After more than 2 years of availability, complement inhibition therapy for geographic atrophy (GA) secondary to age-related macular degeneration (AMD) is no longer new with unknown efficacy and safety outcomes. Indeed, we have started to accumulate data on how pegcetacoplan (Syfovre, Apellis Pharmaceuticals) and avacincaptad pegol (Izervay, Astellas Pharmaceuticals) work in real-world patients.
Although these treatments are not new, we still have a lot to learn about them. Data from well-controlled pivotal studies tell us about the safety and efficacy of a specific treatment, but we must turn to real-world experiences to learn how patients are interacting with these treatments in clinical settings.
To that end, I sat down with my colleagues Ferhina S. Ali, MD, MPH; Margaret Chang, MD, MS; and David A. Eichenbaum, MD, to film a video and podcast series in which we shared cases that illustrated the breadth of GA treatment strategies and patient profiles we manage in our clinics.
We decided that, in addition to sharing clinical data, we would include a social history of each patient to emphasize the humanity of the patients before us—they are, after all, people with complex lives whose circumstances drive their decisions, and we cannot understand our patients in the chair if we discount their lives outside of it.
You can find videos and podcasts from our discussion via QR codes in this compendium. The discussion reproduced herein has been edited for brevity and clarity. I hope you find it useful.
—John W. Kitchens, MD
Case No. 1: Bilateral Geographic Atrophy in an Active, Motivated Patient With Good Vision in Her Right Eye
David A. Eichenbaum, MD: A 79-year-old White woman presented to the clinic with complaints of subjectively declining vision. Her VA was 20/32 OD and 20/100 OS. She had an ocular history of cataract surgery several years ago. Her medical history included controlled hypertension, treated breast cancer, and mild COPD from a prior history of smoking; none of these were remarkable, in my estimation.
The patient’s social history is important here: she cares for her elderly husband and a great-grandchild. As the primary driver in her family, she was worried about losing vision in her better eye. Losing the ability to drive safely would significantly hinder her family’s life.
I performed fundus autofluorescence (FAF) imaging on presentation (Figure 1). Her left eye, which is her worse eye, showed extensive GA lesions, while her right eye showed multifocal, medium-sized lesions. Optical coherence tomography (OCT) of the right eye showed evidence of limited hypertransmission (Figure 2) whereas the left eye showed significant hypertransmission (Figure 3).
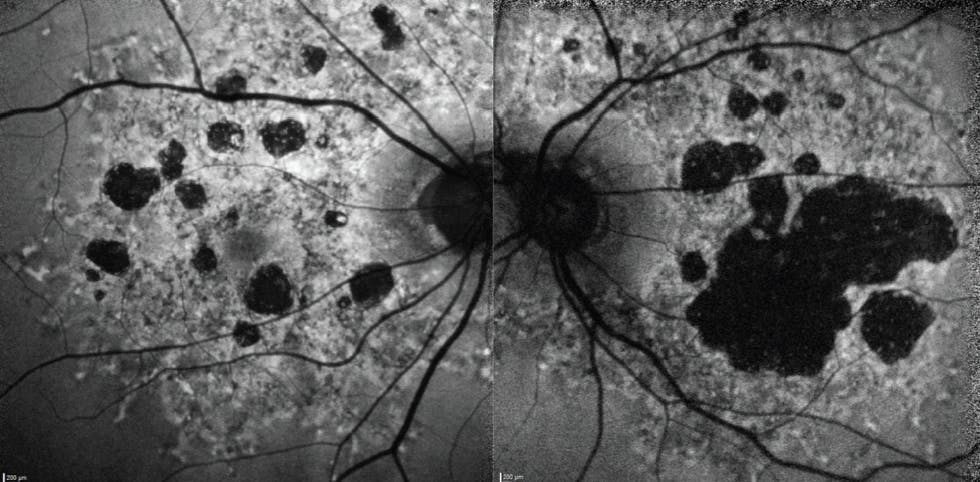
Figure 1. FAF imaging revealed extensive bilateral GA lesions. The patient’s right eye, which was her better-seeing eye, had medium-sized multifocal lesions, while her left eye had a large central lesion.

Figure 2. OCT imaging of the patient’s right eye showed modest (but noticeable) hypertransmission consistent with outer retinal atrophy.

