In February 2023, the FDA approved pegcetacoplan (SYFOVRE®, Apellis Pharmaceuticals, Inc. Waltham, MA) for treatment of geographic atrophy (GA) secondary to age-related macular degeneration (AMD). Pegcetacoplan acts on the complement cascade by targeting and inhibiting C3 and C3b (Figure 1). Overactivation of the complement system is implicated in the pathogenesis of geographic atrophy.1 C3 is the most abundant complement protein. It occupies a central position in the complement system, being the convergence point for all three pathways of complement activation (classical, lectin, and alternative pathways).2 Inhibiting C3 and C3b can effectively down-regulate the entire downstream cascade, preventing the generation of all downstream effectors such as C3a, C3b (and other C3 fragments), C5a, and the membrane attack complex (MAC).

Figure 1. The complement cascade, a major component of the immune system, is comprised of a network of proteins working together to promote inflammation and destroy pathogens. It operates via three pathways – classical, alternative, and lectin – eventually leading to the formation of a destructive complex (the membrane attack complex or MAC) that attacks a pathogen’s cell membrane. Dysregulation of the cascade may lead to photoreceptor (PR) and retinal pigment epithelium (RPE) dysregulation and has been implicated in GA. Pegcetacoplan acts on C3 and C3b, the critical point for all three pathways of complement activation.
Pegcetacoplan is effective in reducing the growth rate of GA secondary to AMD, as demonstrated in two large, 24-month phase 3 clinical trials, OAKS (ClinicalTrials.gov: NCT03525613) and DERBY (NCT03525600),3 alongside an ongoing 3-year extension study, GALE. The initial 12-month results from GALE encompass 3 years of continuous pegcetacoplan in eyes randomized to pegcetacoplan treatment groups. Slope analysis from these trials indicates increasing efficacy, culminating in a 42% reduction in the rate of GA progression with monthly treatment in the 3rd year in patients with nonsubfoveal GA lesions versus projected sham (Figure 2). The overall reduction in the rate of GA growth versus sham over 3 years for the total population was 25% in the pegcetacoplan monthly (PM) group and 20% for the pegcetacoplan every-other-month (PEOM) group. After 24 months pegcetacoplan treatment reduced growth of GA compared to sham by 26% and 23% in nonsubfoveal GA, in 18% and 16% in subfoveal GA eyes, and by 21% and 17%, across all eyes receiving PM and PEOM, respectively in OAKS and DERBY combined. There was no statistically significant difference between pegcetacoplan-treated arms and sham pooled in the endpoints on visual functional measures at 2 years, including BCVA, reading speed, functional reading independence index, and mean sensitivity for microperimetry.

Figure 2. Mean rate of change in the total area of GA lesions from baseline to month 36 in the pooled OAKS and DERBY studies and in the GALE extension study assuming a piecewise linear trend in time with a knot every 12 months. The dotted line designates projected sham, given the absence of a sham arm in the GALE study following the crossover of sham patients to pegcetacoplan. Projected sham was estimated from the mean rate of change in each period in months 0-24. Open-label studies can allow for selection bias. This is a prespecified analysis but there is no statistical testing hierarchy.
Natural history studies demonstrate an increased rate of GA growth in eyes with nonsubfoveal GA compared to subfoveal lesions,4 which may account for the greater efficacy of pegcetacoplan in nonsubfoveal-located GA. Nonsubfoveal GA was defined as ≥1 µm of the GA lesion border from the foveal center by an independent reading center (DARC) based on OCT and fundus autofluorescence (FAF) imaging. The lack of available treatments until recently means that retina specialists frequently encounter advanced, subfoveal GA cases in clinical practice. Pegcetacoplan offers a solution for a heterogenous group of GA lesion types because the inclusion criteria of the clinical trials were designed to reflect patients who may be seen in real-world clinical practice. At baseline in OAKS/DERBY, 624 (81%) of included eyes with subfoveal GA had BCVA better than 20/200 (the threshold for legal blindness). Thus, patients with subfoveal GA might benefit from treatment that slows lesion growth to delay permanent vision loss. This conclusion is supported by recent post-hoc analyses. The first demonstrated reduced risk in the development of absolute scotoma in the central 4- and 16-loci using microperimetry in patients treated with pegcetacoplan versus sham (OAKS). The other showed a reduction of 38% in monthly treated eyes and of 12% in those treated every other month versus sham in the risk of loss of vision to less than 35 letters (20/200) (OAKS and DERBY).
