Pneumatic retinopexy is an excellent addition to the retina surgeon’s armamentarium. It’s very rewarding to take a patient with hand motion vision back to 20/20 in less than 2 weeks, all while avoiding the OR. However, becoming adept at pneumatic retinopexy requires practice. Incorporating a new technique early in solo practice can be daunting, making effective skill acquisition during training even more important. Here, I present some pearls in pneumatic retinopexy management and surgeon development.
PEARL NO. 1: PRACTICE MAKES PERFECT
Beyond all other tips and tricks, becoming better at pneumatic retinopexy requires practice. As with any procedure, particularly one with as many possible variations as pneumatics, the art is in understanding the nuances. And that can only happen with experience and the confidence gained through repeat success.
You must accept that failure will occur, as is the case with any other skill acquired during fellowship or residency. The fear of failure can be debilitating; however, it is important to realize that a failed pneumatic does not necessarily entail a worse final outcome. In fact, the opposite is often true. A macula-off retinal detachment (RD) can be transformed into a macula-on RD by the time you are in the OR for a vitrectomy. A macula-off detachment can have an attached fovea within hours of bubble injection.
PEARL NO. 2: START EASY
Depending on who you ask, indications for pneumatic retinopexy can range from restrictive to liberal. Gaining confidence early, however, is more easily done when the ideal candidate is in your chair. Patients with quadratic RDs with single superior tears who are phakic and have no additional peripheral pathology and a clear posterior vitreous detachment make for a much easier initial learning curve.
After five to 10 successful cases, you will feel more comfortable with the mechanics and the pace of pneumatic retinopexy. By this point, you’ll likely have mastered the large-volume paracentesis and gas bubble injection without excessive fish eggs and be accustomed to the pace of bubble expansion and the timing of staged laser retinopexy, helping you feel more confident in proceeding with more complex cases.
This is also the time to experiment with staged laser versus cryotherapy at the time of the pneumatic retinopexy. Both approaches have pros and cons, so weighing both is important. While cryotherapy has the benefit of potentially being a one-and-done treatment, patients will have significant chemosis and postoperative discomfort. Laser, on the other hand, can be more difficult through a gas bubble, particularly on a blonde fundus. Bullous RDs can mask the true location of a break, particularly one that is not marked at the time of the pneumatic retinopexy.1
PEARL NO. 3: PATIENT SELECTION AND EXPECTATIONS
Patient selection is critical. In the OR, patients are sedated, and you are in control. In the clinic, the patient is awake, and participation is critical. During the initial examination, consider how the patient tolerates the depressed examination. Let them know that the pneumatic will involve significantly more manipulation in the chair both on the initial day and during follow-up. Make sure they know that the procedure may have a lower rate of primary success, require more manipulation in the clinic, and, in the event of failure, result in the same surgery that is initially being deferred.2,3
It is useful to know what kind of support, if any, patients have at home and how much of a burden additional clinic visits might be for them.4 A patient with little support and a long commute is at higher risk for loss to follow-up than the highly motivated patient who has a care provider who is committed to taking them to their frequent visits and enforcing positioning. While we do not have a regimented visit schedule, we see most patients the first day following pneumatic retinopexy. At this visit, we check IOP and can typically apply laser, as even the most bullous of detachments are flat with the use of a large bubble. Patients are then typically extended to weekly visits until the gas resolves (one to three more visits) and then to monthly visits in a pattern similar to surgical patients.
Clinically, consider peripheral pathology, such as atrophic holes, lattice, or even abnormal pigmentary changes. Frequently, a pneumatic will fail not because of inadequate treatment of the original break, but because a new break develops or is initially missed. Meticulous laser of retinal holes and lattice degeneration at the original visit, as well as during the follow-up period once the bullous fluid has resolved, is critical to long-term success.
PEARL NO. 4: LARGE BUBBLE
One of the most effective tricks that will significantly increase your success rate is using a large gas bubble. The large bubble allows for more effective examination during follow-up, makes staged laser significantly easier, more quickly displaces submacular fluid, and makes positioning much easier for the patient (Figure 1).
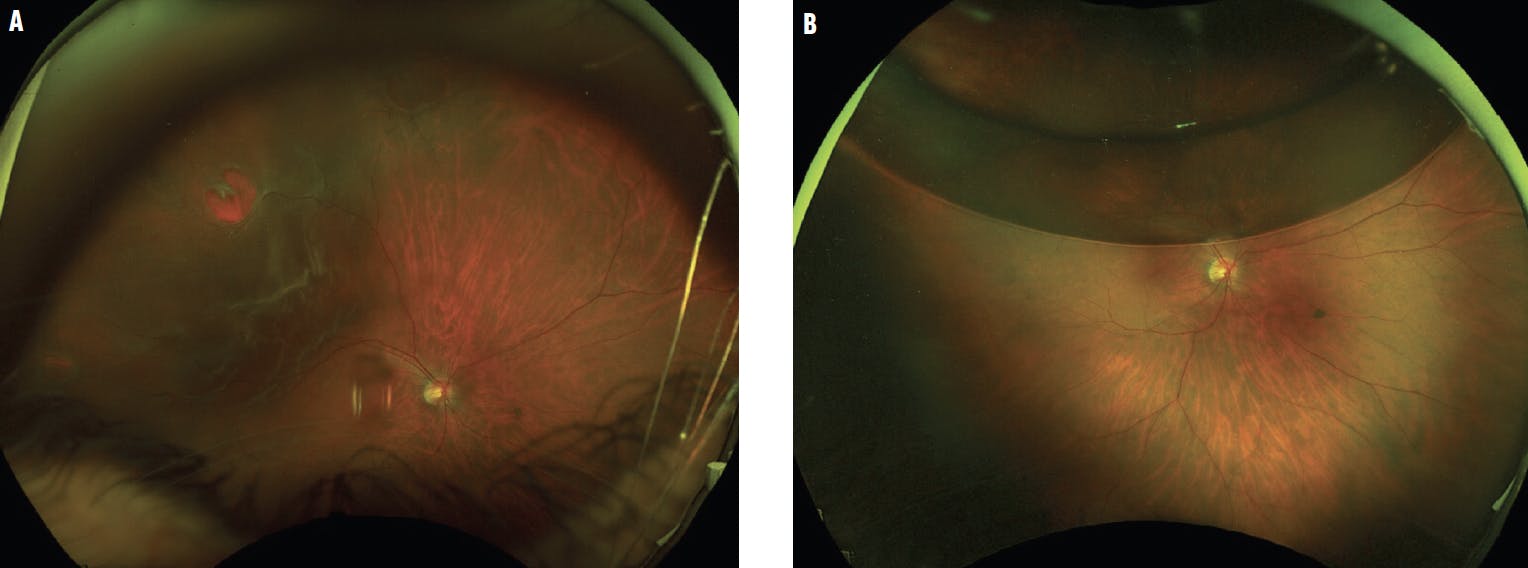
Figure 1. This macula-off RD had breaks at the 9, 10, and 12 clock hours (A). Pneumatic retinopexy with a large bubble allowed for complete staged laser on postoperative day 2 following gas injection (B). At the postoperative month 4 visit, the patient’s VA was 20/25, and the retina remained attached.
To achieve a large bubble, you must become comfortable with a large-volume anterior chamber paracentesis. In our clinic, we perform the procedure with a speculum while the patient lies on their back in the chair and looks up at the ceiling. With practice, you can use a cotton tip applicator—with 0.4 cc to 0.5 cc taps—to attain persistent pressure near the limbus, even in the phakic patient. This allows you to confidently inject 0.6 cc of gas without having to worry about next-day IOP spikes.
For added security, you can consider prescribing prophylactic dorzolamide/timolol alongside a steroid/antibiotic combination as part of the standard postoperative drop regimen, which can be tapered off during 1 to 2 weeks of follow-up.
PEARL NO. 5: COMMIT TO THE CAUSE
With the fundamentals in place, progressively more complex cases can quickly become successful cases.
The rare case in which the break cannot be identified, but a superior location is suspected, can be positioned accordingly. Apply laser to the superior clock hours where the break is expected (we prefer laser in cases where broad areas require treatment).5
In pseudophakic cases where you might expect to find multiple small far peripheral breaks, close inspection on depressed examination with treatment of areas of peripheral vitreous traction can lead to similar success rates as the clear phakic patient (Figure 2).

