Many studies over the past 2 decades have shown that real-world outcomes in neovascular AMD (nAMD) often fall far short of the results achieved in the clinical trial setting. The reasons for this underperformance are numerous and often specific for an individual patient; however, this variability is almost certainly driven by the high injection and office visit burden that our current anti-VEGF agents require for optimal disease control. Recently approved longer acting anti-VEGF drugs may help with increasing treatment intervals, but even more durable solutions, like sustained delivery of tyrosine kinase inhibitors (TKIs), could be key to addressing this unmet need in patient care and help us achieve better long-term outcomes for our nAMD patients.1
Unmet Needs in Age-related Macular Degeneration
Retina Today (RT): What are the key reasons that the visual outcome results of randomized, controlled clinical trials have been difficult to replicate in the real world? Is the burden of frequent injections a factor?
Ferhina S. Ali, MD, MPH: The difference between the results in the clinical trial setting versus real world outcomes is widely recognized by retina specialists. Patients in the clinical trial setting must follow tight, controlled visit and treatment protocols with strict retreatment intervals and disease activity assessments. Importantly, patients in sponsored clinical trials are well supported by trial sponsors for these visits (e.g., transportation and follow-up) as well as in the clinic with a strong commitment of the research and clinical staff.
Naturally, once the trial is complete and the product is hopefully approved by the FDA, it can often be difficult for patients to adhere to the same sort of regimens.
This is validated when we look at real-world database studies, such as the Intelligent Research in Sight (IRIS) registry, showing that on average, patients receive far fewer injections in the first year of treatment compared to clinical trial protocols.2 We also know that patients who receive treatments at the intervals as prescribed in the FDA label have much better outcomes, particularly in the first year of real-world treatment.2
Roger A. Goldberg, MD, MBA: This difficulty is not only seen at the patient level, but also at the clinic level, where staff often do not have a line of sight into what may be preventing/prohibiting patients’ ability to maintain regular follow up. Real-world data that are available clearly show that patients are being undertreated with current anti-VEGF agents, making it difficult to achieve the results—both in terms of visual gains and retinal thickness improvements – that have been seen in the clinical trials. Does it feel like I am undertreating my patients? No, because I only see the patients that come in and remain adherent to their treatment regimen. I am not seeing the patients who no-show, until they eventually come back in, and sometimes it’s a bad outcome, like a bleed, with irrecoverable vision loss, and sometimes it’s more subtle, with seemingly stable vision despite worsening exudation. Yet the ‘big data’ tell me this yo-yoing of fluid isn’t good for my patients’ vision over time.
Additional data from the IRIS registry have shown that as many as 12% of patients being treated for nAMD were lost to follow up in the first year of treatment.3 Advancing age correlated with higher rates of loss to follow-up.
I was fortunate to be able to work with one of the CPMC residents to look at our own practice’s data on missed visits at the start of the COVID pandemic. If a patient missed their scheduled follow up by 1 month or less, their risk of significant vision loss of three lines or more was 5%. However, if they missed their follow up by 3 or more months, that number skyrocketed to 33%.4
COVID was obviously a strange time, but even outside of a global pandemic, patients may have many and varying factors that contribute to their inability to adhere strictly to an injection schedule. I don’t see this as the patient’s fault, but as a limitation of the current treamtents.
Patients may have concurrent medical issues requiring them to see other doctors, are often dependent on others for transportation, and often stop seeing a ‘benefit’ after the first few injections.
Dr. Ali: Our electronic medical record systems come equipped with scheduling platforms, but myriad factors can complicate these set schedules. Managing multiple prior authorizations, step-therapy appeals, and ensuring alignment with desired and optimal follow-up schedules can be significant challenges for a practice. On the patient side, many patients do not meet the visual acuity criteria to operate a vehicle and/or do not feel comfortable doing so, requiring a family member or friend to take them back and forth to appointments. Essentially, they are at the mercy of a confluence of logistical issues in the ongoing management of a chronic disease, in addition to any other ailments or debilities they may have. It’s easy to see how this can unravel. While advances in imaging technology have allowed earlier detection, the challenges of regular follow-up and monitoring remain.
To Dr. Goldberg’s point, we, as retina specialists, see the patients that adhere to our recommendations, and in my experience, patients tend to be diligent about making sure they’re getting the treatment as outlined by the specialist, especially in the first year of treatment. We do know, however, that treatment fatigue exists and even increases over time.