Additional support for pegcetacoplan’s efficacy has been shown by comparing GA lesion growth between treated eyes and fellow eyes with GA. Natural history studies have shown high correlation in the GA progression rates in two eyes of the same patient,5 supporting a comparison with the fellow eye as an internal control. Over 3 years, overall GA growth was reduced by 21% in the PM arm and 19% in the PEOM arm compared to the fellow eyes meeting study inclusion criteria, with an increasing effect up to 28% in the third year in patients receiving monthly pegcetacoplan injections.
The increasing effect of pegcetacoplan over time noted in clinical trials is hypothesized to manifest after the damaged photoreceptors and RPE cells, which are committed to progress toward cell death in untreated GA eyes, have cleared. The full neuroprotective benefits of complement inhibition may only become evident once these compromised cells die off, allowing cells earlier in the disease process to be effectively shielded from irreversible damage.
An estimated 250000 pegcetacoplan injections have been administered across clinical trials and in the real world as of the latest data cut on March 31, 2024 (See Pegcetacoplan: Favorable Benefit-Risk Profile). Events of retinal vasculitis and/or retinal vascular occlusion have been reported in the real-world setting. Cases may occur with the first dose of pegcetacoplan and may result in severe vision loss. Since the initial reports, the estimated rate of vasculitis after pegcetacoplan has remained stable at approximately 1:10000 injections (or 0.01% per injection) overall. To date, cases of vasculitis appear to be a first-injection phenomenon and the rate of first injection is estimated at 1:4000 (or 0.025%). Based on an initial investigation with samples, it is hypothesized that the risk is linked to pre-existing immunity against polyethylene glycol. Apellis is committed to sharing data with the retina community and to patient care. Please contact pharmacovigilance at Apellis if you experience an adverse event, and they will also report your case to the FDA and regulatory bodies as mandated. Discontinue treatment with pegcetacoplan in patients who have signs and symptoms of vasculitis.
Panel section: The following section includes views and opinions of retina physicians. The information is being provided for educational purposes only and is not intended to provide medical advice or replace and/or substitute the medical treatment and care provided by a patient’s health care provider. When treating retinal vasculitis, physicians should use their independent medical judgement based on the patient’s individual clinical history and symptoms.
Which patients do you consider treating with SYFOVRE?
Eleonora Lad, MD, PhD: I offer pegcetacoplan to all patients with GA, except those with a history of uveitis or ocular inflammation or active infections. My ideal eye has an uninvolved foveal center, and my goal in providing treatment to slow lesion progression is to preserve uninvolved retinal tissue for as long as possible. If the patient has a history of neovascular AMD, I still offer treatment, but I discuss the risk of reactivation and the potential need for two injections—the complement inhibitor and anti-VEGF therapy. I think monocular patients are especially motivated to receive treatment. Also, eyes with fast-progressing lesions (multifocal or diffuse GA, hyperautofluorescence patterns associated with active GA growth) should be preferentially considered.
Marco A. Zarbin, MD, PhD, FACS: Several factors influence my decision to offer pegcetacoplan treatment to patients with GA, including the distance of the GA to the fovea, the patients’ visual acuity in the index and fellow eye, and the presence or absence of choroidal neovascularization (CNV). My ideal patient for treatment is someone with confirmed GA secondary to AMD, whose BCVA is better than 20/100 but worse than 20/30 due to GA and who has no CNV on exam or OCT. A history of uveitis is a relative contraindication. The literature describes several risk factors for rapid GA growth, but to me, relying on the actual documented progression rate in a given patient when discussing the risk for disease progression is less speculative.
John W. Kitchens, MD: I have been surprised at how many patients want pegcetacoplan treatment. In my view, those who have the most to gain from this therapy include patients with nonsubfoveal GA in one eye and subfoveal GA in the fellow eye, and those with multifocal, small eccentric GA. Conversely, in individuals with center-involved GA and established poor vision, pegcetacoplan may be an option in selected cases.
