Anti-VEGF intravitreal injection is the current standard of care for the treatment of wet AMD. Although many patients experience dramatic visual and anatomic improvements with this treatment, some eyes may show a poor response. Recently, anti-VEGF resistance has been shown to be more commonly associated with a subtype of AMD, polypoidal choroidal vasculopathy (PCV).1-4
PCV is characterized by dilated polyp-shaped vascular lesions within the choroidal neovascular network, often associated with a branching vascular network.5,6 This subretinal neovascularization is usually anatomically located between Bruch membrane and the retinal pigment epithelium (RPE; type 1) but can be above the RPE (type 2).7,8
The diagnosis of PCV has become important in the management of wet AMD because it can predict anti-VEGF resistance. Further, eyes with PCV may exhibit a better visual response with less frequent anti-VEGF treatments combined with verteporfin photodynamic therapy (PDT; Visudyne, Bausch + Lomb).9,10
Although the current standard for the diagnosis of PCV is indocyanine green angiography (ICGA),5,6,11 it is often not available or not ordered in the routine evaluation of wet AMD. OCT is, however, readily available, making it another useful tool in this clinical scenario. The most characteristic signs of PCV on B-scan OCT are its polypoidal lesions and dilated aneurysm-shaped lesions. These appear as inverted-U-shaped elevations of the RPE with heterogeneous reflectivity (Figure 1). Because these lesions can be seen on OCT and are characteristic of PCV, B-scan OCT can be useful for diagnosing PCV in most clinical settings.

Figure 1. ICGA shows large polypoidal lesions of PCV that correlate point-to-point on B-scan OCT with the inverted-U-shaped elevation of the RPE. Note the heterogeneous reflectivity of the polypoidal lesion on OCT and the serous detachment.
B-SCAN CHARACTERISTICS
Kokame and colleagues evaluated the possibility of making a diagnosis of PCV based on OCT B-scan alone in eyes confirmed to have PCV based on ICGA.12 The polypoidal lesions were confirmed with point-to-point localization to the lesions on the ICG angiogram. B-scan OCT showed visible polypoidal lesions in 56.5% of eyes with PCV (Figure 1). The specificity was 97.7%, the positive predictive value was 97.5%, and the negative predictive value was 58.3%.
This study showed that, even without more specific testing or dye imaging, PCV can be identified on OCT alone in more than half of cases with high specificity. However, the low negative predictive value implies that the absence of the inverted-U-shaped lesion does not necessarily rule out PCV. Often, PCV is suspected only after a poor response to anti-VEGF therapy.
This study further showed that after 6 to 9 months of anti-VEGF therapy the presence of polypoidal lesions on B-scan OCT decreased from 56.5% of eyes to 24.6%.12 This finding indicates that, when PCV is suspected, it is important to look at the baseline OCT before anti-VEGF treatment to have the best chance of diagnosing PCV on B-scan OCT.
Another characteristic finding on OCT that can help to identify PCV is a higher prevalence of subretinal fluid in eyes with PCV than in eyes with typical wet AMD. There was no difference regarding macular edema, subretinal hyperreflective material, or RPE detachment.
Members of the Asia Pacific Ocular Imaging Society also looked at non-ICGA characteristics of PCV and found that OCT and color fundus photographs can help differentiate PCV from typical wet AMD in eyes with persistent disease activity.13 In addition, adjunctive PDT treatment could be chosen with OCT guidance, without the need for ICGA, to allow complete coverage of the polypoidal lesions.
TREATMENT CONSIDERATIONS
Identifying PCV can significantly affect patient care, as the diagnosis suggests the possibility of anti-VEGF resistance and a poor response to anti-VEGF injections. Based on the EVEREST II study,9,10 which found that combined PDT and ranibizumab (Lucentis, Genentech) improved vision with fewer injections than ranibizumab monotherapy, alternative primary therapy can be considered.
For eyes with PCV, the target of PDT is based on the location of polypoidal neovascular lesions on ICGA. The treatment spot is limited to a diameter matching the greatest linear dimension of the polypoidal neovascular lesion or a localized 300 µm border around the lesion (Figure 2). The combination of PDT and anti-VEGF injection can markedly decrease the need for injections with good visual outcomes and can result in a significant benefit in eyes demonstrating anti-VEGF resistance (Figure 3).
