Retina specialists are fortunate to practice in an era in which they can provide therapy to patients with geographic atrophy (GA) secondary to age-related macular degeneration (AMD). FDA-approved complement inhibitors have shown some efficacy and represent a new dawn for the retina specialty.
Still, therapies have been shown to only modestly slow disease progression, and no therapy has been shown to truly maintain or even restore vision in GA patients. We now hope to usher in the next generation of therapies that act by other mechanisms to enable better preservation, or even potential improvement, of vision.
Attacking the fundamental cellular pathogenesis of AMD and GA has drawn the attention of innovators seeking to improve the performance of GA therapies. At Aviceda Therapeutics, we have developed novel glycoimmune therapies by harnessing the body’s natural immune system to modulate inflammation by synthesizing highly selective ligands, with maximum specificity toward target immune cells and diseases, and aiming predominantly at the cells that cause the damage in GA. The company’s lead product candidate, AVD-104, is a glycan-coated nanoparticle with a dual mechanism of action. Most directly, it inhibits the activity of phagocytic/inflammatory macrophages, resolves VEGF-producing macrophages, and repolarizes activated microglia and macrophages to their neuroprotective state. Secondarily, it binds to complement factor H (CFH) to reduce (but not completely deplete) the overactivation of C3.
How AVD-104 Repolarizes Activated Macrophages to Resolution Macrophages in AMD Patients
In patients with AMD, it has long been known that macrophages are present at the leading edge of GA lesions. Disease activation of these macrophages leads to the release of proinflammatory mediators that create the conditions under which inflammation, apoptosis, neovascularization, and fibrosis flourish, leading to both GA and wet AMD.1-6 Repolarization of these proinflammatory macrophages to their healing state leads to resolution of disease activity, and represents a novel treatment mechanism for GA (Figure 1). AVD-104 repolarizes activated macrophages by mediating the sialic acid–Siglec interaction which is at the heart of most immune cell interactions.

Figure 1. The release of proinflammatory mediators occurs after activation of macrophages. The resulting inflammation, apoptosis, neovascularization, and fibrosis lead to both GA and wet AMD. The repolarization of macrophages leads to resolution of disease activity.
Siglecs, or sialic acid–binding immunoglobulin-like lectins, are receptors expressed on all immune cells, and function primarily as mediators of immune resolution (Figure 2).7-10 When bound to agonizing ligands, Siglec 7, 9, and 11 activity resolves inflammation and repolarizes activated macrophages to their resolution phenotype.