Roger A. Goldberg, MD, MBA: After excluding conditions that mimic GA, the decision to offer pegcetacoplan treatment is based on patient and clinical factors including symptoms, appearance and growth of GA over time, and the status of the fellow eye. Characteristics such as multifocal GA and nonsubfoveal GA location are associated with more rapid growth of GA. Patient factors include age, mental status, and overall health to understand their visual needs and how long treatment will be viable. As the data demonstrates an increasing benefit of pegcetacoplan the longer a patient is on therapy, I want to treat someone who will recognize that benefit. Clinical factors include visual acuity in the index and fellow eye; the GA status in the fellow eye; whether there is wet AMD in either eye; and whether the patient is already receiving injections. Lastly, a history of uveitis or prior retinovascular disease may be a relative contraindication. There are multiple factors to consider consistent with our clinical practice as we translate the science into the art of medicine and apply large data to the specific patient sitting in front of us. That said, if a patient has GA of any size, whether I feel like they would be a good candidate for therapy or not, I always at least inform them that (1) they have GA, and (2) there is therapy now available for GA. Then, we dive into the details of their specific situation.
How do you discuss SYFOVRE with your patients?
Dr. Goldberg: I inform patients that pegcetacoplan has been shown to slow the growth of GA in two large clinical trials. How well this medicine works depends on the duration of therapy: it works better at slowing GA in the third year of therapy than in the first year. I always tell patients that pegcetacoplan cannot reverse already established vision loss that is permanent, but it slows the future progression of GA. I explain that it delays lesion progression, which protects healthy retinal tissue for longer. As for safety, pegcetacoplan treatment involves inserting a needle in the eye, which carries a risk of infection and inflammation, including inflammation of the optic nerve or the blood vessels of the retina. The occurrences of retinal vasculitis appear to be happening primarily after the first injection, with a rate of about 1:4000 for the first injection. Due to this risk, I recommend treating the worst-seeing eye first and waiting an interval of 4 to 6 weeks before treating the contralateral eye. I also discuss the risk of developing wet AMD. Finally, I describe my preference for a 6- to 8-week treatment interval, due to a more favorable risk-benefit ratio with a more tolerable regimen.
Dr. Zarbin: Likewise, I emphasize that pegcetacoplan treatment slows the rate of lesion progression and does not restore vision that has already been lost. In the clinical trials across all GA subtypes in AMD, pegcetacoplan reduced the rate of growth versus sham by 21% in the monthly arm and by 17% in the every-other-month arm at 2 years, and the benefit seems to increase with continued pegcetacoplan injections. Regarding safety, I explain that anti-complement treatments like Syfovre increase the risk of CNV (after 24 months ~12% in monthly treatment, 7% every other month, and 3% in controls), and that if this develops, a separate injection will be required. Lastly, I tell patients there is less than 1% risk of catastrophic complications such as endophthalmitis or retinal vascular occlusion, that could necessitate additional surgery and/or cause complete and permanent loss of vision in the eye.
Has treatment with SYFOVRE demonstrated a functional benefit in previous analyses?
Dr. Lad: It is notable that over 60% of eyes in the OAKS and DERBY trials had subfoveal GA (Figure 3), and many of those eyes had characteristics associated with fast GA progression. Due to the advanced GA stage in many eyes, and the known fact that BCVA is not reflective of GA lesion growth, no difference in BCVA was seen at 24 months between the treated and sham arms. Alternative endpoints have been considered and even accepted by the FDA for GA trials such as the rate of GA growth on autofluorescence.