From an optimal treatment algorithm and discussion standpoint, I tell patients that in the first year of treatment, they can expect to receive anywhere from 6 to 12 injections. How I extend their treatment interval is determined by vision and anatomic outcomes. The pharmacokinetics of bolus anti-VEGF injections have peaks and troughs; the troughs may be magnified with extended breaks during a treatment cycle. In a treat-and-extend scenario, the OCT scans should remain consistent with little variability, and in this treatment algorithm, the dosing intervals should be decreased or increased based on disease activity.
The newer generation of anti-VEGF treatments, such as brolucizumab (Beovu®, Novartis), faricimab (Vabysmo®, Genentech), and aflibercept 8 mg (Eylea HD®, Regeneron), were designed for the potential to provide greater durability, and the way they were studied, aside from efficacy and safety, was to understand just that.5-7 However, we are still in the determination phase of seeing how long these agents work in the real-world setting. The trial designs for these agents were such that they looked closely at disease activity, but at times allowed for more fluid accumulation than the average retina specialist has historically tolerated. Perhaps this may end up meaning that retina specialists have been too strict and there may be more tolerance for fluid, especially subretinal, in the setting of stable vision. We need longer term experience and data to know that. Regardless, it’s clear that greater durability remains an unmet need.
Retinal Thickness and the Impact on Long-term Outcomes
RT: How does variation in retinal thickness contribute to poor long-term visual outcomes/real-world outcomes that differ from clinical trials?
Dr. Goldberg: Data collected over the course of 4 years from CATT and IVAN show that more variations in retinal thickness correlate to poorer visual acuity and anatomic outcomes.8
With a treat-and-extend protocol, we are trying to increase the intervals between injections and push the limits of durability with minimal/no fluid reaccumulation between visits. What we are trying to avoid, which isn’t always possible, is the yo-yo effect with retinal thickness variability which has been shown to negatively impact visual outcomes in the long term.
Dr. Ali: I agree. Take the scenario in which I am trying to get to an 8-week interval using a treat-and-extend protocol. If on follow-up I see that the vision has worsened or the fluid has increased, I will likely increase the frequency of injection. As Dr. Goldberg stated, fluid fluctuations in the retina have negative effects on functional vision, so there continues to be a need for longer-acting treatments for nAMD that deliver continuous control to maintain anatomic stability.
Voloranib: Mechanism of Action
RT: How does *DURAVYU™ (vorolanib intravitreal insert, EyePoint Pharmaceuticals) target the VEGF pathway in a way that is unique among currently available nAMD therapies?
Dr. Ali: The mechanism of action of Vorolanib, a TKI, is quite different from current anti-VEGF agents. While anti-VEGF’s block some VEGF ligands extracellulary, Vorolanib provides pan-VEGF inhibition by blocking all three VEGF receptors (VEGFR-1, -2, and -3) intracellularly inhibiting angiogenic and inflammatory effects of the VEGF pathway but it does not inhibit the Tie-2 pathway. This is particularly important, as we know that by inhibiting the Tie-2 pathway, vascular instability is promoted via the cascade of pericyte drop out and inflammatory responses.9
Dr. Goldberg: All of the FDA-approved agents bind to free VEGF-A ligands circulating in the extracellular space. However, inhibiting the VEGF-A ligand can also lead to an upregulation in VEGF-C, which binds to VEGF receptors 2 and 3 potentially activating the angiogenic pathway. Vorolanib is a TKI that intracellulary binds to all VEGF receptors inhibiting the angiogenic pathway regardless of the presence of extracellular VEGF-A or -C ligands. The TKI approach is a different way of targeting the disease via pan-VEGF suppression to halt the disease cascade. Because TKIs are small molecules, bolus injections of them would rapidly disperse; therefore, a drug delivery system is required to enable the drug to exert its anti-VEGF effect over a pre-determined and extended amount of time (Figure 1).

Figure 1. Vorolanib, delivered in the Durasert E platform, provides controlled release of drug for at least 6 months.
Courtesy of Regillo CD, on behalf of the DAVIO 2 Investigators.
DURAVYU, being developed by EyePoint, delivers this innovative combination via its bioerodible insert technology, Durasert E. DAVIO 2,11 the largest sustained-release TKI study to date, evaluating the intravitreal insert with Vorolanib, demonstrated visual acuity stability (Figure 2A) and anatomic control over 32 weeks (Figure 2B), with 63% of study participants requiring no supplemental injections of aflibercept 2mg (Figure 3). The data presented suggests that having a TKI on board provides the potential for longer intervals between anti-VEGF injections, and less variability in vision and retinal thickness.