Figure 3. OCT imaging of the patient’s left eye depicted extensive hypertransmission consistent with advanced GA.
John W. Kitchens, MD: How did you decide to treat this patient?
Dr. Eichenbaum: We discussed treatment with pegcetacoplan. I dosed her left eye (ie, her worse-seeing eye) with a single injection to see if she would have an adverse reaction. After she did not show evidence of inflammation, the patient elected bilateral monthly pegcetacoplan treatment. Over the first year of treatment, she did not have any adverse events. She preserved vision centrally in her right eye but steadily lost vision in her left eye.
During that time, her husband grew sicker, which meant more driving and more medical appointments. Given that her left eye’s vision had deteriorated significantly, she elected for every-other-month (EOM) treatment in her left eye but wished to maintain monthly injections in her right eye (Figure 4). FAF imaging showed minimal growth of lesions in her left eye, and her BCVA OD was 20/40+ (down from 20/32 at presentation).

Figure 4. FAF imaging showed modest growth of GA lesions in the patient’s right eye, and she elected to continue monthly injections in that eye. Given the massive atrophic regions in her left eye, the patient elected for EOM treatment in that eye.
After another year of monthly treatment, her right eye retained good vision (ie, 20/40 BCVA OD) even though FAF imaging showed continued GA lesion growth (Figure 5).

Figure 5. FAF imaging showed growth of GA lesions in the right eye. Still, the patient’s BCVA remained mostly unchanged after the second year of treatment.
At present, the patient is widowed and cares for two great-grandchildren. She still drives and has elected for injections every 6 weeks in her right eye as her schedule has grown busier. We’ll do everything we can to maintain her vision, but I fear that she will soon lose vision that allows safe driving.
Dr. Kitchens: Tell us more about how you arrived at a 6-week pegcetacoplan regimen.
Dr. Eichenbaum: The data from the pivotal studies that led to the FDA approval of pegcetacoplan did not include a 6-week treatment arm, so I understand why this strategy might garner some focus from my colleagues. I was grateful to keep her on monthly therapy for 2 years; when the circumstances of her life changed and the best she could do was a 6-week interval, I accommodated her. In fact, even if she wanted 8-week dosing intervals, I’d still feel that I’m within the parameters of the evidence from the phase 3 studies.
After 2 years of monthly dosing, her BCVA only dropped from 20/32 OD to 20/40 OD. If part of our goal as providers is to buy patients time, then I am satisfied with this outcome.
Dr. Kitchens: This patient presented with poor vision in her left eye, so she understood the natural history of GA. Motivation might have been easier for her. How do you motivate patients who have good bilateral vision and bilateral GA?
Dr. Eichenbaum: You’re correct that this patient was motivated, very likely in part by her subjective experience with vision deterioration. And you’re also correct that patients with good vision are challenging, as they are often skeptical about initiating treatment. I often instruct patients to return in 3 to 4 months, at which point I ask if they are experiencing compromised quality of vision—if, for example, they have trouble recognizing faces or difficulty reading at night. If so, they may begin to better understand the nature of their vision loss and may be more open to therapy.
Ferhina S. Ali, MD, MPH: I have noticed that patients with a family history of vision loss quickly grasp their prognosis and the stakes of GA. I also adopt Dr. Eichenbaum’s technique of requesting follow-ups with patients whose GA has not yet adversely affected their central vision, as continued communication and touchpoints are often the key to educating patients, particularly with relatively new treatment options.
Margaret Chang, MD, MS: Don’t rule out sharing images with patients. I have found that images such as the ones from Dr. Eichenbaum’s case help patients quickly understand GA. Longitudinal FAF photographs illustrate how GA is growing in that particular eye and sometimes moves them toward treatment.
Dr. Kitchens: This patient’s right eye does not have significant foveal involvement just yet, which likely accounts for her in-tact functional vision. Did you consider the lesions’ distance from the fovea when evaluating this patient?
Dr. Eichenbaum: For most patients, I assume that GA lesions are moving toward the fovea, regardless of when we begin treatment. So much disease activity takes place in the junctional zone, which is the area of active disease immediately outside a GA lesion. Emerging treatment will likely address this precise area, as it represents tissue that is not yet atrophied.
CASE NO. 1: BILATERAL GEOGRAPHIC ATROPHY IN AN ACTIVE, MOTIVATED PATIENT WITH GOOD VISION IN HER RIGHT EYE