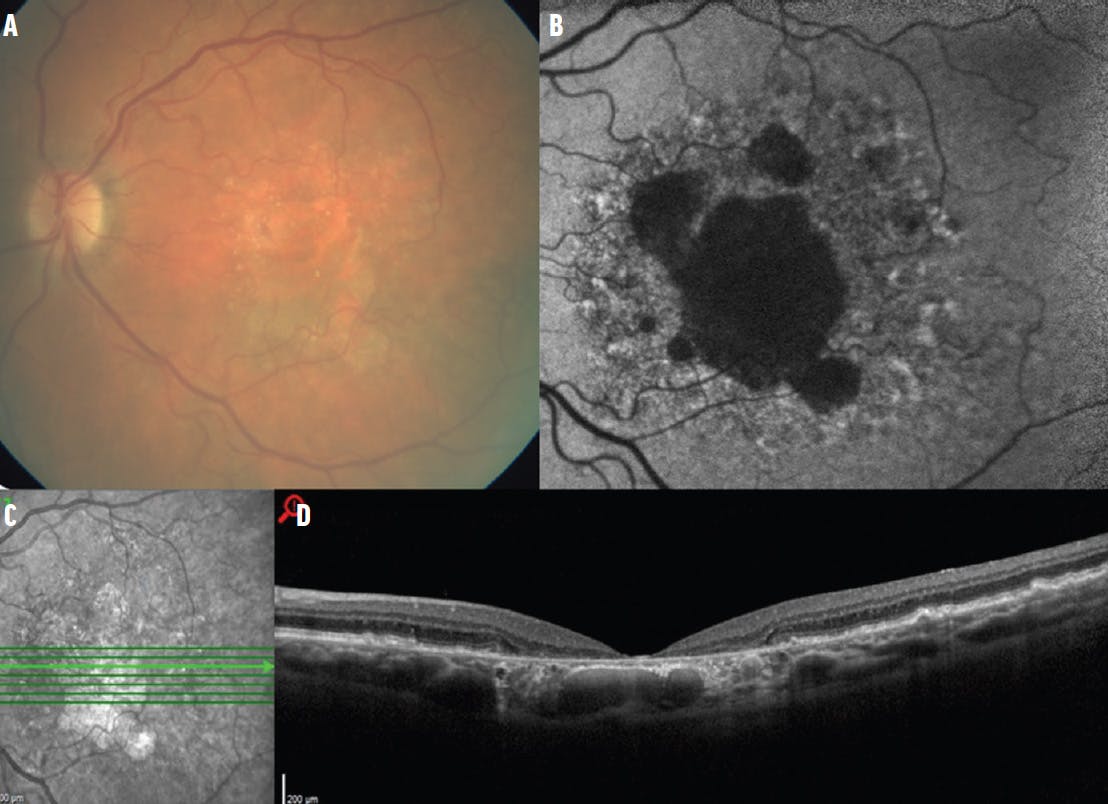
Figure 3. A majority of patients in the OAKS and DERBY trials had subfoveal GA. The average lesion size at baseline was approximately 8.2 mm2 as seen in this case of a patient with a similar lesion size (8.3 mm2). A – colour fundus photograph. B – Fundus autofluorescence. C – Infrared image. D - OCT scan showing central CRORA (Complete RPE and Outer Retinal Atrophy).
There are compelling data from multiple post hoc analyses supporting the efficacy of pegcetacoplan including delayed time to severe visual impairment in pegcetacoplan treated eyes (Figure 4), and preservation of vision with pegcetacoplan treatment in eyes with extrafoveal GA. One of the most robust pieces of data was microperimetry, demonstrating increased preservation of retinal light sensitivity in the junctional zone (the area adjacent to the GA lesion border) and in the central macula in pegcetacoplan-treated eyes. The latter analysis showed that eyes with preservation of central macular sensitivity had better vision than those with a loss of this sensitivity.

Figure 4. A time-to-event analysis was conducted for the persistent drop of visual acuity in treated or sham eyes to less than 35 letters (equivalent to 20/200). There was a delay in those events compared with sham in eyes treated with pegcetacoplan monthly (risk reduction of 38% over 2 years vs sham) and in those treated with pegcetacoplan every other month (risk reduction of 12% vs sham).
Dr. Goldberg: Much of what we have learned about functional microperimetry endpoints comes from the CHROMA and SPECTRI trials, which demonstrated that perilesional or junctional zone microperimetry is a functional correlate of GA growth. The post-hoc analysis of OAKS used this measure to show a trend of preservation of light sensitivity in pegcetacoplan-treated eyes. Combined with the FAF primary endpoint, which evaluated the RPE, the totality of data demonstrates the benefit of treatment with pegcetacoplan in preserving outer retinal tissue, and with it, retinal sensitivity to light. Intuitively, this structure-function correlation makes sense.
