Classification of AMD
The Beckman classification, widely adopted in epidemiologic studies, defines early AMD by the presence of medium sized drusen (63-125µm) without pigmentary changes, using X vein width at the edge of the optic nerve as an approximate internal calibration marker. Intermediate AMD is characterized by the presence of larger drusen (≥125µm) or pigmentary abnormalities, while late-stage AMD encompasses GA and neovascular AMD.
The Age-Related Eye Disease Study (AREDS) simplified classification system defines four-levels. Stage three (intermediate AMD) is defined by the presence of at least one large drusen (≥125µm) or multiple intermediate-sized drusen (>63µm), along with possible pigmentary changes and no late AMD in the fellow eye. Stage four (advanced AMD) represents the presence of choroidal neovascularization (CNV) or GA in one eye.
However, neither the Beckman nor AREDS classification systems account for subretinal drusenoid deposits (reticular pseudodrusen, or RPD), which are located above the retinal pigment epithelium (RPE). Recent findings from AREDS highlight RPD as a significant risk factor for progression to late-stage AMD,1 underscoring the necessity for refining current risk assessment tools.
Advances from the Classification of Atrophy Meeting (CAM)
CAM brought together an international panel of experts to establish a consensus classification system for structural changes detected by OCT, which precede atrophy secondary to AMD.
An OCT-defined correlate of GA, termed complete RPE and outer retinal atrophy (cRORA), has been proposed as an endpoint for atrophy developing in the context of intermediate AMD. The criteria for cRORA include: (1) a region of hypertransmission ≥250µm, (2) RPE attenuation or disruption ≥250µm, and (3) evidence of overlying photoreceptor degeneration. All these criteria must be met in the absence of RPE tear.2
The term incomplete RORA (iRORA) describes an earlier stage of GA, where OCT findings do not fully meet the cRORA criteria.3 A precursor stage, termed nascent GA, includes features such as subsidence of the outer plexiform layer and inner nuclear layer, and/or the presence of a hyperreflective wedge-shaped band, without choroidal signal hypertransmission or RPE attenuation or disruption.4
Additional subtypes include complete outer retinal atrophy (cORA) and incomplete outer retinal atrophy (iORA), typically associated with subretinal drusenoid deposits. These subtypes involve outer retinal atrophy without initial RPE loss.
Interpreting the intricate features revealed in a single B-scan can be challenging. To overcome this, ongoing efforts aim to develop fully automated models for quantifying key retinal features linked to early- and late-stage AMD.5 The integration of these tools into clinical practice is crucial for improving efficiency, particularly in busy clinics. Automated systems that seamlessly interface with electronic medical records could greatly enhance diagnostic accuracy, enabling timely and reliable assessments.
GA Progression and Predictive Factors
Progression from non-central to central GA takes on average 2.5 to 3 years (Figure 1), while progression from unilateral to bilateral GA averages around seven years.6 Lesion growth occurs at a rate of approximately 1.78mm2 per year.6 The Proxima A and B studies have underscored the severe functional impact of GA, highlighting its rapid progression within two years, even in earlier disease stages such as unilateral GA.7
