Perioperative vision loss in ocular surgery is rare. Among the most common etiologies are retinal vascular ischemia, direct optic nerve trauma during air-fluid exchange or perioperative anesthesia, and nonarteritic anterior ischemic optic neuropathy (NAION).1 In cases of vision loss related to retinal or optic nerve vascular ischemia, the perfusion pressure of the globe during surgery is essential. Adequate blood flow to organs such as the eye occurs due to a pressure gradient. Here, we present a case in which low intraoperative ocular perfusion pressure secondary to systemic hypotension resulted in transient ocular ischemia (Video).
CASE REPORT
A 73-year-old man presented with a several-month history of decreased vision in the left eye. Ophthalmic examination was notable for decreased VA to 20/80, a normal anterior segment examination, and evidence of an epiretinal membrane. The patient underwent uncomplicated pars plana vitrectomy (PPV) with epiretinal membrane peeling without internal limiting membrane removal.
Five months following the initial surgery, the patient developed a recurrent epiretinal membrane, and a decision was made to proceed with repeat PPV, epiretinal membrane removal, and additional internal limiting membrane peeling. At the onset of the vitrectomy, progressive optic nerve head pallor, boxcarring of the retinal vessels, and signs of dense retinal ischemia were noted (Figure 1).
Examination of the periorbita revealed no evidence of retrobulbar hemorrhage or orbital compartment syndrome. The decision was made to lower the preset infusion pressure from 30 mm Hg to 10 mm Hg. As machine errors can occur, we also confirmed a low IOP by manually palpating the globe. The patient had a systemic blood pressure of 80/60 mm Hg and a mean arterial pressure of 67 mm Hg. Given the concern for decreased ocular perfusion, intravenous fluids and vasopressors were administered to temporarily increase the patient’s mean arterial pressure.
Over the next several minutes, the patient’s systemic blood pressure increased to 110/90 mm Hg, and we noted a return of retinal perfusion (Figure 2). We decided to proceed with PPV and membrane peeling as planned. The rest of surgery was uneventful. At postoperative month 1, the patient’s VA was 20/100, with a posterior segment examination revealing evidence of temporal optic nerve head pallor, suggesting the possible development of (NAION).
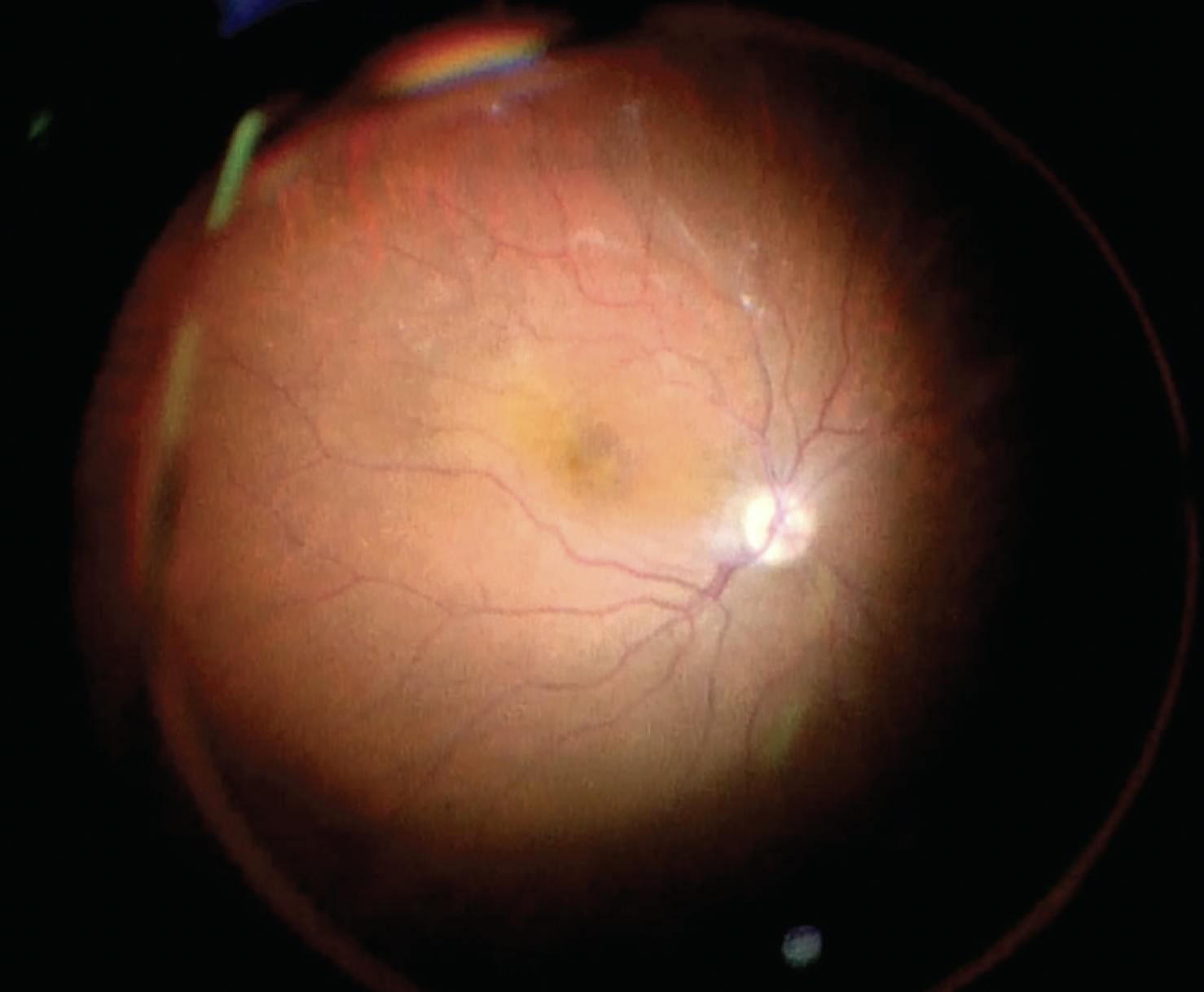
Figure 2. A few minutes after increasing the patient’s mean arterial pressure, we noted retinal reperfusion.