Dr. Kitchens: BCVA is a limited endpoint in GA, because progression and growth of GA lesion size often do not correlate with vision loss. Vision is also noisy and variable in these elderly patients with GA in the macula and we see that in the office. This makes vision a less reliable measure for GA studies. Because the treatments we have for GA are intended to slow its progression, their studies were designed with anatomic outcomes as the primary endpoint. I think we are so accustomed to anti-VEGF therapy improving visual acuity for our patients with neovascular AMD, but that is a completely different paradigm, and so we have to recalibrate our expectations for the outcome in patients with GA. A post-hoc analysis of OAKS and DERBY showed that eyes in which the GA was more than 250 µm from the foveal center lost fewer letters than sham-treated eyes over the course of treatment with pegcetacoplan.
When do you start your patients on SYFOVRE?
Dr. Goldberg: There is a sweet spot to the timing when pegcetacoplan treatment is started. A study by Chakravarthy et al showed that 2/3 of patients with GA became ineligible to drive within a median of 1.6 years.6 We can’t wait until all the vision is gone to start treating, but whenever we start, we are obligating patients to repeated injections over time. The eyes that are going to benefit the most from treatment are those where the GA is likely to threaten vision. If I find a small lesion far away from the fovea, I might be inclined to closely observe for progression before offering treatment.
Dr. Kitchens: Every patient is an individual and has an individual decision to make. For patients newly diagnosed with GA, I generally do not want to inject pegcetacoplan at the first visit. For our existing patients, if we have the data, I like to show them the GA and its progression, but I still send them home to think about the pros and cons of treatment. Most of the time, we must do a benefits investigation anyway, so that pause gives us time to figure out the insurance coverage options. For patients who either remain undecided or forgo treatment, I request they return in 3 to 6 months to reassess the atrophy.
How do you decide between monthly and every-other-month treatment?
Dr. Lad: The 24-month efficacy data for both treatment frequencies are similar although favoring monthly treatment. Typically, I preferentially offer every-other-month treatment to decrease the injection burden. However, some patients may benefit from a monthly treatment—for example, those with a high risk of lesion progression, monocular patients, and those with a strong family history of vision loss from AMD.
Dr. Goldberg: There is always a balance between efficacy and injection risk. While the efficacy was similar between both treatment intervals at 24 months, there was a more pronounced benefit for monthly pegcetacoplan treatments in the GALE extension study, with up to 42% reduction in nonsubfoveal GA growth between months 24 and 36. However, the effect was still meaningful in-every-other month dosing in this subpopulation in GALE with up to 28% reduced GA growth, and safety outcomes favor this regimen. Fortunately, the pegcetacoplan US approval label offers flexibility with dosing every 25 to 60 days and I recommend 6- to 8-week treatment intervals for patient convenience, as well as for compliance.
Dr. Zarbin: I use an interval of 6 to 8 weeks between injections. An 8-week interval has been shown in the clinical trials to be associated with a lower risk of CNV than monthly dosing and a comparable chance of benefit. I might opt for monthly treatments in some situations; for example, if the fovea is threatened acutely in the better-seeing eye (ie, because the patient presented to me late in the course of their disease). In that scenario, I would probably opt for monthly injections initially, then after 1 year, I might consider decreasing the frequency to every 6 to 8 weeks.
What are the best techniques to prepare a SYFOVRE injection?
Dr. Goldberg: Pegcetacoplan should be at room temperature for at least 15 minutes before administration according to the USPI. This is because it is viscous when cold, making it difficult to draw up. For preparation, I always use the injection kit supplied by Apellis that includes an 18-gauge filter needle for withdrawing the medication from the vial and a 29-gauge, thin-walled needle for the intravitreal injection. I maintain the vial upright and tilted slightly, as I place the filter needle into the vial. I keep the bevel of the filter needle completely submerged in the liquid as I draw up the medicine. It is not necessary to pull the plunger all the way, as that will not help draw the medicine up any faster. Instead, I pull back to about 0.3 or 0.4 mL and target a volume of 0.2 mL, which will leave me with a roughly 0.1 mL air bubble between the plunger of the syringe and the medicine. I do not flick the syringe; instead, with one back-and-forth motion, I draw the medicine and that air bubble down, along the sides of the syringe, and then I bring the plunger up relatively quickly to catch the medicine with the plunger base. That brings the medicine down onto the plunger, and the air bubble goes to the hub of the needle. The other thing to mention is that the actual injection of the medicine should be performed slowly; it takes a little bit longer than an anti-VEGF injection because it is a higher volume (0.1 mL vs 0.05 mL) and more viscous. For full preparation instructions, you should consult the SYFOVRE prescribing information.
