AT A GLANCE
- The initial surgical approach for advanced diabetic retinopathy (DR) prioritizes techniques that achieve surgical goals while minimizing risks of reoperation.
- Indications for reoperation include recurrent vitreous hemorrhage, epiretinal membranes, residual vitreous traction, subretinal fluid, redetachment, and proliferative vitreoretinopathy.
- When reoperating on a challenging DR case, ensure all new retinal breaks are cleared of traction from residual vitreous and adjacent preretinal membranes.
Managing complex cases of diabetic retinopathy (DR) in the OR can pose significant challenges. The primary goals of surgical treatment include removal of vitreous hemorrhage (VH), relief of vitreoretinal traction, and application of laser to achieve quiescence, restoring visual function and mitigating the need for subsequent surgical interventions. However, when a second surgery becomes necessary, understanding the failures from the initial surgery and strategically planning for reoperation are critical for improving patient outcomes.
GETTING IT RIGHT THE FIRST TIME
The initial surgical approach for advanced DR prioritizes techniques that achieve surgical goals while minimizing risks that can lead to reoperation. Success begins with the management of preoperative and systemic risk factors. Collaboration with the patient’s primary care physician and endocrinologist to optimize blood pressure control and discuss anticoagulation medications in the perioperative period can decrease the risk of intraoperative and postoperative VH. Surgical scheduling should consider hemodialysis schedules and time for medical clearance, which, when not coordinated, can result in delays in surgical care. This is particularly important for patients receiving preoperative anti-VEGF injections, as surgical delays put the patient at risk for retinal detachment (RD) if surgery is unexpectedly canceled. Lastly, administering panretinal photocoagulation prior to surgery in areas of the retina not obscured by VH and away from areas of vitreoretinal traction can stabilize the retina and decrease the risk of complications during fibrovascular membrane dissection.
Beware of Hemorrhage
Early postoperative VH following diabetic vitrectomy can delay visual recovery, negatively affect a patient’s perception of surgical success, and increase the risk of postoperative complications. Mild postoperative VH can often be observed, but severe and persistent cases may require additional surgery. Several factors contribute to early postoperative VH: blood in the residual anterior vitreous, inadequate diathermy, relative hypotony after surgery, and residual traction on fibrovascular membranes. To reduce the risk of postoperative VH, surgeons can employ several measures, including lowering the infusion pressure to physiologic levels to identify sources of bleeding and removing as much anterior VH as possible, which is easier in pseudophakic eyes and with scleral depression. Surgeons can also perform a complete fluid-air exchange to remove residual VH and minimize oozing in the early postoperative period, confirm that sclerotomies are sealed (and, when necessary, suture wounds to prevent hypotony), and thoroughly segment and/or delaminate fibrovascular membranes. Triamcinolone staining can be useful to confirm the absence of residual vitreous adhesions. Additionally, preoperative and intraoperative anti-VEGF injections may be beneficial.1
Reduce Traction
Residual vitreoretinal adhesions and traction can lead to a series of complications, including recurrent VH, epiretinal membrane formation, and tractional RD that can progress to combined rhegmatogenous and tractional RD.
Successful relief of vitreoretinal traction begins with accurate identification of the posterior hyaloid face. Induction of a posterior vitreous detachment (PVD) in diabetic eyes can be challenging due to the presence of vitreous schisis and tight adhesions of the vitreous to the retinal surface.2 Triamcinolone acetonide staining can aid in the identification of the posterior hyaloid face. In some cases, a partial vitreous detachment may be present in the midperiphery.
A useful first step is to segment the posterior vitreous 360° from the vitreous base (Figure 1). Partial midperipheral vitreous detachments can often be identified and propagated circumferentially. Furthermore, segmentation of the anterior and posterior vitreous can minimize the risk of anterior breaks during membrane dissection.