DISCUSSION
Maintaining blood flow to all ocular structures during surgery depends on an adequate mean ocular perfusion pressure (MOPP). This is calculated by subtracting the IOP from the mean ophthalmic arterial pressure, which is the mean arterial blood pressure adjusted to the level of the eye.2 Prolonged alteration of either the IOP or the mean ophthalmic arterial pressure during surgery can lead to ocular ischemia and a risk of permanent vision loss. Most hypothesized mechanisms suggest a link between intraoperative hypotension and/or increased IOP.3
Our patient experienced transient retinal ischemia due to intraoperative systemic hypotension, most likely related to anesthetic sedation in the setting of preoperative low blood pressure. He did not have significant cardiovascular comorbidities; however, advanced age and atherosclerotic vascular changes can contribute to altered optic nerve perfusion regulatory mechanisms.4
Ellabban et al described the occurrence of a central retinal artery occlusion (CRAO) during vitrectomy that resolved following induction of a posterior vitreous detachment.5 The researchers thought the cause of the CRAO was related to the local anesthetic bolus and direct compression of the central retinal artery, as there was no evidence of systemic hypotension or elevated IOP. They hypothesized that detaching the hyaloid from the optic disc may have released any abnormal adhesion around the central retinal artery. Suction from the vitreous cutter may have also created a negative pressure force that dislodged a potential thrombus.5
Other possible links between local anesthesia and a CRAO during ocular surgery have been described, including elevated IOP with secondary occlusion and a localized vasoconstrictive effect due to adrenaline in the anesthetic.6
The temporal optic nerve head pallor noted postoperatively in our patient likely represents the development of ischemic optic neuropathy. NAION is a multifactorial disease resulting from hypoperfusion of the prelaminar optic nerve head. NAION following vitrectomy has been reported, and while exact mechanisms are unclear, it is likely that decreased ocular perfusion pressure during surgery precipitated ischemia of the anterior optic nerve. Other factors contributing to NAION after vitreoretinal surgery may include direct optic nerve head trauma due to stretching and elongation of axons during the separation of the posterior hyaloid.
Rossi et al used a novel device to calculate the MOPP during PPV.2 The authors reported both a reduction in the mean arterial pressure due to anesthetic sedation and periods of increased IOP throughout surgery. This combination resulted in a reduction in the MOPP below critical limits, placing the eye at risk of ischemic damage due to limited autoregulation at the level of the optic nerve head. Prior studies have also measured intraoperative IOP during a variety of procedures, including cataract extraction and scleral buckle placement.
It is critical to remain vigilant of the retinal and optic nerve perfusion pressure during vitrectomy and to balance the intraocular infusion pressure against the blood pressure. The typical infusion cannula pressure may need to be reduced to improve optic nerve and retinal perfusion pressure. The best way to maintain an adequate MOPP is likely appropriate control of IOP during intraocular surgery; however, increased intravenous fluids and vasopressors may be needed in cases of extremely low blood pressure under sedation.
1. Jain N, McCuen BW 2nd, Mruthyunjaya P. Unanticipated vision loss after pars plana vitrectomy. Surv Ophthalmol. 2012;57(2):91-104.
2. Rossi T, Querzoli G, Gelso A, et al. Ocular perfusion pressure control during pars plana vitrectomy: testing a novel device. Graefes Arch Clin Exp Ophthalmol. 2017;255(12):2325-2330.
3. Hayreh SS. Ischemic optic neuropathies - where are we now? Graefes Arch Clin Exp Ophthalmol. 2013;251(8):1873-1884.
4. Riva CE, Hero M, Titze P, Petrig B. Autoregulation of human optic nerve head blood flow in response to acute changes in ocular perfusion pressure. Graefes Arch Clin Exp Ophthalmol. 1997;235(10):618-626.
5. Ellabban AA, Patil AD, Costen MT, Babar AR. Central retinal artery occlusion during vitrectomy: Immediate retinal revascularization following induction of posterior vitreous detachment. Am J Ophthalmol Case Rep. 2018;9:38-40.
6. Swamy BN, Merani R, Hunyor A. Central retinal artery occlusion after phacoemulsification. Retin Cases Brief Rep. 2010;4(3):281-283.













_1773249222.png?auto=compress,format&w=75)