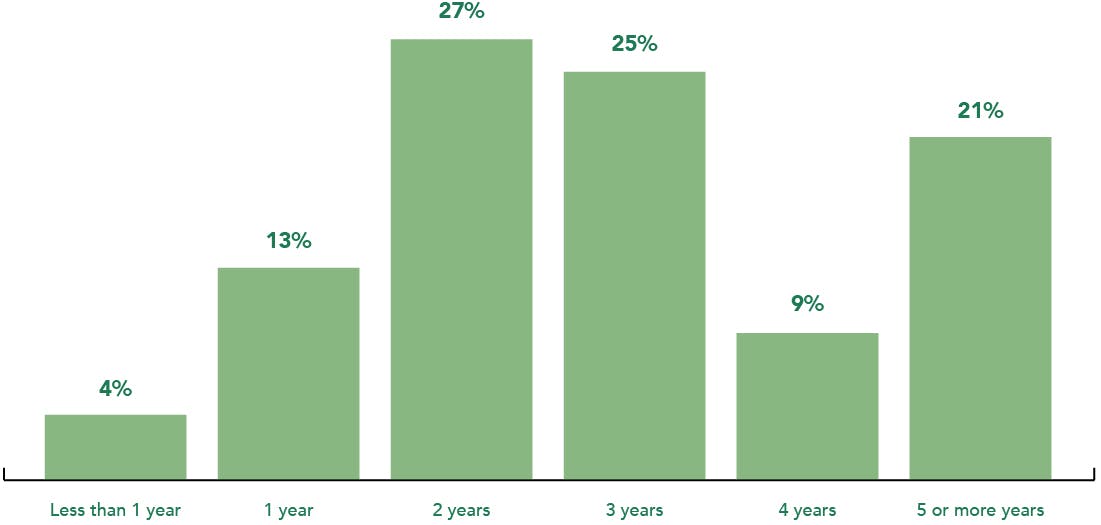
Figure 1. Based on the 2023 EURETINA Clinical Trends Survey, clinicians estimate that, on average, it takes 3 years for non-central GA to progress to central GA.*
Several key factors influence the rate of GA progression, namely baseline lesion size, lesion centrality, lesion focality, and the status of the fellow eye.8 Additional predictors include a larger baseline low luminance deficit,7 lesion edge location,9 the presence of specific FAF patterns,10 and the presence of RPD or subretinal drusenoid deposits.11
OCT imaging markers associated with GA progression include: (1) choroidal hypertransmission, caused by disruption of overlying RPE, (2) subretinal drusenoid deposits or pseudodrusen, (3) persistent basal laminar deposits, and (4) hyperreflective foci.

Conclusions
Early intervention is essential to slow or prevent GA progression. Anatomical changes that precede vision loss present clinically relevant primary endpoints. Redefining clinical trial endpoints is critical to advancing emerging therapies. Furthermore, AI is poised to play a transformative role in automating and streamlining workflows in busy clinical settings, ultimately improving both efficiency and the quality of patient care.
*European Society of Retina Specialists (EURETINA) Clinical Trends Survey 2023.
1. Agrón, E. et al. An Updated Simplified Severity Scale for Age-Related Macular Degeneration Incorporating Reticular Pseudodrusen: Age-Related Eye Disease Study Report Number 42. Ophthalmology 131, 1164–1174 (2024).
2. Sadda, S. R. et al. Consensus Definition for Atrophy Associated with Age-Related Macular Degeneration on OCT: Classification of Atrophy Report 3. Ophthalmology 125, 537–548 (2018).
3. Guymer, R. H. et al. Incomplete Retinal Pigment Epithelial and Outer Retinal Atrophy in Age-Related Macular Degeneration: Classification of Atrophy Meeting Report 4. Ophthalmology 127, 394–409 (2020).
4. Wu, Z. et al. Prospective Longitudinal Evaluation of Nascent Geographic Atrophy in Age-Related Macular Degeneration. Ophthalmol Retina 4, 568–575 (2020).
5. Liefers, B. et al. Quantification of Key Retinal Features in Early and Late Age-Related Macular Degeneration Using Deep Learning. Am J Ophthalmol 226, 1–12 (2021).
6. Lindblad, A. S. et al. Change in area of geographic atrophy in the Age-Related Eye Disease Study: AREDS report number 26. Arch Ophthalmol 127, 1168–1174 (2009).
7. Holekamp, N. et al. Natural History of Geographic Atrophy Secondary to Age-Related Macular Degeneration: Results from the Prospective Proxima A and B Clinical Trials. Ophthalmology 127, 769–783 (2020).
8. Keenan, T. D. et al. Progression of Geographic Atrophy in Age-related Macular Degeneration: AREDS2 Report Number 16. Ophthalmology 125, 1913–1928 (2018).
9. Lindner, M. et al. Directional Kinetics of Geographic Atrophy Progression in Age-Related Macular Degeneration with Foveal Sparing. Ophthalmology 122, 1356–1365 (2015).
10. Holz, F. G. et al. Progression of geographic atrophy and impact of fundus autofluorescence patterns in age-related macular degeneration. Am J Ophthalmol 143, (2007).
11. Marsiglia, M. et al. Association between geographic atrophy progression and reticular pseudodrusen in eyes with dry age-related macular degeneration. Invest Ophthalmol Vis Sci 54, 7362–7369 (2013).

_1749218012.png)