We are fortunate to work in an era where many diseases we treat have excellent therapeutic options. This progress has shifted our goal from merely preserving vision with minimal collateral damage to improving anatomy and visual function. Moreover, newer molecules have enabled us to extend the intervals between treatments so that we can better meet the needs of our patients and our clinics. As the global population ages and diabetes rates rise,1 we are seeing a growing number of patients with neovascular (wet) age-related macular degeneration (AMD), diabetic macular edema (DME), and retinal vein occlusion (RVO), which are leading causes of vision loss among adults.
A pivotal aspect of treating this growing population, along with restoring and maintaining vision, is the resolution of intraretinal and subretinal fluid, which are critical indicators of disease activity and progression. In essence, our clinical decisions, including treatment selection and the determination of treatment intervals, are guided by the drying of the retina. Achieving a dry retina is associated with vascular stability, eliminating inflammation and reducing harmful excess fluid.2 This disease stability, in turn, may contribute to vision preservation. By drying excess retinal fluid as swiftly, durably, and effectively as possible, we optimize visual outcomes and minimize treatment burden, particularly the need for frequent clinic visits.
When discussing treatment options with our patients, we sometimes use the analogy of a flooding library. The quicker we can extract the water and dry the room, the more long-term damage we hope to avert to maintain the integrity of the books. From our clinical experience with wet AMD and DME, we have found that resolving retinal fluid and achieving disease stability as soon as possible post-diagnosis translate into long-term treatment success by forestalling irreversible damage.
Novel MOA in the Treatment Landscape for Retinal Diseases
Until recently, retina specialists had only anti-vascular endothelial growth factor (VEGF) and a handful of other treatments for retinal conditions such as wet AMD and DME. However, the landscape has changed, with different options becoming available in the last several years.
Vabysmo® (faricimab-svoa, Genentech) introduced an innovative strategy for managing wet AMD, DME, and RVO. Vabysmo is a first-in-class, bispecific, that inhibits both VEGF and angiopoietin-2 (Ang-2). In retinal vascular health, Ang-1 and Ang-2 play opposing roles in regulating stability: Ang-1 acts as a stabilizing force that maintains vascular homeostasis and prevents abnormal growth or inflammation, while Ang-2 acts as a destabilizing force.3 In retinal disease, Ang-2 displaces Ang-1, triggering vascular instability, inflammation, and the sensitization of VEGF.4 Though the contribution of Ang-2 inhibition on treatment effect and response has not been established, targeting Ang-2 alongside VEGF-A aims to restore this balance and is a novel approach to managing retinal vascular diseases like wet AMD and DME.5-7
Evidence from Vabysmo Trials for Wet AMD and DME
Evidence from the Vabysmo pivotal trials, including TENAYA and LUCERNE in wet AMD and YOSEMITE and RHINE in DME, consistently demonstrated that this treatment yields rapid and durable retinal drying when employed as a first-line therapy. Moreover, patients showed anatomical improvements and, crucially, sustained vision gains. Of note, the pivotal trials in wet AMD and DME met their primary endpoint showing Vabysmo was non-inferior versus aflibercept 2 mg given every 8 weeks in the mean change from baseline best corrected visual acuity (BCVA) at year 1 (average of weeks 40, 44, and 48*). Drying was also studied and rapid and sustained reductions in central subfield thickness (CST) were observed across all treatment arms.
Our experience and clinical decision-making build on results from the trials; fluid resolution and subsequent disease stability is a large deciding factor in determining treatment intervals. When we see patients, we are certainly guided by visual function, visual acuity and symptoms, but the presence or absence of macular fluid more commonly and directly impacts our clinical decisions. The drying effect we have observed in our clinics following treatment with Vabysmo is impressive, with durability to match, thus allowing us the possibility to adopt treat and extend regimens after loading doses,† which patients often appreciate.
Case Studies of First Line Use in Wet AMD and DME
Case 1: A 79-Year-Old Woman With Wet AMD
This first case study involves a 79-year-old treatment-naïve patient with a long history of bilateral wet AMD. The patient’s BCVA was 20/200, with a CST of 731 µm, when she received her first Vabysmo treatment. This patient experienced rapid improvement in vision and retinal fluid 1 month after her first Vabysmo injection during the loading dose period (Figure 1). This improvement was maintained over the course of 4 months and the patient’s treatment interval continues to be extended.