How can clinicians assess lesion growth in the clinic?
Dr. Kitchens: In the phase 3 trials, fundus autofluorescence (FAF) was the primary endpoint to measure GA lesion growth monitored by an independent reading center. While this is available for use in the clinic, it is tricky to compare serial FAF images over time. Some of the OCT devices have built-in software modules to assess change in GA area. I use the RPE analysis function, which allows me to follow GA lesion changes over time, as well as to educate patients about the disease.
Dr. Goldberg: I obtain an OCT at every visit, consistent with my practice for AMD as well as to evaluate for CNV. GA growth can be assessed via hypertransmission on the en face OCT. I also perform FAF every 6 months to have a multimodal imaging assessment of GA. Different OCT devices can show an en face view of the macula, which can highlight GA, whether as an OCT reflectance image or as an infrared image. The review software will often then let you draw around the areas of GA to facilitate size measurements. Multiple companies are developing artificial intelligence-based GA analysis algorithms as add-ons to OCT devices.
What is the risk of developing vasculitis with SYFOVRE, and why does this risk exist?
Retinal vasculitis and/or retinal vascular occlusion, typically in the presence of intraocular inflammation, have been reported with the use of SYFOVRE. Cases may occur with the first dose of SYFOVRE and may result in severe vision loss. Discontinue treatment with SYFOVRE in patients who develop these events. Patients should be instructed to report any change in vision without delay.
Dr. Lad: Based on the latest data, there have been 250000 injections administered, with 25000 doses administered so far in the clinical program. These events are estimated at 1 in 10000 (or 0.01%) per injection. Apellis is collecting information and forming hypotheses on the cause of these events, with the most promising theory under investigation being a type III hypersensitivity reaction to PEG in the medication.
Dr. Goldberg: The confirmed cases identified to date have occurred after the first injection, so the estimated rate per first injection is probably to 1 in 4000. Early detection is crucial, which is why I see patients back 2 to 3 weeks after the first injection to look for any signs of inflammation.
Dr. Zarbin: I agree, these events could be a hypersensitivity reaction to the polyethylene glycol in the medication. There is histology pending on an enucleated eye, which should tell us more. If hypersensitivity is the cause, I wonder if skin testing before injection might be useful.
What should clinicians do if they suspect a case of vasculitis?
Dr. Lad: After intravitreal injections, patients may complain of irritation for 24 to 48 hours. I always inform my patients to report to the office if there are symptoms over 48 hours after an injection. In retinal vasculitis after pegcetacoplan, the onset of symptoms was 6 to 14 days following injection. Symptoms included blurred or reduced vision, or eye pain. Patients should be brought in immediately for evaluation, which includes a slit lamp examination, OCT, fundus photography, and fluorescein angiography (FA), either wide-field or standard FA with peripheral sweeps. Signs vary between patients but may include conjunctival injection; mild corneal edema and elevated IOP; anterior chamber and/or vitreous inflammation, which may be mild; retinal hemorrhage; and signs of nonperfusion. Consider an infectious etiology, especially if hypopyon and fibrin are present in the anterior chamber.
After an infectious cause has been definitively excluded, rigorous corticosteroid treatment should commence and includes ocular steroids (intravitreal or subtenon and topical) and systemic steroids. Steroid treatment should be individualized based on the individual case. Elevated IOP should be treated with IOP-lowering drops. Patients should be monitored closely while inflammation is still active. Panretinal photocoagulation may be a consideration if areas of nonperfusion or neovascular glaucoma are present.
Dr. Zarbin: Endophthalmitis should be excluded as a first step, but the two conditions should be distinguished. Signs of endophthalmitis may include but are not limited to the following: conjunctival injection, inflammatory cells in the anterior chamber and the vitreous, hypopyon, and retinitis/vasculitis may or may not be present. Non-infectious vasculitis, on the other hand, may or may not have vitreous cells and hypopyon is absent. Elevated IOP with corneal edema may be a feature. An FA should reveal vasculitis although one may be able to detect it on fundus exam alone. I try to begin treatment of vasculitis immediately upon diagnosis. Generally, I would consider including systemic steroids and/or a dexamethasone implant, unless contraindicated. The best way to follow patients after the first dose of pegcetacoplan is still not clear. I currently do not call patients back 1 week after an initial injection to monitor for vasculitis, but I do ask them to contact me immediately if they detect any floaters or changes in peripheral or central vision, and I provide them with my direct contact information.
