive CTGF levels driving fibrosis, which results in scarring and eventual blindness.2
TREATMENT CONCERNS
Parikh et al showed that when eyes presented with PDR and vitreous hemorrhage, there was a 34% probability of needing vitrectomy within 2 years.3 As demonstrated in the DRCR Retina Network Protocol S study, which compared PRP with ranibizumab (Lucentis, Genentech/Roche) in patients with PDR, just one-third of eyes treated with either PRP or ranibizumab exhibited a regression of new vessels and an improvement in the DR grading from PDR to nonproliferative DR. Among these eyes, half presented with vitreous hemorrhage at some stage during the 5-year follow-up period, 6% to 15% showed a TRD, and 11% to 20% required vitrectomy.4
In addition, anti-VEGF agents lack long-term effectiveness with vascular growth potentially recurring, which may result in patients presenting with pronounced retinal neovascularization, particularly when follow-up appointments are missed. Despite the substantial resources of the DRCR Retina Network to engage with patients, the 5-year results indicated a high rate of loss to follow-up of 39%.4
THE CASE FOR EARLY VITRECTOMY
The DRCR Retina Network’s Protocol AB was a prospective surgical study (n = 205 eyes) that compared the efficacy of immediate vitrectomy with anti-VEGF therapy with aflibercept (Eylea, Regeneron) for vitreous hemorrhage due to PDR.5 In the 2-year follow-up period, 33% of eyes in the aflibercept group required vitrectomy compared with 8% of eyes in the initial vitrectomy group, which also experienced a significantly lower recurrence rate of vitreous hemorrhage or active neovascularization (3%) compared with the aflibercept group (49%). About a tenth of eyes treated initially with aflibercept presented with TRD over the course of the observational period.
Patients who have experienced permanent vision loss in one eye due to diabetic TRDs are at an elevated risk of facing visual loss due to similar causes in the fellow eye. A recent series published by Schreur et al reported a 5-year cumulative incidence of up to 25% in such cases.6

Figure 2. A 26-year-old woman with type 1 diabetes and PDR. With triamcinolone staining, note the facile separation of the vitreous cortex, despite several vascular pegs/bleeding points. Such stage of vitreoretinal interface changes from PDR is ideally suited for early vitrectomy with a likely good long-term visual prognosis.
The traditional indication for vitrectomy in PDR is waiting (a reactive approach) until a relevant vision loss has occurred due to non-clearing vitreous hemorrhage, TRD, macular traction, or dense premacular subhyaloid hemorrhage. One of the concerns with diabetic vitrectomy has been the high rate of complications shown in the Diabetic Retinopathy Vitrectomy Study, reporting a risk of significant vision loss of up to 40%.7
Over the past few decades, advancements in small-gauge vitreoretinal surgery—including enhanced fluidics and highly efficient vitreous cutters—have significantly enhanced the safety of the procedure with notable success rates in achieving retinal reattachment and fewer occurrences of severe vision loss in these eyes.8
The need for vitrectomy in the fellow eye rises to up to 36% within 5 years following the first vitrectomy.9 Moreover, a suboptimal visual outcome after vitrectomy in the first eye serves as a predictor for a less favorable outcome in the fellow eye, particularly when the indication for vitrectomy is still considered reactive.10
Although it is feasible to enhance retinal structure and function reactively using current surgical techniques, restoring good vision is a rare outcome once traction has been established.11 In TRD cases, vitrectomy combined with delamination or segmentation of the preretinal fibrovascular membranes showed an intraoperative complication rate of 30%, with 58% of eyes requiring an intravitreal tamponade and 15% needing a second vitrectomy.12 Although there was an overall visual improvement after surgery, fewer than one-third of eyes analyzed in the Royal College of Ophthalmologists’ National Ophthalmology Database maintained the standard visual requirement for driving.12
IS EARLY VITRECTOMY CLINICALLY JUSTIFIED?
Tan et al investigated the safety and efficacy of preemptive diabetic vitrectomy in patients with severe, non-fibrotic retinal neovascularization (before the angiofibrotic switch).13 They reported that the overall intraoperative complications were less frequent compared with those reported in the UK National Ophthalmological Database (UKNOD) study for diabetic vitrectomy (10% vs 23%).12,13 The primary intraoperative complication noted was iatrogenic breaks, occurring at a rate of 10%, which was similar to that reported in the UKNOD series when delamination was not necessary but lower when delamination was performed (28%).12,13 Tan et al reported the need for gas tamponade in 10% of eyes and no case of silicone oil, while 60% to 100% of eyes required a tamponade in reactive diabetic vitrectomy with silicone oil rates reaching up to 20%.13,14
The reduced complication rate may be attributed to the relatively normal vitreoretinal interface allowing a less traumatic surgical separation of the vitreous from the retina, which also contributes to long-term stabilization.15 This contrasts with eyes that had already undergone an angiofibrotic switch, where fibrovascular complexes with traction are present.
![<p>Figure 3. Long-term course of early vitrectomy in PDR (patient used in dataset for Tan et al. Eye [Lond].<sup>13</sup>). Preoperatively (A), the patient’s VA was 0.6 (0.2 logMAR). Four years after surgery (B), the patient’s VA was 0.8 (0.1 logMAR).</p>](https://core4-cms.imgix.net/0124RT_Cover_Pollreisz-12_1706543903.jpg?auto=compress,format)
Figure 3. Long-term course of early vitrectomy in PDR (patient used in dataset for Tan et al. Eye [Lond].13). Preoperatively (A), the patient’s VA was 0.6 (0.2 logMAR). Four years after surgery (B), the patient’s VA was 0.8 (0.1 logMAR).