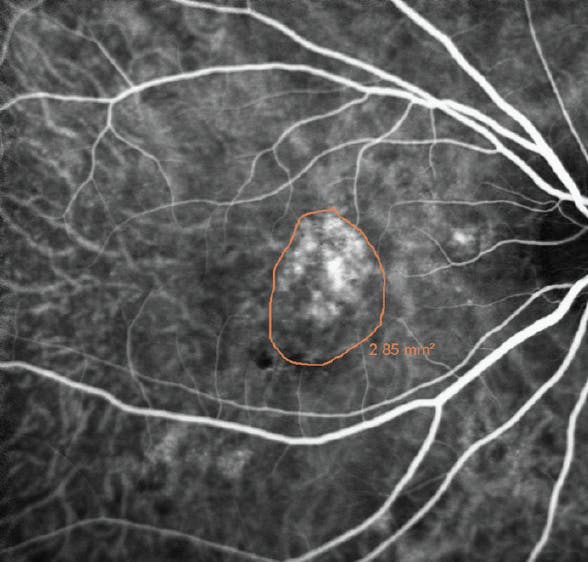
Figure 2. ICGA shows polypoidal vascular complex with the target (orange outline) around the lesion. The PDT beam is set at the greatest linear dimension of the lesion or 300 µm larger.
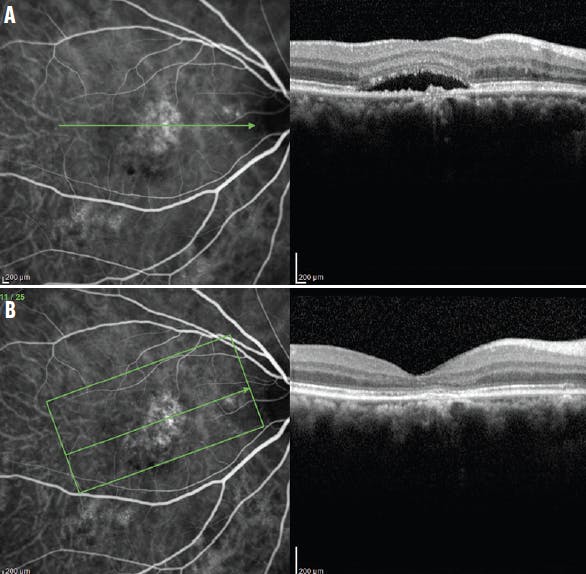
Figure 3. This patient with wet AMD had recurrent subretinal fluid and VA of 20/50 despite 27 ranibizumab injections administered once every 5 weeks, indicating anti-VEGF resistance. ICGA correlated with B-scan OCT showed significant serous retinal detachment (A). ICGA after combined reduced-fluence PDT and intravitreal bevacizumab showed a persistent branching vascular network but decreased vascular complex, with correlated B-scan OCT showing resolution of the subretinal fluid (B). The subretinal fluid remained resolved without treatment for 30 months with VA of 20/40.
Furthermore, evidence suggests that PCV can have a differential anatomic response depending on the anti-VEGF agent chosen. Aflibercept (Eylea, Regeneron) is the treatment of choice in Asia for PCV, as it has shown a significantly better response in some eyes treated previously with other anti-VEGF agents.14,15 The HAWK study, which included eyes with PCV, found evidence that brolucizumab (Beovu, Novartis) had a better drying effect than aflibercept in those eyes.16 Thus, the anti-VEGF agents that elicit the most significant anatomic response in the PCV subtype of wet AMD are brolucizumab, followed by aflibercept, then ranibizumab, and then bevacizumab (Avastin, Genentech). The recently reported risk of uveitis and vasculitis with brolucizumab must be considered in the selection of therapy.