Case No. 2: Rapid Geographic Atrophy Growth in an Active Man Who Elected for Bimonthly Treatment
Dr. Chang: A 71-year-old White man presented in 2014 upon referral for an intermediate dry AMD evaluation. Indeed, I diagnosed the patient with intermediate dry AMD. He was a former smoker (he quit 20 years prior to presentation), and his medical history included hypertension and hypercholesterolemia. He was particularly concerned about his vision, as he had a family history of blindness, albeit from unknown origin. He was instructed to use AREDS vitamins and return for follow-up visits.
In November 2019, I began to observe the type of hypertransmission defects on OCT that indicate the presence of GA in his right eye (Figure 6). His BCVA was 20/30 OD and 20/50 OS.

Figure 6. Early hypertransmission defects on OCT imaging in the right eye indicated early GA. The left eye appeared to still have intermediate dry AMD per OCT imaging.
GA grew quickly in his right eye. When the patient retuned in December 2020, GA in his right eye was pronounced on OCT (Figure 7). BCVA fell from 20/30 OD to 20/50 OD; the vision improved slightly in the contralateral eye, moving from 20/50 OS to 20/40 OS.

Figure 7. Hypertransmission on OCT suggested significant GA growth in the right eye in the span of 1 year, which corresponded with a drop in BCVA from 20/30 OD to 20/50 OD. The left eye remained stable with intermediate dry AMD.
The patient developed concomitant cataracts, which were removed in approximately 2022. It did little to change his BCVA. In January 2024, GA had progressed in his right eye, as seen on FAF (Figure 8). His left eye had progressed from intermediate dry AMD to GA. His BCVA was 20/100 OD and 20/40 OS. The patient elected for bilateral treatment with avacincaptad pegol EOM.

Figure 8. FAF imaging revealed GA in the right eye with foveal involvement. The patient’s left eye showed the first signs of GA after years of stability with intermediate dry AMD.
After 1 year, GA had progressed in both eyes, as seen on FAF (Figure 9). Both eyes had foveal involvement. The patient’s BCVA dipped to 20/150 OD and 20/60 OS. Despite this vision loss, the patient remains committed to treatment as of the time of this conversation.

Figure 9. Bilateral GA lesion growth involving the fovea led to significantly decreased vision, even after 1 year of treatment with complement inhibition therapy.
Dr. Kitchens: Why did the patient want EOM treatment rather than monthly treatment?
Dr. Chang: The patient has an active lifestyle and sees monthly treatment as too disruptive. As of 2025, which is 11 years after I first saw him, he continues to visit my office. At 82 years, he still actively manages a casino he owns and says he plans to keep working until he can’t work any further.
Dr. Kitchens: I’m surprised at how quickly this patient’s GA lesion in the right eye grew from 2019 to 2020. Did it surprise you?
Dr. Chang: It did. Obviously there is no way to tell how quickly GA will grow in each patient, but when you see rapid growth such as this, it challenges the often-held notion that GA is a slow-moving disease. In some patients, it progresses rapidly.
Dr. Kitchens: Some providers might have stopped treating the patient’s right eye, which exhibited significant GA growth even during treatment. Why do you continue to treat it?
Dr. Chang: The patient drives this decision. He knows that GA is a progressive disease that, if left unchecked, will rob him of his vision. He wants to preserve vision in both eyes for as long as possible, and bilaterial complement inhibition therapy is the best strategy available. If the patient is motivated, then I’ll support continued treatment.
Dr. Kitchens: Still, at some point, treatment is futile. When does that point occur?
Dr. Eichenbaum: Our current therapeutic options are limited, and they do not restore vision. I am much less aggressive with treatment when BCVA reaches 20/200 or worse. Of course, I listen to patients: some may have poor measurable vision but still have useful subjective vision. If that patient still wants treatment, I’ll continue to treat. But if they’re not motivated to continue injections, I won’t press them to do so.
Dr. Kitchens: What roles do caregivers play in the lives of patients with GA?
Dr. Ali: Caregivers are the key to compliance for many of our patients, especially those who cannot drive to their appointments due to vision loss. With this in mind, I typically ask patients whom I have diagnosed with GA to return with a caregiver at their next appointment. Having an extra set of ears in the room helps the patient decide what’s best for them. Plus, inviting the caregiver to the appointment acquaints him or her to the treatment burden required for adherent therapy and serves as a chance for us to educate the caregiver on GA.
Dr. Kitchens: Engaging family members is such a critical step for patients, as they often are the ones who encourage a patient to initiate treatment.
Dr. Ali: Giving patients literature that they can review at home helps educate them about the importance of GA. The manufacturers of the two available complement inhibitor treatments provide useful pamphlets that seem to fly off the shelves in my office.
Dr. Kitchens: Can you tell us why you were comfortable with EOM dosing for this particular patient?
Dr. Chang: In this case, the patient lived an active lifestyle and found monthly treatment too burdensome. Because he was a motivated patient who I believed would remain compliant, I was comfortable with EOM therapy.
But you must be careful with EOM treatment: if you suspect a patient won’t comply with EOM treatment, they may end up only receiving treatment every 3 or 4 months. We have little data on the efficacy of treatment dosed that infrequently.
Dr. Eichenbaum: I start almost all my patients on monthly dosing for this exact reason. If they feel the burden of treatment is too high, we can scale back to treatment every 6 weeks or EOM. But if I start them on EOM treatment and they request a frequency reduction, then they’re essentially requesting a dosing regimen unsupported by clinical trial evidence.
CASE NO. 2: RAPID GEOGRAPHIC ATROPHY GROWTH IN AN ACTIVE MAN WHO ELECTED FOR BIMONTHLY TREATMENT

