Dr. Noemi Lois
The Diabetic Macular Oedema aNd Diode Subthreshold micropulse laser (DIAMONDS) clinical trial was commissioned and funded by the United Kingdom’s National Institute for Health and Care Research. Set within specialist hospital eye services at 16 sites in the United Kingdom, the trial used the Iridex IQ 577® Laser in MicroPulse® mode to evaluate the clinical effectiveness, safety, and cost-effectiveness of subthreshold MicroPulse laser therapy (SML) compared with continuous-wave laser therapy (CWL)* for the treatment of people living with diabetes with center-involved DME.
Patients were followed for 24 months, and SML therapy was found to have equivalent efficacy and cost to CWL, suggesting that either treatment could be offered to patients with central involved DME of <400 µm suitable for macular laser treatment.1
- DIAMONDS was a prospective, double-masked clinical trial carried out at 16 UK sites.
- 266 patients were recruited, with 133 randomized into the micropulse arm for treatment with the Iridex IQ 577 laser and the TxCell™ pattern scanning delivery device, and 133 randomized into the continuous-wave arm and treated with 532-nm laser.
- Investigators used standardized protocols.
- Participants were followed for 2 years.
Background | Dr. Noemi Lois
The absence of a scar or burn following SML led to some healthcare professionals to doubt the effectiveness of SML compared to CWL. This reluctance to believe that SML was effective existed despite the several small randomized clinical trials suggesting that SML was as effective as CWL.
Recognizing the need for a trial that was robust and that evaluated outcomes not previously investigated, the United Kingdom’s National Institute for Health and Care Research commissioned a study, which was then conducted with the support of the Northern Ireland Clinical Trials Unit. The trial included patient input from the beginning. In the design phase, patients with diabetes and DME helped to identify outcomes central to people living with DME. Maintaining good sight and, specifically, meeting driving standards was named as an especially valuable outcome.
Following the completion of the study, patients also have said that reducing the number of visits to the clinic is important, as is maintaining retinal heath. Patients pointed out that diabetes care is now better than before, and they expected to live longer. They therefore wanted a treatment that would not cause damage, preserving the health of their retina over the course of their life.
Study Summary | Dr. David Steel
DIAMONDS study was a government-sponsored, randomized double-masked noninferiority (and equivalence) clinical trial, carried out at 16 sites in the UK. Of the 266 patients recruited, 133 were randomized into the SML group where they received treatment with the Iridex IQ 577 laser and TxCell™ Pattern Scanning Delivery Device, and 133 were randomized into the continuous-wave group, where they were treated with 532-nm laser using the modified Early Treatment Diabetic Retinopathy Study (ETDRS) protocol.
MicroPulse® technology chops a continuous-wave laser beam into short bursts, allowing the tissue to cool between each application.

A treatment mode in the Iridex IQ 577 laser.
Investigators used standardized protocols, and a contact lens with laser magnification of 1.0x ± 0.06x was recommended for all treatments. For SML, laser was applied confluently to the macular area, 500 µm from the foveal center, using three 7x7 spot grids, with zero-spot spacing above and below the fovea, and one 7x7 spot grid, with zero-spot spacing at each side of the fovea (temporal and nasal). Treatment was also applied to other areas of thickening, if present.
Laser titration using continuous-wave mode was performed in an area of edematous retina away from the fovea before administering SML treatment. The power at which a barely visible reaction was seen in continuous-wave mode was multiplied by four and used with a 5% MicroPulse duty cycle, 200-µm spot size, and 200-ms exposure duration for treatment.
SML Protocols
- Laser was applied confluently to the macular area.
- 500 µm from the foveal center with three 7x7 spot grids with zero-spot spacing above and below the fovea.
- One 7x7 spot grid with zero-spot spacing at each side of the fovea (temporal and nasal).
- Treatment was also applied to other areas of thickening, if present.
- The power at which a barely visible reaction was seen in continuous-wave mode was multiplied by four and used with a 5% MicroPulse duty cycle.
- 200-µm spot size and 200-ms exposure duration for treatment.
For patients in the CWL arm of the trial, treatment was applied to areas of thickened retina, macular nonperfusion (away and noncontiguous with the perifoveal capillaries), and leaking microaneurysms, in accordance with the ETDRS and the guidelines of the United Kingdom Royal College of Ophthalmologists.
Participants were followed for 2 years. If necessary, laser retreatments were carried out with the same technology allocated by randomization.
“THE ABSENCE OF A SCAR OR BURN FOLLOWING SML LED TO SOME HEALTHCARE PROFESSIONALS TO DOUBT THE EFFECTIVENESS OF SML COMPARED TO CWL.”
