AT A GLANCE
- Accurate diagnosis of inherited retinal disease (IRD) remains a challenge due to significant overlap between genetically distinct conditions and the immense number of genes implicated in IRD pathogenesis.
- Ultra-widefield fundus photography establishes visible pathology and IRD staging information, whereas fundus autofluorescence and OCT provide dynamic metrics for monitoring disease stability and progression that serve as benchmarks in clinical trials.
- Advances in AI are beginning to uncover phenotype/genotype associations from input images to aid in IRD diagnosis; however, current studies are largely limited to a subset of genes and diseases.
With more than 300 gene mutations implicated in inherited retinal diseases (IRDs), there is an extreme diversity of clinical presentation and disease progression, with significant overlap between genetically distinct conditions.1 This poses a significant diagnostic and therapeutic challenge that requires a multidisciplinary team and an expansive toolkit.2 Alongside detailed history-taking and examination, ophthalmic imaging and genetic testing are central pillars of precise IRD diagnosis.3,4 While next-generation sequencing is imperative to identify causative gene mutations, imaging remains crucial for monitoring disease progression. Additionally, advances in AI have led to the beginnings of imaging-based algorithms that might one day be helpful in identifying IRDs.5-7 In this article, we focus on the primary imaging approaches used in IRD diagnosis and management.
FUNDUS PHOTOGRAPHY
Fundus photography offers the benefit of establishing visible retinal pathology and helps clinicians detect pathognomonic signs and distinguish IRDs from more common conditions such as AMD and inflammatory disorders (Figure 1). Photographs also assist with disease staging. Stargardt disease, the most common macular dystrophy, harbors a staging system based on the presence, extension, and disappearance of retinal flecks and the progression of central atrophy (Figure 2).8 Best disease, the second most common inherited macular dystrophy, has a staging system based on posterior segment changes that correlate with visual acuity.9 Additionally, a hallmark of retinitis pigmentosa (RP), the most common IRD, includes fundus findings such as optic disc pallor, arteriolar attenuation, and bony spicule pigment clumping. Efforts are underway to develop machine-learning algorithms to analyze fundus photographs, which could enhance the sensitivity and efficiency of IRD screening and expedite patient care and diagnosis.7

Figure 1. This 34-year-old man was diagnosed with gyrate atrophy based, in part, on the fundus imaging (A) and UWF fundus imaging (B) of the right eye, which are notable for RPE atrophy with scalloped posterior borders.

Figure 2. Multimodal imaging of the right eye of a 12-year-old with Stargardt disease includes a fundus image showing faint yellow flecks and atrophy within the macula (A) and FAF demonstrating central hypoautofluorescence surrounded by a ring of hyperautofluorescence and prominent hyperautofluorescent flecks within the macula (B). OCT shows subfoveal and parafoveal RPE and outer retina loss (C). Visual acuity was 20/250 at the time of imaging. Multimodal imaging of the patient’s asymptomatic sibling at a less advanced stage of disease includes fundus imaging with faint yellow flecks (D) and FAF showing prominent hyperautofluorescent flecks within the macula without atrophy (E).
FUNDUS AUTOFLUORESCENCE
On fundus autofluorescence (FAF), hyperautofluorescence often indicates increased metabolic activity potentially in the setting of cellular stress or inflammation, whereas hypoautofluorescence indicates retinal pigment epithelium (RPE) atrophy. This ultimately offers a snapshot of disease activity, and serial imaging can document disease stability and progression. Because of the degenerative nature of IRDs, many conditions display a sequence of hyperautofluorescence in early stages with a transition to hypoautofluorescence in later atrophic stages. In RP, a ring of hyperautofluorescence often surrounds the fovea and macula encircling the central viable retina, which slowly constricts with more advanced disease (Figure 3).10 FAF also offers the added benefit of visualizing perturbations to cellular homeostasis before overt damage appears on fundoscopy. In classic Stargardt disease, FAF demonstrates macular hyperautofluorescence that precedes posterior segment findings on fundoscopy. This possibly indicates a therapeutic window for repairing damage before it becomes irreversible. In later stages of Stargardt disease, there is progression of hypoautofluorescence moving outward from the macula surrounded by a leading wave of hyperautofluorescence, creating the classic bull’s eye appearance, with areas of decreased autofluorescence serving as a measurable marker of disease progression in clinical trials.11 This pathological hallmark is also seen with drug-induced maculopathies (eg, hydroxychloroquine toxicity) and cone dystrophy, highlighting the importance of thorough history-taking when assessing patients for IRD.