Case No. 3: Unilateral Treatment in a Patient With Bilateral Geographic Atrophy
Dr. Ali: A 73-year-old White man was referred to my clinic from optometry. He had a family history of AMD; his mother lost her vision due to AMD, but he was unsure if she had a history of treatment. He was pseudophakic in both eyes and reported a developing blind spot in his right eye.
Importantly, at the time of the consultation, the patient reported being an active retiree who lived alone. He said he was passionate about his work as a mixed media artist, with a particular focus on wheel pottery. This patient explained to me that his artistry required both close-up work and depth perception, and he was worried that losing vision would mean that he could no longer practice this hobby. Given these facts, it was clear to me that he was a motivated patient.
Color fundus photography (CFP) and OCT imaging from his initial visit revealed an extrafoveal lesion with encroaching subfoveal involvement in the right eye. The left eye demonstrated a small extrafoveal GA lesion (Figure 10). His BCVA was 20/60 OD and 20/40 OS.

Figure 10. CFP imaging (top row) and OCT imaging (bottom row) showed evidence of bilateral GA. Slight foveal involvement was observed in the right eye, while the left eye showed strictly extrafoveal disease.
Dr. Kitchens: This seems like a patient who could greatly benefit from complement inhibition therapy. How did you broach the subject with him?
Dr. Ali: He first presented in June 2023, which means that he had two options to choose from. He had heard from the referring optometrist that treatments were an option for GA, and he returned 3 months after the initial appointment to revisit the idea of treatment. I performed FAF imaging at that appointment (Figure 11). I observed perilesional hyperautofluorescence in his right eye, which suggested that the lesion was likely to grow in that direction. Imaging of the left eye confirmed the finding from CFP on the first visit of an extrafoveal lesion, and I observed no hyperautofluorescence. The patient’s BCVA was 20/50+ OD and 20/30 OS, which was relatively similar to BCVA measurements from his first appointment.

Figure 11. FAF imaging on the patient’s second visit showed an area of hyperautofluorescence in the right eye suggestive of potential growth. The left eye showed no such hyperautofluorescence.
The patient elected to proceed with complement inhibition therapy in the right eye, requesting a regimen of every 6 weeks.
I performed FAF 1 year after initiating treatment (Figure 12). The expected GA growth in his right eye did indeed manifest, and the GA lesion area was much larger on this visit. Although BCVA was 20/60 OD, he maintained good central vision. Remarkably, his left eye demonstrated little change over time. The BCVA was 20/40 OS, which means both eyes were relatively stable from baseline.