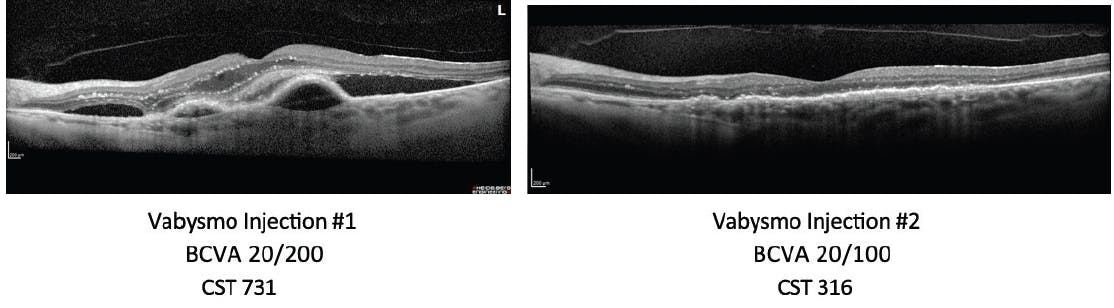
Figure 1. OCT scan at 4 weeks post-initial Vabysmo injection shows rapid retinal fluid resolution in a treatment-naïve wet AMD patient.
Case 2: A 48-Year-Old Treatment-Naïve Male Patient With DME
A treatment-naïve patient with proliferative diabetic retinopathy and DME was experiencing blurry vision and presented with neovascularization, vitreous hemorrhage and extrafoveal edema. The patient was started on Vabysmo treatment and, after four initial loading doses, extended out to 4-month dosing intervals. The patient’s vision improved from 20/30 to 20/20 after eight Vabysmo doses and his CST reduced from 456 µm to 290 µm (Figure 2).
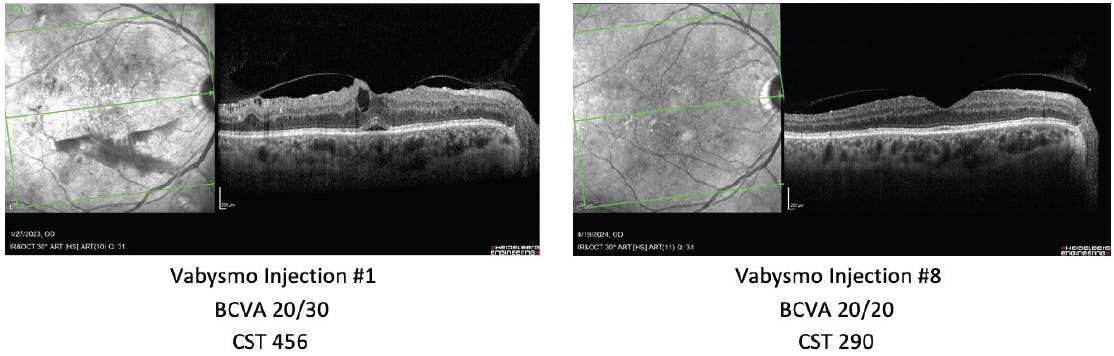
Figure 2. Initial OCT of treatment-naive DME patient revealed advanced neovascularization, vitreous hemorrhage, and extrafoveal edema. Vabysmo treatment, extended out every 4 months, improved vision and reduced retinal fluid.
With more than 2,000 patients enrolled in the clinical trials and additional real-world experience, there is also a strong foundation of safety data supporting Vabysmo as a first-line therapy. Moreover, these studies indicate that Vabysmo achieves these vision and drying results with extended intervals between treatments for some patients. The real-world evidence as seen in our clinical practice points to Vabysmo’s durability and drying capabilities.
Safety Data from Clinical trials
The clinical trials demonstrated that Vabysmo has a comparable safety profile to aflibercept. It is important to note that with any intravitreal injection that inhibits VEGF, there is a risk of endophthalmitis, retinal detachments, and a temporary increase in intraocular pressure, as well as arterial thromboembolic events and retinal vasculitis with or without occlusion, typically in the presence of intraocular inflammation – which have been reported with the use of Vabysmo in the postmarketing setting. The most common adverse reactions reported in patients receiving Vabysmo were cataract (15%) and conjunctival hemorrhage (8%).8
The Possibility of Fewer Visits for Patients
Traditionally, patients with wet AMD and DME required ocular injections every 1 to 2 months. However, with Vabysmo, patients may be able to go 1 to 4 months between treatments after four monthly loading doses. As clinicians, we understand that our elderly wet AMD and working-age diabetic patients may find themselves experiencing frequent medical appointments with a comprehensive team of health care professionals, including cardiologists, dentists, podiatrists, nephrologists, endocrinologists, and more. For patients with diabetes, life sometimes gets in the way. They are in search of retinal solutions that can provide durable stability for their conditions with the chance of fewer injections. The introduction of Vabysmo as a first-line treatment is a significant advance in achieving these objectives.
Our experience in practice closely aligns with the trials and real-world data. Ultimately, we want to stabilize our patients as quickly as possible and keep them stable while allowing them an opportunity for fewer visits and injections. This durability is important for patients with DME in particular, given it may be harder to get them into the clinic frequently.