Figure 3. FAF of a 21-year-old woman with early-stage RP shows a notable ring of hyperautofluorescence around the macula (A). FAF of a 48-year-old man shows a broad ring of hyperautofluorescence around the macula and peripheral hyorautofluorescence from pigment deposition and RPE atrophy (B).
Research suggests that machine learning may be able to use FAF images to help distinguish between IRDs. In one study, machine learning achieved a global accuracy of 0.95 when assigning FAF images as either RP, Best disease, Stargardt disease, or healthy controls.5 Another study found that a machine-learning algorithm was as accurate as retina specialists at distinguishing FAF images of pattern dystrophy and Stargardt disease.6 However, in both studies, the accuracy of algorithms was below 100%, which reflects the need to use multimodal imaging and combine imaging with other diagnostic tools to improve IRD identification.
ULTRA-WIDEFIELD IMAGING
Ultra-widefield (UWF) technology allows up to a 200° view of the retina, offering the chance to incorporate peripheral pathology into IRD classification systems.12 Ongoing studies are redefining the grading of Stargardt disease and RP using UWF FAF imaging with a focus on extension of pathology into the periphery.13 In the setting of RP, for example, hypoautofluorescent patterns in the midperiphery correlate with scotomas on visual field testing.14 Investigators are working to incorporate UWF imaging into machine-learning algorithms to further refine the algorithms’ diagnostic accuracy.
OCT
OCT allows for 3D dissection of the integrity of the individual retinal layers during the progression of IRD. Ellipsoid zone (EZ) disruption, RPE atrophy, outer retinal layer atrophy, and loss of the choriocapillaris are core OCT components of numerous IRDs. EZ disruption has garnered significant attention as a measure of disease progression and is being used as a clinical endpoint in RP trials and in investigations of complement inhibitors in Stargardt disease (NCT03364153).15,16 OCT can also assist with diagnosis and disease staging as seen in achromatopsia, which starts with EZ disruption followed by formation of optically empty space and eventual outer retinal loss (Figure 4).17
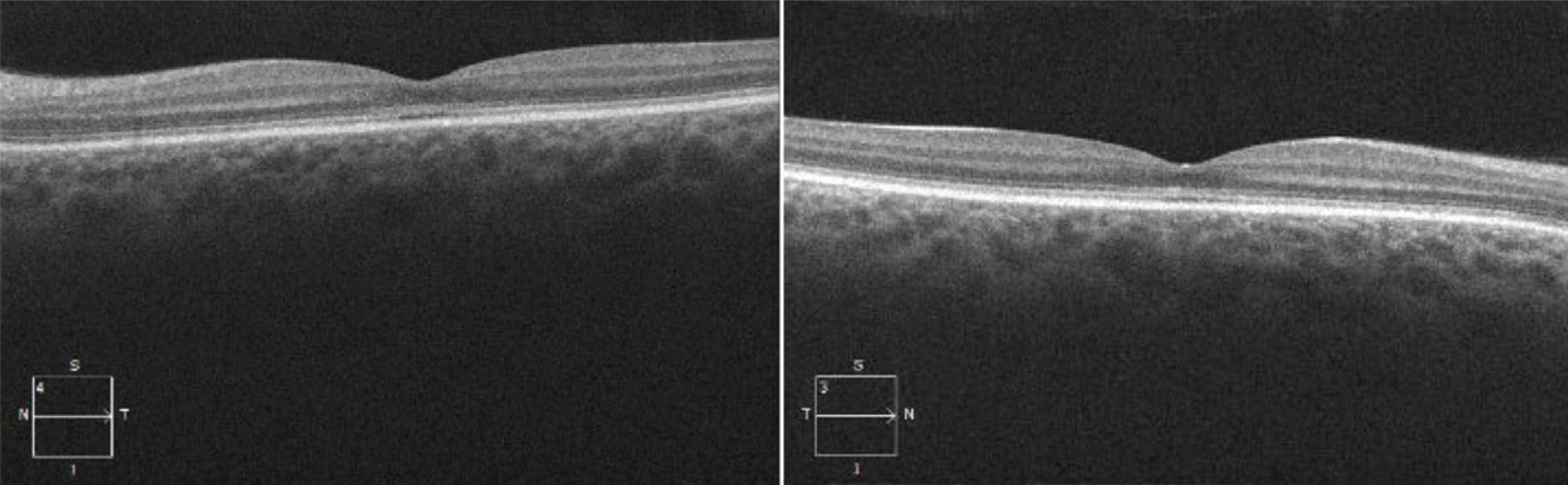
Figure 4. OCT of the left and right eye of a 28-year-old woman with achromatopsia (homozygous for CNGB3 mutations) shows the beginnings of subfoveal EZ disruption and formation of optically empty space in each eye.