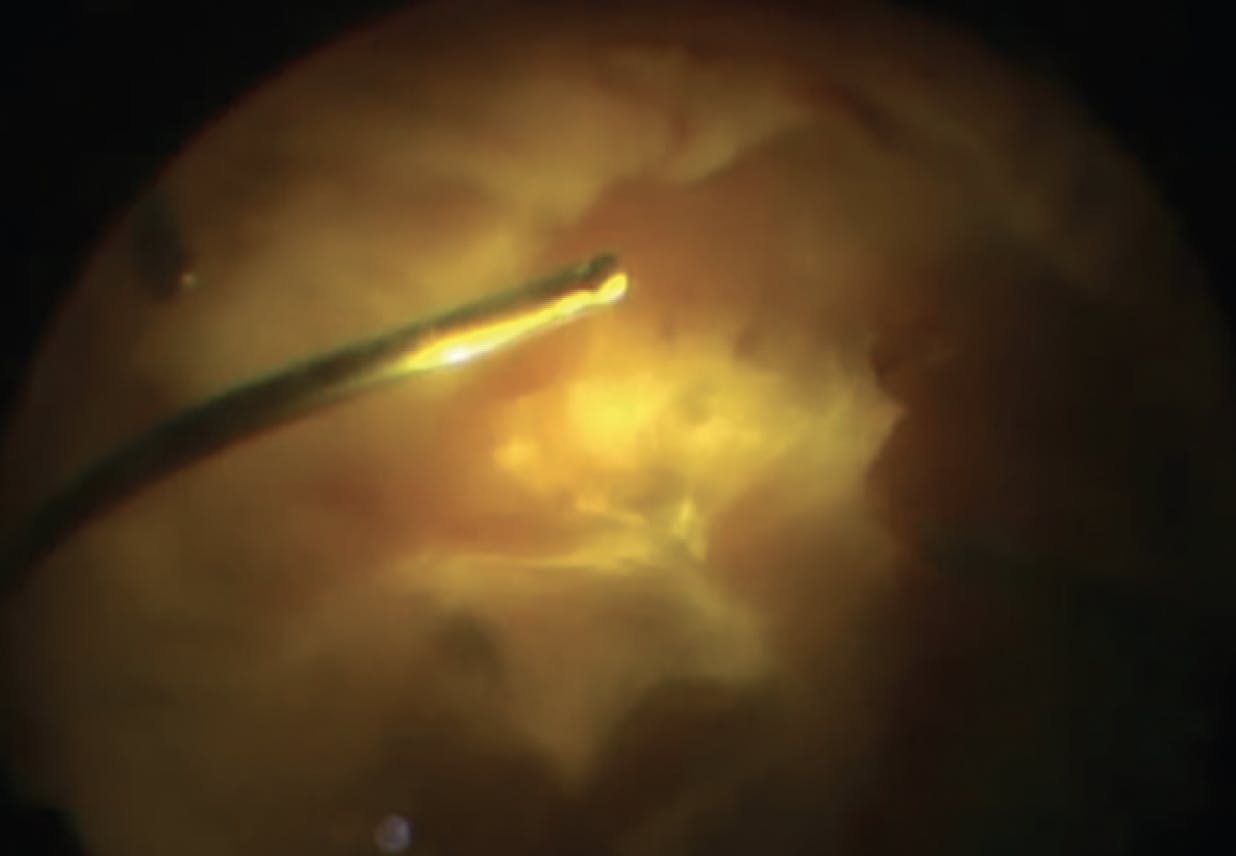
Figure 1. During diabetic vitrectomy, releasing anterior-posterior traction helps to prevent the development of vitreous base tears when dissecting tractional RDs.
When a midperipheral vitreous detachment is not present, it may be necessary to induce a PVD by aspirating or peeling the vitreous anteriorly from the optic nerve (Figure 2). When tackling broad fibrovascular membranes, segmentation into smaller, more manageable foci alone may be sufficient to achieve surgical goals (Figure 3). Segmentation can also minimize the risk of unwanted traction on adjacent areas of fibrosis when delamination is necessary.

Figure 2. Surgeons can use ILM forceps to induce a PVD over the nerve. This is a safe location to enter the correct surgical plane in the setting of vitreous schisis.

Figure 3. Segmentation of membranes into individual islands allows dissection of membranes without undue traction, decreasing the risk of iatrogenic retinal breaks.
