Tyrosine kinase inhibitors (TKIs) have garnered a lot of interest of late as a therapeutic strategy in retinal vascular disease. What is a TKI, and why should the retina community be paying attention?
Peter K. Kaiser, MD: When we speak colloquially about vascular endothelial growth factor (VEGF) activation, what we mean is that when VEGF binds to its receptor tyrosine kinase (RTK), it initiates a cascade of tyrosine kinase activation that triggers the signaling pathways that ultimately result in the signs of retinal vascular disease that we identify in the clinic: angiogenesis, increased permeability, etc. The current primary strategy for stopping that is anti-VEGF agents that work in the extracellular space, with the limitation that they need to be continually injected to achieve enough VEGF suppression to keep the disease in check. That’s a balance that, frankly, we don’t always achieve in the clinic. TKIs are small molecules that work intracellularly to stop the downstream activation of VEGF and other tyrosine kinase cascades. TKIs work whether VEGF is present extracellularly and interacting with the RTK or not. Another advantage TKIs have is they can be used to target multiple pathways: for example, platelet-derived growth factor is also involved in angiogenesis, and it is not targeted by anti-VEGF agents.
TKIs have been pursued in other parts of medicine. What can ophthalmology learn from that experience?
Dr. Kaiser: Many of the TKIs being tested in ophthalmology actually got their start in oncology. For instance, axitinib is FDA-approved for the treatment of patients with advanced renal cell carcinoma. Sunitinib, another TKI being tested in ophthalmologic indications, has indications for renal cell carcinoma, gastrointestinal tumors, and neuroendocrine tumors. Taken together, that should give us as ophthalmologists some confidence in the mechanism of action, because there are many similarities in eye disease and cancer with respect to new vessel growth and leakage.
Axitinib is the drug being used in Ocular Therapeutix’s pipeline candidate OTX-TKI, which is being developed for treatment of retinal vascular diseases: age-related macular degeneration (AMD), diabetic macular edema (DME), and retinal vein occlusion (RVO). What is important for ophthalmologists to know about OTX-TKI?
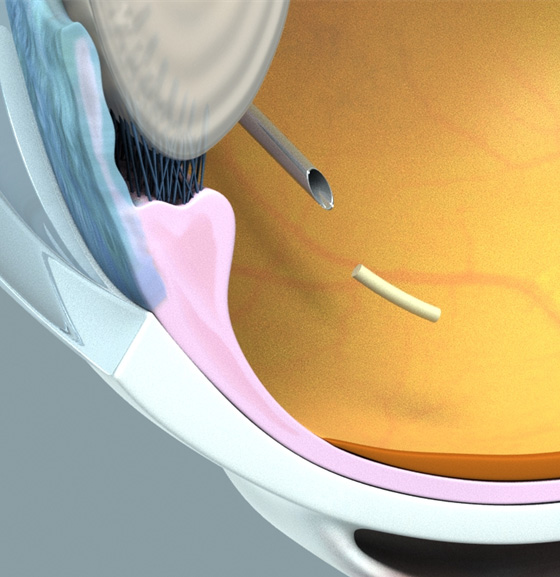
Antony Mattessich: Technically speaking, OTX-TKI is an investigational bioresorbable, intravitreal hydrogel implant incorporating axitinib, a small molecule TKI with anti-angiogenic properties, that is being evaluated for the treatment of wet AMD and other retinal diseases (Figure).1 At Ocular, we see OTX-TKI as a potential solution to a problem. To date, intravitreal anti-VEGF therapy has been incredibly successful in improving outcomes in these eye diseases, but the complexities of figuring out the ideal interval and managing the associated treatment burden is onerous for patients and providers. That’s a problem, and we think our technology may provide a solution in solving that problem, because our proprietary bioresorbable polymer matrix, a polyethylene glycol (PEG) hydrogel, can be designed to elute a targeted amount of drug for the intended duration of therapy. With OXT-TKI, we have taken the most potent TKI available, axitinib, and formulated it within our proprietary polymer matrix. Ocular Therapeautix has built strong science to support the mechanism of action associated with TKIs, with axitinib in particular, and we also have relevant market experience with the hydrogel platform in the form of commercially-available DEXTENZA® (dexamethasone ophthalmic insert) 0.4 mg. That foundation gives us cautious optimism in moving the OTX-TKI development program forward.
The pipeline in retinal vascular disease is robust and highly competitive. Is hydrogel how Ocular will differentiate itself?
Mr. Mattessich: Ocular is focused on finding the perfect marriage between the right polymer and the right molecule with the right potency to be able to solve the basic problem in treatment of retinal vascular disease, which is durability. Our approach makes us different. Along with the fact we are working with a highly-targeted molecule, axitinib, our hydrogel technology is one of the most important things that sets us apart in this crowded space. The hydrogel platform is exceedingly programmable and provides an adjustable medium into which we can introduce our drug of interest, in this case, axitinib. The main advantage of the polymer is that it eventually resorbs by bulk hydrolysis. That means there is no build-up of particle fragments in the eye to create visual disturbances.2 It also means a potential lower risk for immunogenicity. In fact, in its various iterations, the hydrogel platform, which is completely synthetic, has been used in over 7 million patients to date with various drugs and in multiple disease states, in some applications in very large amounts, and yet we have not become aware of any immune responses that have been triggered by the use of hydrogel. We look forward to further testing and in-human studies, and after that, hopefully partnering with the retina community to answer some of the challenges faced in real-world clinical practice.