One advantage of Vabysmo is its flexible dosing options, facilitating treatment intervals tailored to each patient’s unique needs driven by OCT and visual acuity, as opposed to more regimented schedules that may be inadequate for individual patients’ needs. The recommended dosing with Vabysmo in wet AMD and DME is 6 mg given in 4 or 6 (DME only) monthly loading doses followed by treatments every 1 to 4 months. Given that Vabysmo can be administered in monthly dosing regimens, or intervals as lengthy as 4 months, it offers a versatile treatment solution that is adaptable to a wide range of real-world circumstances.
Merging Safety and Efficacy With Simplicity and Access
As retinal specialists, safety and efficacy are paramount in our treatment decisions.
Fortunately, beyond the clinical trials, our experience in our practices is that anti-VEGF treatments, including dual inhibition Vabysmo, have well-established safety data to guide clinical management.
We also value simplicity. The pre-filled syringe for Vabysmo marks a significant step forward in improving the treatment of wet AMD, DME, and RVO. This streamlined administration process, combined with the flexibility of dosing schedules, has provided a better overall experience for us as clinicians and our patients.
Another reality and key consideration in our treatment decisions is insurance coverage. Vabysmo’s broad coverage makes it accessible to a significant portion of patients with wet AMD, DME, and RVO. Eligibility largely depends on insurance coverage, but thankfully, nearly 90%9 of patients in the United States are covered to start Vabysmo either as a first-line treatment or immediately following step therapy.‡ Specific coverage may vary for individuals and plans, but for those patients who are not covered, there are programs in place that may help patients get access to Vabysmo.
A Valuable First-Line Option for Patients and Specialists
Vabysmo represents the first dual-pathway therapy specifically designed to target both Ang-2 and VEGF-A, attacking these retinal diseases from multiple fronts. It is thought to restore vascular stability, reduce inflammation, and control leakage in the eye. Clinical trials provide compelling evidence that Vabysmo leads to rapid and robust drying of retinal fluid, enabling retinal specialists the opportunity to prolong intervals between injections, with safety data available across six large clinical trials.
As physicians, our ultimate goal is to provide our patients with a comparative treatment, ideally with as few clinic visits as necessary. Given its efficacy, durability, safety, and flexible dosing along with the introduction of a prefilled syringe, Vabysmo emerges as a logical choice for first-line treatment for wet AMD, DME, and RVO.
1. World Health Organization. “DIABETES.” World Health Organisation, WHO, 2023, www.who.int/news-room/fact-sheets/detail/diabetes.
2. Goldberg R, Kolomeyer AM, Nudleman E, et al. Faricimab reduces macular leakage vs aflibercept in patients with DME. Presented at: the Association for Research in Vision and Ophthalmology; April 23-27, New Orleans, LA.
3. Regula JT, Lundh von Leithner P, Foxton R, et al. Targeting key angiogenic pathways with a bispecific CrossMAb optimized for neovascular eye diseases [published correction appears in EMBO Mol Med. 2019. EMBO Mol Med. 2016;8(11):1265-1288.
4. Oshima Y, Deering T, Oshima S, et al. Angiopoietin-2 enhances retinal vessel sensitivity to vascular endothelial growth factor. J Cell Physiol. 2004. 199: 412-417. https://doi.org/10.1002/jcp.10442.
5. Khanani AM, Kotecha A, Chang A, et al. TENAYA and LUCERNE: Two-Year Results from the Phase 3 Neovascular Age-Related Macular Degeneration Trials of Faricimab with Treat-and-Extend Dosing in Year 2. Ophthalmology. 2024;131(8):914-926.
6. Wong TY, Haskova Z, Asik K, et al. Faricimab Treat-and-Extend for Diabetic Macular Edema: Two-Year Results from the Randomized Phase 3 YOSEMITE and RHINE Trials. Ophthalmology. 2024;131(6):708-723
7. Tadayoni R, Paris LP, Danzig CJ, et al. Efficacy and Safety of Faricimab for Macular Edema due to Retinal Vein Occlusion: 24-Week Results from the BALATON and COMINO Trials. Ophthalmology. 2024;131(8):950-960.
8. Vabysmo (faricimab-svoa) Prescribing Information. Genentech, Inc. 2024.
9. “Access & Support: VABYSMO® (Faricimab-Svoa).” Vabysmo-Hcp.Com, 6 Sept. 2024, www.vabysmo-hcp.com/namd/financial-support/access.html.