The authors would like to acknowledge Apellis Pharmaceuticals for providing medical writing support.
1. Boyer DS, Schmidt-Erfurth U, van Lookeren Campagne M, et al. The pathophysiology of geographic atrophy secondary to age-related macular degeneration and the complement pathway as a therapeutic target. Retina. 2017; 37:819-835.
2. Merle NS, Church SE, Fremeaux-Bacchi V, Roumenina LT. Complement system part I—molecular mechanisms of activation and regulation. Front Immunol. 2015;6:262.
3. Heier JS, Lad EM, Holz FG, et al; OAKS and DERBY study investigators. Pegcetacoplan for the treatment of geographic atrophy secondary to age-related macular degeneration (OAKS and DERBY): two multicentre, randomised, double-masked, sham-controlled, phase 3 trials. Lancet. 2023;402(10411):1434-1448.
4. Schmitz-Valckenberg S, Sahel JA, Danis R, et al. Natural history of geographic atrophy progression secondary to age-related macular degeneration (Geographic Atrophy Progression Study). Ophthalmology. 2016;123(2):361-368.
5. Sunness JS, Margalit E, Srikumaran D, et al. The long-term natural history of geographic atrophy from age-related macular degeneration: enlargement of atrophy and implications for interventional clinical trials. Ophthalmology. 2007;114(2):271-277.
6. Chakravarthy U, Bailey CC, Johnston RL, et al. Characterizing disease burden and progression of geographic atrophy secondary to age-related macular degeneration. Ophthalmology. 2018;125(6):842-849.
SYFOVRE® (pegcetacoplan injection) Important Safety Information
INDICATION
SYFOVRE is indicated for the treatment of geographic atrophy (GA) secondary to age-related macular degeneration (AMD)
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
SYFOVRE is contraindicated in patients with ocular or periocular infections, and in patients with active intraocular inflammation
WARNINGS AND PRECAUTIONS
• Endophthalmitis and Retinal Detachments
– Intravitreal injections, including those with SYFOVRE, may be associated with endophthalmitis and retinal detachments. Proper aseptic injection technique must always be used when administering SYFOVRE to minimize the risk of endophthalmitis. Patients should be instructed to report any symptoms suggestive of endophthalmitis or retinal detachment without delay and should be managed appropriately.
• Retinal Vasculitis and/or Retinal Vascular Occlusion
– Retinal vasculitis and/or retinal vascular occlusion, typically in the presence of intraocular inflammation, have been reported with the use of SYFOVRE. Cases may occur with the first dose of SYFOVRE and may result in severe vision loss. Discontinue treatment with SYFOVRE in patients who develop these events. Patients should be instructed to report any change in vision without delay.
• Neovascular AMD
– In clinical trials, use of SYFOVRE was associated with increased rates of neovascular (wet) AMD or choroidal neovascularization (12% when administered monthly, 7% when administered every other month and 3% in the control group) by Month 24. Patients receiving SYFOVRE should be monitored for signs of neovascular AMD. In case anti-Vascular Endothelial Growth Factor (anti-VEGF) is required, it should be given separately from SYFOVRE administration.
• Intraocular Inflammation
– In clinical trials, use of SYFOVRE was associated with episodes of intraocular inflammation including: vitritis, vitreal cells, iridocyclitis, uveitis, anterior chamber cells, iritis, and anterior chamber flare. After inflammation resolves, patients may resume treatment with SYFOVRE.
• Increased Intraocular Pressure
– Acute increase in IOP may occur within minutes of any intravitreal injection, including with SYFOVRE. Perfusion of the optic nerve head should be monitored following the injection and managed as needed.
ADVERSE REACTIONS
• Most common adverse reactions (incidence ≥5%) are ocular discomfort, neovascular age-related macular degeneration, vitreous floaters, conjunctival hemorrhage.
Please see full Prescribing Information for more information.
©2024 Apellis Pharmaceuticals, Inc. All rights reserved. 05/24 MED-ALL-PEGGA-24-00020 v1.0