Outcome Summary | Dr. David Steel
SML had equivalent efficacy and cost to CWL suggesting that either treatment could be offered to patients with central involved DME of <400 µm suitable for macular laser treatment. Both laser treatments had equivalent efficacy in clearing the fluid from the retina and maintaining vision for at least two years, and both were cost-effective.
No statistically significant difference was found in the percentage of patients meeting driving standards. The same was true of mean change in binocular best corrected visual acuity, central retinal thickness (CRT), Humphrey visual field mean deviation, side effects, and rescue treatments.
In my opinion, when patients are armed with knowledge of this equivalency and an understanding of the superior risk profile of SML, they will likely opt for SML, which does not burn the retina.
Benefits of MicroPulse® Technology | Dr. Noemi Lois
The use of MicroPulse® technology in retinal treatments has been shown in other studies to preserve photoreceptor cells,2-4 retinal pigment epithelium (RPE) and neurosensory retina.2,3 Even treating the fovea has been shown to be possible with studies showing no evidence of damage,2 which makes the procedure easy and safe to perform. Histopathology studies have also shown lack of laser burns following SML when applied using 5% duty cycle.5
SML should therefore be easy to teach to junior ophthalmologists and general ophthalmologists. When treating patients with edema, fluid can make it difficult to locate the center of the macula and the fovea may not be clearly recognizable; however, use of SML is appropriate in these cases, because studies have shown use of SML resulted in no evidence of damage to the fovea.2 Similarly, patient movement and compliance during treatment also is not as critical with SML given the limited risk of fovea damage.
With the number of adults with diabetes and clinically significant macular edema projected to rise from an estimated 18.83 million globally in 2020 to 28.61 million by 2045,6 ease of training is an important consideration as the need for care increases.
MicroPulse® technology has been shown to preserve photoreceptor cells, RPE and neurosensory retina.
Case Study | Dr. Noemi Lois
A 65-year-old female with DME was randomized to SML. Her baseline best corrected visual acuity was 50 ETDRS letters, and her CRT was 375 µm. At 24 months, the edema was resolved, and the patient’s CRT was reduced to 258 µm. Her visual acuity had improved to 73 ETDRS letters. Optical coherence tomography (OCT) shows preservation of all retinal layers following treatment.
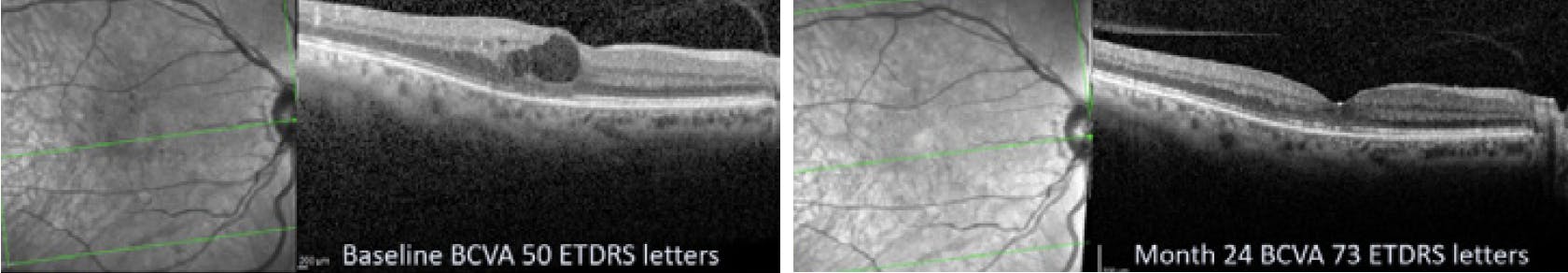
Patient with center involving DME with <400 μm CRT on OCT. Left, prior to treatment (baseline), and right, 24 months following SML.
“SUBTHRESHOLD MICROPULSE LASER IS CLEARLY THE FUTURE. IT IS AS EFFECTIVE AS STANDARD LASER AND DOES NOT PRODUCE RETINA BURNS. IT’S A NO BRAINER!” -TOM RUSH, Person living with diabetes (PLWD)
Clinical Pearls
David Steel MBBS, FRCOphth, MD(Res)
- Be extremely attentive and methodical about applying the grid blocks that make up the pattern in your protocol. Since you do not see a visible reaction to the laser treatment, you will not be able to tell where you have lasered, and you must know you have covered the necessary area.
- Use a contact lens with laser magnification of one and as wide of a field as possible. This prevents the need to alter spot size to compensate for lens magnification and it allows you to see the whole area that you are treating, making it easy to maintain your bearings.
- Titrate the power. I recommend the titration protocol used in DIAMONDS, as well as the application protocol, as they have been clinically proven to be safe and effective.
- Do not be afraid of repeating SML treatment. I did not see any adverse events with retreatment, and we did have patients who responded on the second or third treatments.