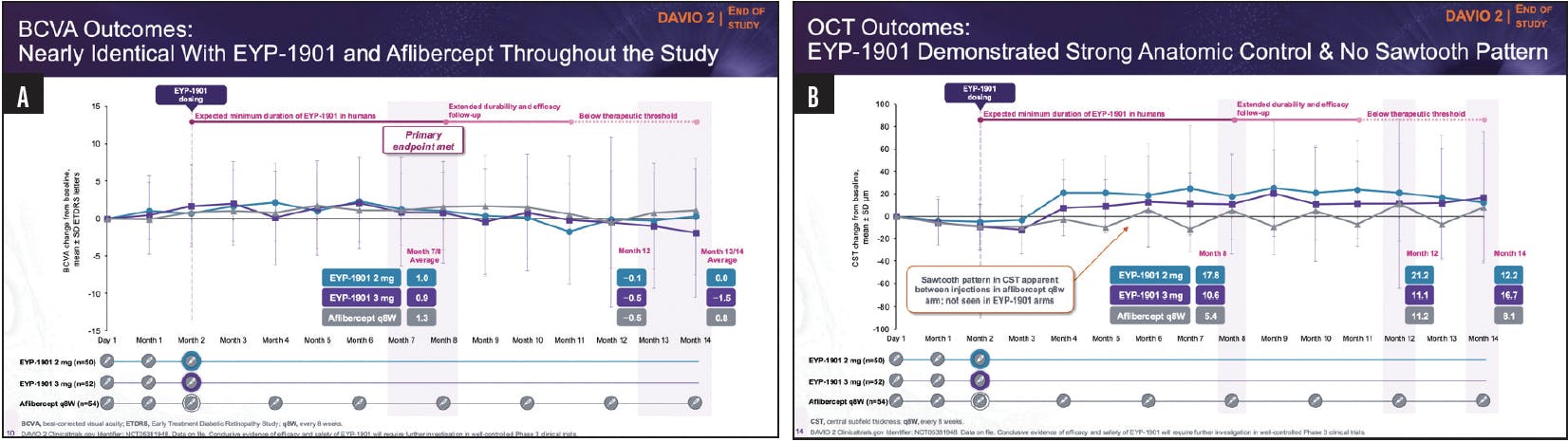
Figure 2. In a phase 2 study, the Voloranib insert met its primary endpoint of noninferiority with both doses compared to aflibercept 2 mg (A). OCT results showed no sawtoothing in the patient treated with the voloranib insert vs aflibercept 2 mg (B).
Courtesy of Regillo CD, on behalf of the DAVIO 2 Investigators.

Figure 3. Sixty-three percent of patients in each voloranib treatment arm did not require supplemental injection up to 32 weeks.
Courtesy of Regillo CD, on behalf of the DAVIO 2 Investigators.
An analogy can be made with long-acting and bolus (short-acting) insulins for optimal control of their diabetes.11 Many diabetic patients will do well managing their blood sugar with only long-acting insulins; this is akin to the patients who received a single DURAVYU in DAVIO 2 and didn’t require any supplemental treatment with aflibercept. Similarly, just like in diabetes, where some patients will require additional short-acting bolus insulin in addition to their long-acting insulin, so too did some patients who received DURAVYU need a periodic bolus injection of aflibercept. My sense is that when long-acting TKIs come to market, many wet AMD patients will be able to go 6 months or longer between injections, while some may require periodic supplementation with shorter-acting bolus anti-VEGF agents. And so, in the real world of nAMD treatment, we will likely see a multi-MOA approach to treating nAMD.
RT: DURAVYU uses zero order kinetics to extend treatment intervals. What is the benefit of zero order kinetics?
Dr. Goldberg: Zero-order kinetics improve durability by delivering a consistent and controlled amount of vorolanib over time. Durasert E leverages zero-order kinetics to continuously deliver a predictable and therapeutic amount of vorolanib to the retina for ~9 months. This eliminates the ‘boom-and-bust’ cycle of traditional anti-VEGF injections, which contributes to the saw-tooth effect or variation in retinal thickness that ultimately affects functional vision over time. The Durasert sustained release platform has already been FDA approved as part of two other agents, and DURAVYU brings a bioerodible version of it to nAMD.
Treatment Algorithms with Voloranib
RT: How do you anticipate using DURAVYU, if it is approved, in practice?