* Primary endpoint in wet AMD trials was measured by the ETDRS letter score and tested for non-inferiority using a margin of 4 letters. Differences in LS means for VABYSMO were +0.7 letters (CI: [95%] -1.1, +2.5) in TENAYA; and 0.0 letters (CI: [95%] -1.7, +1.8) in LUCERNE. Primary endpoint in DME trials was measured by the ETDRS letter score and tested for non-inferiority using a margin of 4 letters. Differences in LS means in YOSEMITE were +0.7 letters (CI: [97.5%] -1.1, +2.5) for VABYSMO Q4W–Q16W and -0.2 letters (CI: [97.5%] -2.0, +1.6) for VABYSMO Q8W. Differences in LS means in RHINE were +0.5 letters (CI: [97.5%] -1.1, +2.1) for VABYSMO Q4W–Q16W and +1.5 letters (CI: [97.5%] -0.1, +3.2) for VABYSMO Q8W. A non-inferiority margin was not available for year 2.
† For wet AMD, intervals are adjusted based on OCT and visual acuity evaluations 8 and 12 weeks after initial four monthly loading doses. For DME, if resolution of fluid based on CST is achieved, the interval may be modified based on CST and visual acuity in ≤ 4-week increment extensions, or ≤ 8-week increment reductions.
‡ Coverage determined based on Medicare Fee for Service and tracking of the largest payers for other books of business. Bevacizumab (not indicated for intraocular use) and biosimilars are excluded from the branded treatment category. Specific coverage may vary for individuals and plans. Data as of January 2024.
Important Safety Information & Indications
INDICATIONS
VABYSMO (faricimab-svoa) is a vascular endothelial growth factor (VEGF) inhibitor and angiopoietin-2 (Ang-2) inhibitor indicated for the treatment of patients with Neovascular (Wet) Age-Related Macular Degeneration (nAMD), Diabetic Macular Edema (DME), and Macular Edema following Retinal Vein Occlusion (RVO).
IMPORTANT SAFETY INFORMATION
Contraindications
VABYSMO is contraindicated in patients with ocular or periocular infection, in patients with active intraocular inflammation, and in patients with known hypersensitivity to faricimab or any of the excipients in VABYSMO. Hypersensitivity reactions may manifest as rash, pruritus, urticaria, erythema, or severe intraocular inflammation.
Warnings and Precautions
Endophthalmitis and Retinal Detachments
Intravitreal injections have been associated with endophthalmitis and retinal detachments. Proper aseptic injection techniques must always be used when administering VABYSMO. Patients should be instructed to report any symptoms suggestive of endophthalmitis or retinal detachment without delay, to permit prompt and appropriate management.
Increase in Intraocular Pressure
Transient increases in intraocular pressure (IOP) have been seen within 60 minutes of intravitreal injection, including with VABYSMO. IOP and the perfusion of the optic nerve head should be monitored and managed appropriately.
Thromboembolic Events
Although there was a low rate of arterial thromboembolic events (ATEs) observed in the VABYSMO clinical trials, there is a potential risk of ATEs following intravitreal use of VEGF inhibitors. ATEs are defined as nonfatal stroke, nonfatal myocardial infarction, or vascular death (including deaths of unknown cause).
The incidence of reported ATEs in the nAMD studies during the first year was 1% (7 out of 664) in patients treated with VABYSMO compared with 1% (6 out of 662) in patients treated with aflibercept.
The incidence of reported ATEs in the DME studies from baseline to week 100 was 5% (64 out of 1,262) in patients treated with VABYSMO compared with 5% (32 out of 625) in patients treated with aflibercept.
The incidence of reported ATEs in the RVO studies during the first 6 months was 1.1% (7 out of 641) in patients treated with VABYSMO compared with 1.4% (9 out of 635) in patients treated with aflibercept.
Retinal Vasculitis and/or Retinal Vascular Occlusion
Retinal vasculitis and/or retinal vascular occlusion, typically in the presence of intraocular inflammation, have been reported with the use of VABYSMO. Healthcare providers should discontinue treatment with VABYSMO in patients who develop these events. Patients should be instructed to report any change in vision without delay.
Adverse Reactions
The most common adverse reactions (≥5%) reported in patients receiving VABYSMO were cataract (15%) and conjunctival hemorrhage (8%).
Pregnancy, Lactation, Females and Males of Reproductive Potential
Based on the mechanism of action of VEGF and Ang-2 inhibitors, there is a potential risk to female reproductive capacity, and to embryo-fetal development. VABYSMO should not be used during pregnancy unless the potential benefit to the patient outweighs the potential risk to the fetus. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for VABYSMO and any potential adverse effects on the breastfed child from VABYSMO. Females of reproductive potential are advised to use effective contraception prior to the initial dose, during treatment and for at least 3 months following the last dose of VABYSMO.
You may report side effects to the FDA at (800) FDA-1088 or www.fda.gov/medwatch. You may also report side effects to Genentech at (888) 835-2555.
Please see additional Important Safety Information in the full VABYSMO Prescribing Information.