Many IRDs are associated with pigment deposition and RPE changes, and OCT can further classify the location of the lesions to provide diagnostic insight. Pattern dystrophy leads to collections of subretinal hyperreflective material and RPE disruption, while deposits seen in Best disease are often hyperreflective vitelliform subretinal domes that transition to neovascular or atrophic lesions (Figure 5).8 OCT also pinpoints complications of specific IRDs, including cystoid macular edema (CME) in RP and choroidal neovascularization (CNV) in pattern dystrophy. Studies have also examined the association of OCT findings with IRD progression and prognosis.18,19
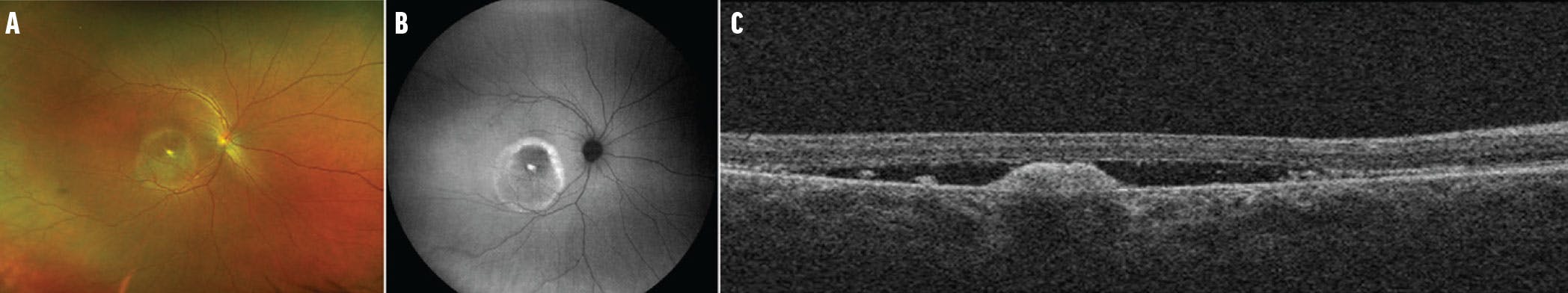
Figure 5. Multimodal imaging of the right eye of a 40-year-old woman with confirmed BEST1-associated macular dystrophy includes fundus images showing the vitelliruptive stage with the “scrambled egg” appearance, or stage IV (A); FAF demonstrating a ring of hyperautofluorescence (B); and OCT showing vitelliform material deposition in the inferior macula (C).
ANGIOGRAPHY
Fluorescein angiography (FA), used to assess vascular integrity, is not commonly employed in monitoring patients with IRD. For example, the traditional FA dark choroid sign in Stargardt disease, attributed to lipofuscin deposition in the RPE masking the underlying choroidal signal, has largely been superseded by FAF, which offers greater sensitivity. However, FA is still useful in detecting complications of some IRDs, such as CNV or non-leaking CME.
A newer and less invasive imaging technique, OCT angiography (OCTA) provides detailed visualization of the superficial and deep retinal capillary plexus and choriocapillaris. In addition to CNV monitoring, OCTA has identified reduced perfusion and an enlarged foveal avascular zone in RP and a degraded choriocapillaris in advanced stages of other IRDs, such as Best disease and choroideremia.20,21 The pathologic consequences of early vascular changes and potential integration of OCTA into machine-learning paradigms for the detection of IRD remain an active area of investigation.
THE FUTURE OF IRD IMAGING
Multimodal imaging is a cornerstone of the diagnosis and monitoring of IRDs because it provides a visual representation of disease status that is crucial for patient and family counseling. Significant challenges lie in the remarkable overlap in imaging findings between various IRDs and diseases such as AMD and drug-induced maculopathy. While researchers are making strides in using machine learning to uncover phenotype/genotype associations, current studies are largely limited to a subset of genes and diseases.5-7 Expanding algorithms to encompass all the genes implicated in IRD pathogenesis remains a formidable challenge but could potentially be improved with multimodal imaging and demographic inputs. This reinforces the importance of a multidisciplinary approach, in which complementary technologies—such as genetic testing and electrophysiology—synergize with imaging studies to streamline IRD diagnosis and enhance clinical trials that aim to provide treatments for these debilitating diseases.
1. Duncan JL, Pierce EA, Laster AM, et al. Inherited retinal degenerations: current landscape and knowledge gaps. Transl Vis Sci Technol. 2018;7(4):6.