Postoperative vitreous hemorrhage is a common complication following diabetic vitrectomy; its reported incidence ranges from 20% to 75%.13,16 The administration of anti-VEGF agents before surgery has been linked to a decrease in this risk, bringing it down to 10% to 20%.17 In the Preemptive Diabetic Vitrectomy study, the incidence of postoperative vitreous hemorrhage was 40%, occurring mainly within 4 weeks after surgery; the authors hypothesized that the elevated occurrence was a result of cutting or avulsion of numerous pegs connecting the retinal and neovascular circulations upon posterior vitreous detachment.13
Thus, it is crucial to inform patients about the potential risk for a temporary decrease in vision in the initial postoperative period and the likelihood of intravitreal injection or second surgery before achieving long-term stabilization of vision.
THE CLINICAL PEARL
We have excellent treatment options available for patients with PDR to preserve vision. While PRP and anti-VEGF therapy continue to play an important role, early vitrectomy, performed before the angiofibrotic switch and before silicone oil filling is necessary, is another important and viable treatment to prevent vision-threatening complications.
1. Writing Committee for the Diabetic Retinopathy Clinical Research Network; Gross JG, Glassman AR, Jampol LM, et al. Panretinal photocoagulation vs intravitreous ranibizumab for proliferative diabetic retinopathy: a randomized clinical trial. JAMA. 2015;314(20):2137-2146. Erratum in: JAMA. 2019;321(10):1008.
2. Kuiper EJ, Van Nieuwenhoven FA, de Smet MD, et al. The angio-fibrotic switch of VEGF and CTGF in proliferative diabetic retinopathy. PLoS One. 2008;3(7):e2675.
3. Parikh R, Shah RJ, VanHouten JP, Cherney EF. Ocular findings at initial pan retinal photocoagulation for proliferative diabetic retinopathy predict the need for future pars plana vitrectomy. Retina. 2014;34(10):1997-2002.
4. Gross JG, Glassman AR, Liu D, et al; Diabetic Retinopathy Clinical Research Network. Five-year outcomes of panretinal photocoagulation vs intravitreous ranibizumab for proliferative diabetic retinopathy: a randomized clinical trial. JAMA Ophthalmol. 2018;136(10):1138-1148. Erratum in: JAMA Ophthalmol. 2019;137(4):467.
5. Glassman AR, Beaulieu WT, Maguire MG, et al; DRCR Retina Network. Visual acuity, vitreous hemorrhage, and other ocular outcomes after vitrectomy vs aflibercept for vitreous hemorrhage due to diabetic retinopathy: a secondary analysis of a randomized clinical trial. JAMA Ophthalmol. 2021;139(7):725-733.
6. Schreur V, Brouwers J, Van Huet RAC, et al. Long-term outcomes of vitrectomy for proliferative diabetic retinopathy. Acta Ophthalmol. 2021;99(1):83-89.
7. Early vitrectomy for severe proliferative diabetic retinopathy in eyes with useful vision. Results of a randomized trial—Diabetic Retinopathy Vitrectomy Study Report 3. The Diabetic Retinopathy Vitrectomy Study Research Group. Ophthalmology. 1988;95:1307-1320.
8. Khan MA, Samara WA, Hsu J, Garg S. Short-term outcomes of hybrid 23-, 25-, and 27-gauge vitrectomy for complex diabetic tractional retinal detachment repair. Retin Cases Brief Rep. 2019;13(3):244-247.
9. Hwang JC, Sharma AG, Eliott D. Fellow eye vitrectomy for proliferative diabetic retinopathy in an inner city population. Br J Ophthalmol. 2013;97(3):297-301.
10. Sulak M, Urbancic M, Petrovic MG. Predicting visual outcomes of second eye vitrectomy for proliferative diabetic retinopathy. Retina. 2018;38(4):698-707.
11. McCullough P, Mohite A, Virgili G, Lois N. Outcomes and complications of pars plana vitrectomy for tractional retinal detachment in people with diabetes: a systematic review and meta-analysis. JAMA Ophthalmol. 2023;141(2):186-195.
12. Jackson TL, Johnston RL, Donachie PH, Williamson TH, Sparrow JM, Steel DH. The Royal College of Ophthalmologists’ National Ophthalmology Database Study of vitreoretinal surgery: report 6, diabetic vitrectomy. JAMA Ophthalmol. 2016;134(1):79-85.
13. Tan SZ, Steel DH, Stanzel BV, et al. Safety and effectiveness of pre-emptive diabetic vitrectomy in patients with severe, non-fibrotic retinal neovascularisation despite panretinal photocoagulation. Eye (Lond). 2023;37(8):1553-1557.
14. Sokol JT, Schechet SA, Rosen DT, Ferenchak K, Dawood S, Skondra D. Outcomes of vitrectomy for diabetic tractional retinal detachment in Chicago’s county health system. PLoS One. 2019;14(8):e0220726.
15. Ono R, Kakehashi A, Yamagami H, et al. Prospective assessment of proliferative diabetic retinopathy with observations of posterior vitreous detachment. Int Ophthalmol. 2005;26(1-2):15-19.
16. Khuthaila MK, Hsu J, Chiang A, et al. Postoperative vitreous hemorrhage after diabetic 23-gauge pars plana vitrectomy. Am J Ophthalmol. 2013;155:757-63, 763.e1-2.
17. Smith JM, Steel DH. Anti-vascular endothelial growth factor for prevention of postoperative vitreous cavity haemorrhage after vitrectomy for proliferative diabetic retinopathy. Cochrane Database Syst Rev. 2015;2015(8):CD008214.














_1773249222.png?auto=compress,format&w=75)