Figure 2. A patient presented with a pseudophakic macula-off rhegmatogenous RD with an operculated retinal hole and inferior lattice (A). The patient’s retina remained attached 6 months after pneumatic retinopexy (B).
Minor setbacks do not have to spell failure. Occasionally, a new tear develops, less gas is introduced than expected, or the patient metabolizes the gas faster than expected. Find the solution. Rather than defaulting to the OR, consider careful re-assessment of the current RD. Rebubbling with positioning to the causative break can turn an apparent failure into a success (Figure 3).

Figure 3. A patient presented with a history of RD and a current macula-off detachment (A). He underwent cryotherapy and pneumatic retinopexy, but a relatively small bubble was used, and he developed new subretinal fluid during follow-up (B). A second bubble was placed, and additional broad peripheral laser was applied. His retina remained attached at the postoperative month 5 visit (C).
1. Muni RH, Kertes PJ. Marking of retinal breaks in detached retina with laser photocoagulation before pneumatic retinopexy: a prospective case series. Retina. 2009;29(3):405-408.
2. Hillier RJ, Felfeli T, Berger AR, et al. The pneumatic retinopexy versus vitrectomy for the management of primary rhegmatogenous retinal detachment outcomes randomized trial (PIVOT). Ophthalmology. 2019;126(4):531-539.
3. Stewart S, Chan W. Pneumatic retinopexy: patient selection and specific factors. Clin Ophthalmol. 2018;12:493-502.
4. Al-Falah M, Muni RH, Kertes PJ. Pneumatic retinopexy: common myths dispelled. Retina Today. 2023;18(1):22-24.
5. Lincoff H, Gieser R. Finding the retinal hole. Arch Ophthalmol. 1971;85(5):565-569.












_1773249222.png?auto=compress,format&w=75)