Figure 2. Sialic acid–binding immunoglobulin-like lectins, or Siglecs, are macrophage immune receptors whose modulation results in macrophages being repolarized to their resolution state.
Sialic acid is one of several sugars found in hundreds of different combinations on all mammalian cell surfaces. Immune cells identify all other cells as either “self” (i.e., native healthy cells) or “non-self” (i.e., diseased, damaged, or foreign cells) through the binding of Siglecs on their cell surfaces to the sialic acids (various glycan structures) on the presenting cells, which act as immune markers.10 If the binding of a macrophage Siglec to a cell’s sialic acid results in recognition of a “non-self” cell, the macrophage is polarized and an immune response is activated. If the Siglec on a polarized macrophage binds to a “self” sialic acid, then the macrophage is repolarized to a healing state and immune activity is resolved. This barcode-like cell recognition system helps modulate cellular immune activity.
Aviceda’s lead drug candidate, AVD-104, is a synthesized sialic acid-coated nanoparticle that binds to Siglecs on the surface of activated macrophages with very high affinity and thereby triggers repolarization of macrophages and restoration of the cell’s resolution state (Figure 3).
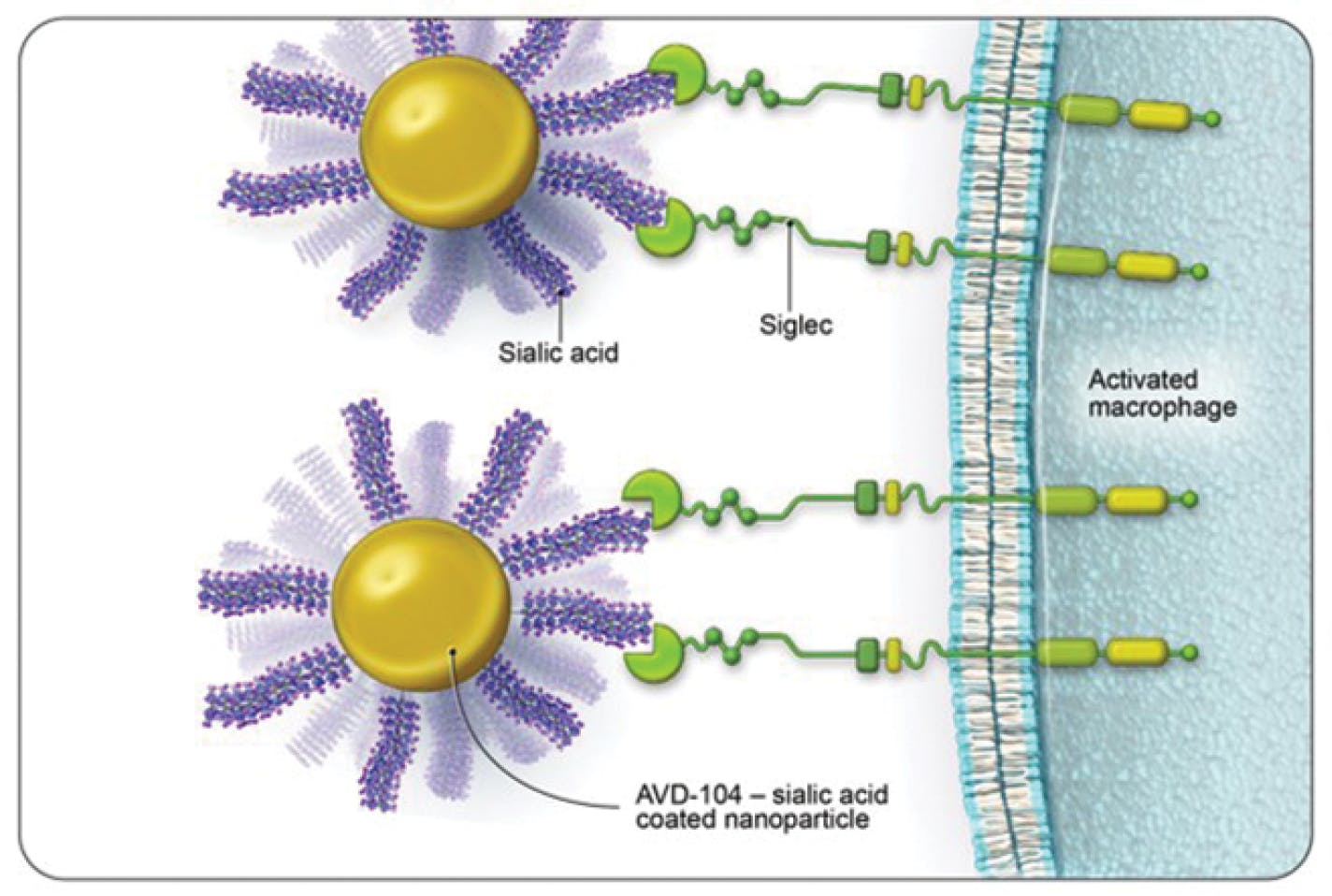
Figure 3. AVD-104 is a sialic acid-coated nanoparticle designed to bind to activated macrophage Siglecs to initiate repolarization and resolution of the immune response.
AVD-104 is delivered via intravitreal injection. Its molecular weight is 20 kDa, and it is 100 nm in size, which allows its penetration through the ILM. Data on its pharmacokinetic profile show that AVD-104 remains at significant levels 3 to 4 months after a single intravitreal injection.
AVD-104 also directly binds to CFH to downregulate complement amplification (Figure 4). This approach does not halt complement signaling through depletion, but rather dampens complement pathway amplification. This dual approach to treating GA in one molecule represents a novel approach to therapy, and looks to set a new standard in the management of GA.

Figure 4. The dual mechanism of action of AVD-104 allows simultaneous repolarization of activated macrophages and downregulation of complement amplification via binding directly to complement factor H on the cellular surface.
The Latest Data On AVD-104: The SIGLEC Trial
The safety and efficacy of AVD-104 for the treatment of GA is under investigation in the Phase 2/3 SIGLEC study, which is divided into two parts.
Part 1 of SIGLEC was a multi-center, open-label, safety and dose-escalation study. Researchers enrolled 30 patients with bilateral GA, assigning a single eye in each patient to one of four cohorts (0.1 mg, 0.5 mg, 1.0 mg, and 3.0 mg). Patients received a single dose of AVD-104 and were followed for 3 months.
AVD-104 had an excellent safety profile, and no ocular or systemic drug-related serious adverse events were observed. At the completion of the study, there had been no cases of ischemic optic neuropathy, retinal detachment, endophthalmitis, choroidal neovascularization, retinal vasculitis, or increased IOP.11
BCVA change from baseline at day 15, month 1, month 2, and month 3 was evaluated. Patients in the 1.0-mg and 3.0-mg dose cohorts experienced mean BCVA improvement from baseline of 4.8 ETDRS letters and 6.5 ETDRS letters, respectively (Figure 5).11 Of all patients, 60% (18/30) experienced BCVA gain in the study eye. Such visual improvements seen after a single dose of AVD-104 are exciting and signal the potential for maintenance or even some restoration of vision from GA rather than simply slowing eventual vision loss. These trends will be further explored in Part 2 of the SIGLEC study, which will be larger and longer than Part 1 of SIGLEC.