2. Murro V, Banfi S, Testa F, et al. A multidisciplinary approach to inherited retinal dystrophies from diagnosis to initial care: a narrative review with inputs from clinical practice. Orphanet J Rare Dis. 2023;18(1):223.
3. Dockery A, Whelan L, Humphries P, Farrar GJ. Next-generation sequencing applications for inherited retinal diseases. Int J Mol Sci. 2021;22(11):5684.
4. Corradetti G, Verma A, Tojjar J, et al. Retinal imaging findings in inherited retinal diseases. J Clin Med. 2024;13(7):2079.
5. Miere A, Le Meur T, Bitton K, et al. Deep learning-based classification of inherited retinal diseases using fundus autofluorescence. J Clin Med. 2020;9(10):3303.
6. Miere A, Zambrowski O, Kessler A, et al. deep learning to distinguish ABCA4-related Stargardt sisease from PRPH2-related pseudo-Stargardt pattern dystrophy. J Clin Med. 2021;10(24):5742.
7. Fujinami-Yokokawa Y, Ninomiya H, Liu X, et al. Prediction of causative genes in inherited retinal disorder from fundus photography and autofluorescence imaging using deep learning techniques. Br J Ophthalmol. 2021;105(9):1272-1279.
8. Rahman N, Georgiou M, Khan KN, Michaelides M. Macular dystrophies: clinical and imaging features, molecular genetics and therapeutic options. Br J Ophthalmol. 2020;104(4):451-460.
9. Querques G, Zerbib J, Santacroce R, et al. Functional and clinical data of Best vitelliform macular dystrophy patients with mutations in the BEST1 gene. Mol Vis. 2009;15:2960-2972.
10. Tee JJL, Kalitzeos A, Webster AR, Peto T, Michaelides M. Quantitative analysis of hyperautofluorescent rings to characterize the natural history and progression in RPGR-associated retinopathy. Retina. 2018;38:2401.
11. Strauss RW, Kong X, Ho A, et al. Progression of Stargardt disease as determined by fundus autofluorescence over a 12-month period: ProgStar Report No. 11. JAMA Ophthalmol. 2019;137(10):1134-1145.
12. Cicinelli MV, Marchese A, Bordato A, Manitto MP, Bandello F, Battaglia Parodi M. Reviewing the role of ultra-widefield imaging in inherited retinal dystrophies. Ophthalmol Ther. 2020;9(2):249-263.
13. Klufas MA, Tsui I, Sadda SR, Hosseini H, Schwartz SD. Ultrawidefield autofluoresence in ABCA4 Stargardt disease. Retina. 2018;38:403-415.
14. Ogura S, Yasukawa T, Kato A, et al. Wide-field fundus autofluorescence imaging to evaluate retinal function in patients with retinitis pigmentosa. Am J Ophthalmol. 2014;158(5):1093-1098.
15. Campochiaro PA, Iftikhar M, Hafiz G, et al. Oral N-acetylcysteine improves cone function in retinitis pigmentosa patients in phase I trial. J Clin Invest. 2020;130(3):1527-1541.
16. Csaky KG, Bok D, Radu RA, Sadda SR. Complement C5 inhibition as a potential treatment for autosomal recessive Stargardt disease (STGD1): design of a clinical trial assessing a novel treatment and primary outcome measure. Invest Ophthalmol Vis Sci. 2018;59:1569.
17. Greenberg JP, Sherman J, Zweifel SA, et al. Spectral-domain optical coherence tomography staging and autofluorescence imaging in achromatopsia. JAMA Ophthalmol. 2014;132(4):437-445.
18. Cai CX, Light JG, Handa JT. Quantifying the rate of ellipsoid zone loss in Stargardt disease. Am J Ophthalmol. 2018;186:1-9.
19. Huang CH, Yang CH, Lai YJ, et al. Hyperreflective foci as important prognostic indicators of progression of retinitis pigmentosa. Retina. 2022;42:388-395.
20. Ling L, Gao F, Zhang Q, et al. optical coherence tomography angiography assessed retinal and choroidal microvasculature features in patients with retinitis pigmentosa: a meta-analysis. Biomed Res Int. 2019;2019:6723917.
21. Iovino C, Iodice CM, Pisani D, et al. Clinical applications of optical coherence tomography angiography in inherited retinal diseases: an up-to-date review of the literature. J Clin Med. 2023;12(9):3170.













_1773249222.png?auto=compress,format&w=75)