Dr. Kaiser: First, we can appreciate some class effects with TKIs in terms of their mechanism, but they’re all different, with different targets, different formulations, and perhaps most importantly, different ways of delivering the active drug. Early efforts to develop TKIs that were delivered topically or in pill form, or just put in solution and injected to the eye, were not as successful as we hoped, but that is not necessarily attributable to the strategy of inhibiting tyrosine kinase activity. For example, in previous studies, oral TKIs were effective, but yielded significant systemic side effects;3 eye drops demonstrated biologic effect but were associated with ocular toxicity;4 and intravitreal injections proved too short-acting to be useful.5 The underlying theme here is that the right drug had not yet been paired with the correct delivery mechanism. The sustained-release polymer used in OTX-TKI seems to answer that question, with evidence suggesting it delivers consistent levels of active drug for 6 to 9 months.1 By doing that, we get the benefit of a TKI plus the longevity that previous programs did not achieve, and the hope is that we eventually get access to this to start addressing treatment burden.
Can you share an update OTX-TKI’s development program?
Dr. Kaiser: Based on positive findings in preclinical work, and with insight into the safety profile of the hydrogel platform with DEXTENZA and other pipeline programs, OTX-TKI is being tested in two phase 1 studies. A phase 1 study is being conducted in Australia, which recruited treatment-naive and previously treated wet AMD patients with retinal fluid. The study design was intended to determine if OTX-TKI can yield disease modification in terms of fluid reduction, which is something that has not been tested previously with other TKIs. Other TKI programs recruited patients who were relatively well-controlled on anti-VEGF agents, with an endpoint of maintaining that control and reducing the need for anti-VEGF rescue. Ocular has set a high bar with the Australia phase 1 study, but again, the strong science on hydrogel and axitinib in AMD support the trial design. A second phase 1 study based in the United States, recruited previously treated patients with no retinal fluid and has similar endpoints as other TKI studies such as prolongation of the need for anti-VEGF rescue. There will be a data readout of a preplanned analysis on the phase 1 US study study at the upcoming AAO meeting in Chicago.6 I can share that Ocular has already started thinking about aspects of a phase 2 study design given the positive outcomes from the Australia phase 1 study. The company is working on a second generation implant, based on some of the lessons learned in the phase 1 studies, to optimize the formulation.
One of the advantages of a versatile platform technology that affects sustained and localized drug delivery is that it has potential applications beyond the retina. Does Ocular’s pipeline extend to any other indications in eye care?
Mr. Mattessich: Yes it does. Beyond retina, our pipeline includes our glaucoma drug candidate in phase 2 clinical development, OTX-TIC, a travoprost intracameral implant, and OTX-DED and OTX-CSI for short term and chronic dry eye disease, respectively. Returning to retina, OTX-TKI is our anchor, and we are encouraged by what we have already learned about this technology, but it’s not our only approach in retina. We have two monoclonal antibodies in complement inhibition that are in the early stages of development that may have longer residence time in the retina—and we believe we can extend that even further with our polymer chemistry. We also have a very exciting gene therapy program where we are exploring the idea of releasing the viral vector over an extended period of time to achieve better transduction with less immune response from the body. Beyond that, as a company, one of the key reasons we are excited to work with Dr. Kaiser and other members of our Scientific Advisory Board is to learn from their substantial clinical experience about other potential targets in ophthalmology.
Watch Related Videos on OTX-TKI From Ocular Therapeutix
For more information on OTX-TKI, visit www.ocutx.com/research/otx-tki
1. Boyer DS, et al. Evaluating safety, tolerability and biological activity of OTX-TKI, a hydrogel-based, sustained-release intravitreal axitinib implant, in subjects with neovascular age-related macular degeneration: interim analysis of a phase 1 clinical trial. Presented at the AAO Annual Meeting. November 13-15, 2020. Virtual.
2. Csaky K, et al. Evaluating safety, tolerability and biological activity of OTX-TKI, a hydrogel-based, sustained-release intravitreal axitinib implant, in subjects with neovascular age-related macular degeneration: preliminary findings from a phase 1 trial. Presented at the EURETINA Annual Meeting. October 2-4, 2020. Virtual.
3. 3.Cohen MN, et al. APEX: a phase II randomised clinical trial evaluating the safety and preliminary efficacy of oral X-82 to treat exudative age-related macular degeneration. Br J Ophthalmol. 2021;105(5):716-722.
4. PAN-90806: Once-daily topical anti-VEGF eye drop for wet AMD and other neovascular eye disease. Presented at Ophthalmology Innovation Summit at the AAO Annual Meeting. October 10, 2019. San Francisco, CA.
5. Prenner JL, Halperin LS, Rycroft C, et al. Disease burden in the treatment of age-related macular degeneration: findings from a time-and-motion study. Am J Ophthalmol. 2015;160(4):725-31.e1.
6. Khanani AM, et al. Interim 28-weeks data from a phase 1 US study of sustained-release axitinib hydrogel implant (OTX-TKI) in previously treated wet AMD subjects. Presented at the AAO Annual Meeting. September 30-October 3. Chicago, IL. Abstract available at: https://aao.apprisor.org/apsSession.cfm?id=PO359
The article discusses an investigational product, OTX-TKI.
Its efficacy and safety profile has not been established and it has not been approved by the FDA.