Figure 12. FAF imaging after 1 year of complement inhibition therapy revealed as-expected GA lesion growth in the right eye. GA growth in the left eye was not observed.
Dr. Kitchens: This is a young patient: he was only 73 at presentation. This challenges the notion that GA patients are elderly. Is he an outlier in your clinic?
Dr. Ali: I’m seeing younger patients present to my clinic because I think our referral networks are catching patients at earlier stages of disease. This is likely for two reasons: we have treatments that make referral worthwhile, and sophisticated imaging modalities are more accessible than ever. Patients who have a family history of AMD also seem to be particularly vigilant with their eyes and will present at the earliest signs of visual disruption.
Dr. Kitchens: This case shows the unpredictable nature of GA. I thought for sure that the left eye’s lesion would grow rapidly, but after 1 year, it hasn’t moved much at all. Meanwhile, the eye you did treat progressed. How do you keep the patient motivated to continue treatment in an eye where GA is progressing?
Dr. Ali: I rely on figurative language. I tell patients that GA is like a runaway train, and we can’t bring it back to the station. But with complement inhibition therapy, we might be able to slow it down. I also emphasize that the two treatment options at our disposal are the first-ever treatments for this disease, and that prior to this, we had no treatments. Even a modest treatment effect is better than no treatment effect.
Dr. Eichenbaum: We retina specialists are so used to restoring vision when we inject treatments. When treating GA, we need to shift our mindset so that it’s more like that of a glaucoma specialist who treats patients to slow their disease progression.
Patients often ask what they can expect with injections. I am direct with them: their vision is never going to improve with these treatments. But we can hope to keep them as close as possible to today’s vision if they undergo treatment. Yes, GA will progress with or without treatment. But with treatment, there’s a chance to blunt that progression.
Dr. Kitchens: No treatment is without risk. How do you articulate the risks of complement inhibition therapy?
Dr. Chang: I don’t want to overwhelm patients with too much information. I typically start with a review of injection-related risks, which are not specific to complement inhibition therapies.
When I move to speaking about risk associated with complement inhibition, I avoid technical terms like vasculitis or intraocular inflammation (IOI), as patients sometimes confuse these for infections. Instead, I describe these complications as something akin to an allergy. Next, I explain that their risk of this reaction is 1 in a few thousand, and that it may lead to irreversible vision loss. For that reason, if we start treatment, we’ll treat the worse-seeing eye just to make sure that a reaction doesn’t develop.
Risks of vasculitis and IOI are top of mind to retina specialists, but in my experience, patients are more concerned about the risk of conversion from GA to wet AMD. They know that wet AMD treatment requires another intraocular injection, and the patients who decline treatment due to the risk profile of complement inhibitors do so more often because they’re concerned about wet AMD conversion rather than vasculitis or IOI. This is true even when I tell them that they’re already at risk of developing wet AMD regardless of whether they initiate complement inhibition, and after I explain that we have several highly effective treatments for wet AMD.
Dr. Kitchens: How do you monitor these patients as they return?
Dr. Ali: CFP and FAF are adequate for tracking disease if you’re looking at 6- or 12-month intervals, but for shorter time frames (eg, patients who present monthly), I use OCT imaging with near-infrared reflectance (NIR). OCT with NIR images help patients understand where their lesion growth has occurred lately. Plus, it helps me detect any potential conversion to wet AMD.
Dr. Eichenbaum: My wet AMD patients don’t necessarily get an OCT image at every visit, but I think assessing GA patients via OCT imaging is valuable, as detecting wet AMD as early as possible is key to successful disease treatment.
CASE NO. 3: UNILATERAL TREATMENT IN A PATIENT WITH BILATERAL GEOGRAPHIC ATROPHY

Further Discussion: Comparing Complement Inhibitor Therapies, Assessing Adoption Hesitancy, and the (Possible) Role of AREDS Vitamins for GA Treatment
Dr. Kitchens: Safety is always a concern when treating patients with GA. How do you weigh safety versus the risk of undertreating or not treating at all?
Dr. Eichenbaum: The inflammatory events we are most worried about—IOI and vasculitis—are associated with the first dose of pegcetacoplan. If we are vigilant with patients dosed with pegcetacoplan, then we can reasonably monitor safety outcomes. As we discussed earlier, bilateral GA patients offer a unique opportunity insofar that we can inject their worse-seeing eye with pegcetacoplan to test for a treatment response.
Dr. Kitchens: What do you do in a monocular patient with GA?
Dr. Ali: In that case, I would lean toward the drug with fewer reported adverse events upon first injection (ie, avacincaptad pegol). It appears that the risk of first-injection inflammation associated with pegcetacoplan is approximately 1 in 4000.1 It’s always useful to keep that statistic handy when discussing safety with patients.