Noemi Lois MD, PhD, FRCS(Ed), FRCOphth
- Focus the laser on the retinal pigment epithelium, below the retinal blood vessels. You want to see the retinal blood vessels running on top of your laser grid, so that you know you are focused below them.
- Treat early in the course of the disease. The ETDRS showed us that macular laser reduces the risk of visual loss (3 ETDRS lines or more) by 50% at 3 years in patients with clinically significant macular edema.7 We should be treating patients before their condition becomes center involving.
- If the edema has progressed and is center involving, we should treat in a timely fashion, while the CRT is <400 μm. This is when the laser treatment has been shown by studies such as DIAMONDS to work well. We should inform and offer treatment with laser to patients early, before they need to be embarked on costly, inconvenient, and more risky long-term intravitreal injections.
Continued Patient Care With MicroPulse® Technology
Patients treated with SML in the DIAMONDS study maintained good vision for two years, and there were no side effects during this time. SML requires fewer visits to the clinic than anti-VEGF injections, and it is not a painful procedure. Patients can be dilated with a short-acting drop and they are not uncomfortable after treatment. They can get right back to normal life.
SML poses no risk of infection, and the treatment can be repeated without damage to the retina. As diabetes patients live longer, a treatment that will not cause any damage becomes increasingly important.
SML is also cost-effective. This is an important factor for patients, insurance companies and national healthcare systems.
“I AM A TYPE 1 DIABETES PATIENT (41 YEARS SINCE DIAGNOSIS) AND I AM VERY GRATEFUL FOR THE BENEFITS OF THE NEW NON-INVASIVE LASER TREATMENT (SUBTHRESHOLD MICROPULSE LASER), WHICH LEAVES NO SCARRING. MY MACULAR EDEMA HAS SETTLED DOWN, AND I HAVE SINCE HAD NO FURTHER NEED OF ANTI-VEGF INJECTIONS. THE PROCEDURE ITSELF LASTS ABOUT 5-10 MINUTES AND IS NOT PAINFUL IN ANY WAY. SIGHT IS PRECIOUS, AND THIS IS A NEW AND EXCITING WAY OF PRESERVING IT.”
-W. ROBERT STEVENSON, Person living with diabetes (PLWD)
DIAMONDS showed SML treatment is a safe, effective, and low-cost procedure. When DME is clearly present (and I am not referring here to subclinical cysts as seen on OCT) and signs of progression have been noted, there is a risk to sight, and I believe patients should be offered treatment with SML there and then.
1. Lois N, Campbell C, Waugh N, et al. Diabetic Macular Edema and Diode Subthreshold Micropulse Laser: A Randomized Double-Masked Noninferiority Clinical Trial. Ophthalmology. 2023;130(1):14-27. doi:10.1016/j.ophtha.2022.08.012
2. Luttrull JK, Sinclair SH. Safety of transfoveal subthreshold diode micropulse laser for fovea-involving diabetic macular edema in eyes with good visual acuity. Retina. 2014;34(10): 2010e2020.
3. Vujosevic S, Martini F, Longhin E, et al. Subthreshold micropulse yellow laser versus subthreshold micropulse infrared laser in center-involving diabetic macular edema: morphologic and functional safety. Retina. 2015;35(8): 1594e1603.
4. Wells-Gray EM, Doble N, Ohr MP, Choi SS. Structural integrity of individual cone photoreceptors after short- wavelength subthreshold micropulse laser therapy for dia- betic macular edema. Ophthalmic Surg Lasers Imaging Retina. 2018;49(12):946e954.
5. Yu AK, Merrill KD, Truong SN, Forward KM, Morse LS, Telander DG. The comparative histologic effects of subthreshold 532- and 810-nm diode micropulse laser on the retina. Invest Ophthalmol Vis Sci. 2013;54(3):2216-2224. Published 2013 Mar 1. doi:10.1167/iovs.12-11382
6. Varma R, Bressler NM, Doan QV, et al. Prevalence of and risk factors for diabetic macular edema in the United States. JAMA Ophthalmol. 2014;132(11):1334e1340.
7. Photocoagulation for diabetic macular edema. Early Treatment Diabetic Retinopathy Study report number 1. Early Treatment Diabetic Retinopathy Study research group. Arch Ophthalmol. 1985;103(12):1796-1806.
Iridex, the Iridex logo, IQ 577, TxCell, and MicroPulse are registered trademarks of Iridex Corporation. All other trademarks are the property of their respective owners. U.S. Patents 7,771,417; 8,945,103
Iridex | 1212 Terra Bella Avenue | Mountain View, CA 94043 | 800.388.4747 (U.S. Inquiries) www.info@iridex.com U.S. & Int’l Inquiries) | www.iridex.com
* In the DIAMONDS clinical trial, continuous-wave laser therapy (CWL) is referred to as standard laser (SL).
LT0770