Dr. Ali: When a new therapy becomes available, particularly a first-in-class medication, it is exciting. The phase 2 results provide promising signals with regard to safety and efficacy. The head-to-head comparisons to aflibercept 2 mg showed noninferiority in efficacy and showed a comparable safety profile.
My approach will be to mirror the clinical trial protocol and I’m excited to see how my patients do. The advantage of vorolanib in the Durasert E platform is durability without potentially sacrificing visual acuity or anatomical control. Furthermore, the ability to use this agent in combination with anti-VEGF injections is encouraging. The trials have outlined a nice approach in combining anti-VEGF therapy initially upfront with the TKIs, which, as mentioned prior, have a different mechanism of action. We know that our patients are not homogeneous, and we know from our history of using anti-VEGF agents that some patients respond differently in terms of how long we can extend the intervals. Treatment for AMD is not one-size-fits fits all and to have another tool in our hands to provide better disease control will be beneficial.
Dr. Goldberg: I believe that TKIs, such as vorolanib, will provide us with more flexibility in how we treat our patients. There is a lot of downside risk with current anti-VEGF therapies because, as previously discussed, not all patients are able to adhere to tightly managed follow up and injections, and they ultimately lose vision over time. It would be advantageous for both my patients and my clinic to be able to extend 50+% of patients to extended intervals. My sense is that most retina specialists won’t feel comfortable extending patients to 6-month intervals without evaluating them at least every three months. That said, if we could move wet AMD to a four-visit-per-year disease, that would be a huge leap forward for most patients.
1. Patel S, Storey PP, Barakat MR, et al. Phase I DAVIO Trial: EYP-1901 bioerodible, sustained-delivery vorolanib insert in patients with wet age-related macular degeneration. Ophthalmol Sci. 2024. 9;4(5):100527.
2. MacCumber MW, Yu JS, Sagkriotis A, et al. Antivascular endothelial growth factor agents for wet age-related macular degeneration: an IRIS registry analysis. Can J Ophthalmol. 2023;58(3):252-261.
3. Khurana RN, Li C, Lum F. Loss to follow-up in patients with neovascular age-related macular degeneration treated with anti-VEGF therapy in the United States in the IRIS® registry. Ophthalmology. 2023;130(7):672-683.
4. Harris S, Goldberg RA. COVID 19 and the effects of missed intravitreal injections on patients with exudative macular degeneration. CPMC Barkan Days lecture. June 2022; San Francisco, CA.
5. Dugel PU, Koh A, Ogura Y, et al. HAWK and HARRIER: Phase 3, multicenter, randomized, double-masked trials of brolucizumab for neovascular age-related macular degeneration. Ophthalmology. 2020;127(1):72-84.
6. Heier JS, Khanani AM, Quezada Ruiz C, et al. Efficacy, durability, and safety of intravitreal faricimab up to every 16 weeks for neovascular age-related macular degeneration (TENAYA and LUCERNE): two randomised, double-masked, phase 3, non-inferiority trials. Lancet. 2022;399(10326):729-740.
7. Lanzetta P, Korobelnik JF, Heier JS, et al. Intravitreal aflibercept 8 mg in neovascular age-related macular degeneration (PULSAR): 48-week results from a randomised, double-masked, non-inferiority, phase 3 trial. Lancet. 2024;403(10432):1141-1152.
8. Evans RN, Reeves BC, Maguire MG, et al. Associations of variation in retinal thickness with visual acuity and anatomic outcomes in eyes with neovascular age-related macular degeneration lesions treated with anti-vascular endothelial growth factor agents. JAMA Ophthalmol. 2020;138(10):1043-1051.
9. Khanani AM, Russell MW, Aziz AA, et al. Angiopoietins as potential targets in management of retinal disease. Clin Ophthalmol. 2021(4);15:3747-3755.
10. Sheth V. Assessing supplemental injection use across groups in the phase 2 DAVIO 2 trial of EYP1901 vs aflibercept in wet age-related macular degeneration. Paper presented by Sheth V on behalf the DAVIO 2 investigators and coauthors at the American Society of Retina Specialists (ASRS) 42nd Annual Meeting. Stockholm, Sweden. July 17-20, 2024.
11. Laranjeira FO, de Andrade KRC, Figueiredo ACMG, et al. Long-acting insulin analogues for type 1 diabetes: An overview of systematic reviews and meta-analysis of randomized controlled trials. PLoS One. 2018;13(4):e0194801.