Many approaches to segmentation and delamination have been described in the literature, including the use of a combination of the vitreous cutter, horizontal and vertical scissors, viscodissection, and bimanual techniques using chandelier illumination (Figure 4). We find that tractional membrane delamination over bullous RDs is more efficiently removed with bimanual techniques, whereas membrane delamination over non-bullous RDs can be achieved with the vitreous cutter alone. Furthermore, the vitreous cutter can be used as a multipurpose tool, functioning as a hook or pick to elevate membranes that can be cut to relieve traction.

Figure 4. With a bimanual technique, surgeons can use end-grasping forceps and horizontal scissors to dissect a tractional membrane from a detached retina, where the lack of counter-tractional forces can make delamination more challenging.
INDICATIONS FOR REOPERATION
Despite optimal management during initial diabetic vitrectomy, some patients may require additional surgery. Indications for reoperation include:
- Recurrent VH. This refers to early postoperative or recurrent VH that does not clear after a period of observation with or without the use of anti-VEGF injections.
- Secondary glaucoma. Persistent VH may lead to ghost cell glaucoma, where red blood cells obstruct aqueous outflow through the trabecular meshwork. This may occur in phakic, pseudophakic, or aphakic patients when the anterior hyaloid face is disrupted.
- Epiretinal membranes or residual vitreous traction. Epiretinal membranes or residual cortical vitreous over the macula may contract, resulting in blurred vision or visually significant metamorphopsia.
- Subretinal fluid. Persistent subretinal fluid may be slow to resorb even after successful release of traction. Monitoring with serial OCTs can help to confirm stable or improving subretinal fluid. When subretinal fluid is increasing in size or extent, reoperation should be considered for a likely rhegmatogenous etiology.
- Redetachment from new retinal breaks. Entry site breaks, stretch breaks from residual vitreoretinal traction, and microbreaks not identified during the primary vitrectomy may lead to postoperative RD.
- Proliferative vitreoretinopathy (PVR). Patients with combined rhegmatogenous and tractional RD or iatrogenic breaks at the time of initial surgery may be at risk for recurrent RD secondary to PVR.
APPROACH TO REOPERATION
When reoperation is indicated, the surgical plan should be tailored to address the reason for primary surgery failure. Additionally, the patient’s visual prognosis should be assessed to determine if reoperation is warranted. If a second surgery is performed, the same principles for primary surgery apply with some additional considerations:
- Consider phacoemulsification in phakic patients to allow for adequate visualization and more thorough anterior vitreous shaving.
- Confirm the presence of a complete PVD with the use of triamcinolone staining.
- Determine if complete segmentation/delamination of fibrovascular membranes is contributing to recurrent VH.
- Examine with scleral depression to help identify new retinal breaks, especially around prior sclerotomy sites.
- Ensure all new retinal breaks are cleared of traction from residual vitreous and adjacent preretinal membranes. We recommend delamination of all preretinal membranes within one to two disc areas from a retinal break.
- Consider adding a scleral buckle to support the vitreous base if anterior retinal breaks are present.
- For patients with secondary glaucoma or rubeosis iridis, comanage the case with a glaucoma specialist and consider tube shunt implantation and/or cyclophotocoagulation to normalize IOP.
- In patients with PVR or when residual traction cannot be easily removed, removal of preretinal membranes, focal or extended retinectomy, anterior vitreous base dissection, and long-acting tamponade (eg, silicone oil) may be necessary.
TRY AGAIN
Surgery for advanced DR requires careful planning and expert execution. Despite best practices during primary diabetic vitrectomy, patients may require reoperation for a variety of indications. When a second surgery is required, it is important to understand and address the failures of the primary surgery to achieve surgical success.
1. Smith JM, Steel DH. Anti-vascular endothelial growth factor for prevention of postoperative vitreous cavity haemorrhage after vitrectomy for proliferative diabetic retinopathy. Cochrane Database Syst Rev. 2015;2015(8):CD008214.
2. Schwatz SD, Alexander R, Hiscott P, Gregor ZJ. Recognition of vitreoschisis in proliferative diabetic retinopathy. A useful landmark in vitrectomy for diabetic traction retinal detachment. Ophthalmology. 1996;103(2):323-328.












_1773249222.png?auto=compress,format&w=75)