Figure 5. At 3 months in Part 1 of the Phase 2/3 SIGLEC study, patients in the 1.0-mg and 3.0-mg dose cohorts experienced mean BCVA gains from baseline of 4.8 letters and 6.5 letters, respectively.
The junctional zone of GA lesions, which is the area immediately outside of a GA lesion where progression occurs, was evaluated for signs of anatomic efficacy (Figure 6). Given that not all photoreceptors and RPE cells in the junctional zone are dead (some may merely be dying or stunned), rescuing cells in the junctional zone may be foundational to structural stabilization and improvement. As measured on fundus autofluorescence (FAF), 67% of patients demonstrated improved hyperautofluorescence 3 months after a single dose of AVD-104; another 10% of patients showed no enlargement of hyperautofluorescence at 3 months. The slowed growth of GA lesions as shown on FAF imaging supports the functional findings outlined above.

Figure 6. Preservation of living cells in the junctional zone, depicted here, may be foundational to stabilization and improvement of GA. In Part 1 of SIGLEC, hyperautofluorescence in the junctional zone improved from baseline in 67% of patients, signaling slowed GA lesion growth.
Part 2 of the SIGLEC study is actively enrolling patients.12 It is a multi-center, double-masked, randomized, controlled trial that will assess safety and efficacy outcomes of AVD-104 1.0 mg given monthly, AVD-104 2.0 mg given bimonthly, and an active comparator. Patients will be treated for 24 months. This head to head noninferiorty/superiority trial has a primary endpoint of rate of change of GA area (as measured by FAF) from baseline, at month 12; secondary endpoints include change from baseline in BCVA at month 12, prevention of BCVA loss from baseline, and loss of RPE or ellipsoid zone as measured by SD-OCT at month12.
The Future Of Therapy For GA
AVD-104’s dual mechanism of action has shown efficacy in GA patients, including visual improvement, with an excellent safety profile, and may well establish a new standard of care for GA treatment.
1. Cao X, Shen D, Patel MM, et al. Macrophage polarization in the maculae of age-related macular degeneration: a pilot study. Pathol Int. 2011;61(9):528-535.
2. Cruz-Guilloty F, Saeed AM, Echegaray JJ, et al. Infiltration of proinflammatory m1 macrophages into the outer retina precedes damage in a mouse model of age-related macular degeneration. Int J Inflam. 2013;2013:503725.
3. Ambati J, Atkinson JP, Gelfand BD. Immunology of age-related macular degeneration. Nat Rev Immunol. 2013;13(6):438-451.
4. Lurier EB, Dalton D, Dampier W, et al. Transcriptome analysis of IL-10-stimulated (M2c) macrophages by next-generation sequencing. Immunobiology. 2017;222(7):847-856.
5. Zizzo G, Hilliard BA, Monestier M, Cohen PL. Efficient clearance of early apoptotic cells by human macrophages requires M2c polarization and MerTK induction. J Immunol. 2012;189(7):3508-3520.
6. Bijarchian F, Taghiyar L, Azhdari Z, Baghaban Eslaminejad M. M2c macrophages enhance phalange regeneration of amputated mice digits in an organ co-culture system. Iran J Basic Med Sci. 2021;24(11):1602-1612.
7. Crocker PR, Paulson JC, Varki A. Siglecs and their roles in the immune system. Nat Rev Immunol. 2007;7(4):255-266.
8. Pillai S, Netravali IA, Cariappa A, Mattoo H. Siglecs and immune regulation. Annu Rev Immunol. 2012;30:357-392.
9. Gray MA, Stanczak MA, Mantuano NR, et al. Targeted glycan degradation potentiates the anticancer immune response in vivo. Nat Chem Biol. 2020;16(12):1376-1384.
10. Macauley MS, Crocker PR, Paulson JC. Siglec-mediated regulation of immune cell function in disease. Nat Rev Immunol. 2014;14(10):653-666.
11. Kaiser PK. Novel Sialic Acid–Coated Nanoparticle, AVD-104, Modulates Macrophages and Complement Dysfunction in Nonexudative AMD in the SIGLEC Phase 2/3 Clinical Trial. Paper presented at: Macula Society Annual Meeting; Palm Springs, CA; February 10, 2014.
12. A Multiple Dose Study of AVD-104 for Geographic Atrophy (GA) Secondary to Age-Related Macular Degeneration (AMD) (SIGLEC). ClinTrials.gov. NCT05839041. Updated February 6, 2024. Accessed February 16, 2024.
The views and opinions expressed here may not necessarily reflect those of Bryn Mawr Communications or Retina Today.