Retina specialists are cautious with new mechanisms of action, so I understand why some providers took a wait-and-see approach to integrating complement inhibition therapies in their clinics. Now that we have data out to 48 months on pegcetacoplan, I think many concerns about the elevated risk of complications have subsided, and a greater understanding of efficacy has been achieved.
Dr. Kitchens: Earlier, we discussed that a mindset shift is needed when employing GA treatments—that is, that we are buying patients time and trying to preserve vision and prevent progression rather than trying to restore vision, as we often do with anti-VEGF agents for exudative disease. Is it possible that the treatment effects associated with current GA options play a role in providers’ reluctance to adopt these therapies?
Dr. Ali: The inability to truly demonstrate efficacy could possibly be a factor for some providers. Clinical trial endpoints focused on vision loss and atrophy growth might be difficult to digest when we typically encounter endpoints centered on vision gains and anatomic improvement. As retina specialists become more comfortable with these endpoints, they may become more comfortable with new treatment options. Even if a retina specialist is unconvinced of the efficacy of complement inhibition therapy, they still owe it to their patients to discuss these FDA-approved options.
Dr. Chang: Retina has a history of successful innovations that had slow launches. It might be difficult to remember, but plenty of surgeons continued to use 20-gauge instruments even after the evidence emerged about the benefits of small-gauge surgery. Now, 20-gauge instruments, while still present in some ORs, are difficult to come by.
Dr. Kitchens: Is the only difference between these two drugs found in their safety profiles?
Dr. Eichenbaum: I wish we had a response to that question, but without a head-to-head study, it’s impossible to have a certain answer. I would estimate that we have a more robust dataset articulating outcomes following pegcetacoplan, which some providers might find useful to lean on.
Dr. Kitchens: The latest data from Keenan et al2 found that AREDS supplementation slowed the progression of extrafoveal GA lesions to the fovea compared with no intervention, but that overall area of lesion growth was similar among patients who took AREDS vitamins and those who did not. What role do AREDS vitamins play in patients with GA?
Dr. Chang: I advise patients with intermediate AMD or GA to consume AREDS vitamins, but always with a caveat similar to the one I offer with complement inhibition therapy: this approach will only slow the progression of disease rather than reverse it. I want to ensure that patients’ expectations are properly calibrated.

Dr. Eichenbaum: I’m still trying to best assess how these data fit into the overall fabric of data we have on GA. Similar to the above discussion about whether one complement inhibitor is better than the other, it’s difficult to conclude anything with confidence if we don’t have a modern study comparing AREDS vitamins to complement inhibition therapy. Of course, there’s the possibility that AREDS vitamins and complement inhibition therapy could work in tandem with each other, removing the either/or framework we often use with other treatments.
Dr. Ali: Let’s not forget that many of our GA patients are on fixed incomes, and AREDS vitamins are expensive. To that end, I limit my suggestion of AREDS vitamins only to those patients who fit strict intermediate AMD criteria.
Dr. Chang: I expect progress in GA therapy in the near future, as the pipeline is packed with investigational candidates. But for now, we need to use what’s at our disposal. These drugs may buy patients with GA more time with their vision. For some, that means a few more years of driving; for others, it may mean they have a bridge therapy that they can use until a more effective treatment arrives. The future is bright, but we can’t overlook the tools we have now because we’re distracted by the possibility of what might be.
FURTHER DISCUSSION: COMPARING COMPLEMENT INHIBITOR THERAPIES, ASSESSING ADOPTION HESITANCY, AND THE (POSSIBLE) ROLE OF AREDS VITAMINS FOR GA TREATMENT

1. Second quarter 2024 financial results conference call [presentation]. Apellis. 2024. Available at: https://investors.apellis.com/static-files/bcbe4eff-ecb5-4029-a924-e8ddcebb468a. Accessed September 3, 2025.
2. Keenan TDL, Agrón E, Keane PA, Domalpally A, Chew EY; AREDS and AREDS2 Research Groups. Oral antioxidant and lutein/zeaxanthin supplements slow geographic atrophy progression to the fovea in agerelated macular degeneration. Ophthalmology. 2025;132(1):1429.