FINAL THOUGHTS
Most ophthalmologists may assume PCV is mainly a disease seen in Asian patients, where a prevalence as high as 50% has been noted in eyes presenting with exudation and bleeding from subretinal neovascularization.17 However, more recent data regarding PCV have shown that it is a common form of wet AMD in Black patients;18 PCV is also much more common in White patients than previously thought with a prevalence of up to 25% of wet AMD actually recognized as PCV.1,19-21 Early studies in White patients with AMD used ICGA fundus camera images, which are less sensitive at detecting PCV than ICGA with the scanning laser ophthalmoscope.22 Thus, PCV is commonly seen in most practices but is not recognized due to lack of appropriate testing such as ICGA. Other more commonly available testing such as OCT B-scan images can also provide a means of diagnosing PCV, even if ICGA is not available.
The use of OCT B-scan imaging can facilitate more widespread diagnosis of PCV and can significantly affect the management approach to include the choice of anti-VEGF agent or the initiation of combination therapy with PDT and intravitreal anti-VEGF injections, which may lead to potential visual benefits and decreased treatment for PCV eyes.
1. Kokame GT, de Carlo TE, Kaneko KN, Omizo J, Lian R. Anti-vascular endothelial growth factor resistance in exudative macular degeneration and polypoidal choroidal vasculopathy. Ophthalmol Retina. 2019;3(9):744-752.
2. Stangos AN, Gandhi JS, Nair-Sahni J, Heimann H, Pournaras CJ, Harding SP. Polypoidal choroidal vasculopathy masquerading as neovascular age-related macular degeneration refractory to ranibizumab. Am J Ophthalmol. 2010;150(5):666-673.
3. Cho M, Barbazetto IA, Freund KB. Refractory neovascular age-related macular degeneration secondary to polypoidal choroidal vasculopathy. Am J Ophthalmol. 2009;148(1):70-81.
4. Hatz K, Prunte C. Polypoidal choroidal vasculopathy in Caucasian patients with presumed neovascular age-related macular degeneration and poor ranibizumab response. Br J Ophthalmol. 2014;98(2):188-194.
5. Khan S, Engelbert M, Imamura Y, Freund KB. Polypoidal choroidal vasculopathy. Simultaneous indocyanine green angiography and eye-tracked spectral domain optical coherence tomography findings. Retina. 2012;32(6):1057-1068.
6. Kokame GT. Prospective evaluation of subretinal location in polypoidal choroidal vasculopathy (PCV) and response of exudative and hemorrhagic PCV to high dose anti-angiogenic therapy (An American Ophthalmological Society Thesis). Trans Am Ophthalmol Soc. 2014;112:74-93.
7. Liang S, Shi X, Rosenfeld PJ, Li X. Type 2 choroidal neovascularization in polypoidal choroidal vasculopathy. Br J Ophthalmol. 2018;102:1570-1574.
8. Sia DI, Ebneter A, Sinkar S, Gilhotra J. Polypoidal choroidal vasculopathy: naked polyp. Int Ophthalmol. 2013;33:67-69.
9. Kokame GT, Kim JE. Treatment for a subtype of exudative macular degeneration: another mountain climbed. JAMA Ophthalmol. 2020;138:942-944.
10. Lim TH, Lai TYY, Takahashi K, et al; EVEREST II Study Group. 24-Month results comparing ranibizumab with or without verteporfin photodynamic therapy for polypoidal choroidal vasculopathy: EVEREST II randomized clinical trial. JAMA Ophthalmol. 2020;138:935-942.
11. Kokame GT. Polypoidal choroidal vasculopathy--an important diagnosis to make with therapeutic implications. Retina. 2012;32(8):1446-1448.
12. Kokame GT, Omizo JN, Kokame KA, Yamane ML. Differentiating exudative macular degeneration and polypoidal choroidal vasculopathy using optical coherence tomography B-scan. Preprint. Published online May 18, 2021. Ophthalmol Retina.
13. Chong Teo KY, Sadda SR, Gemmy Cheung CM, et al. Non-ICGA treatment criteria for suboptimal anti-VEGF response for polypoidal choroidal vasculopathy: APOIS PCV Workgroup Report 2. Preprint. Published online April 15, 2021. Ophthalmol Retina.
14. Kokame GT, Lai JC, Wee R, et al. Prospective clinical trial of Intravitreal aflibercept treatment for polypoidal choroidal vasculopathy with hemorrhage or exudation (EPIC study): 6 month results. BMC Ophthalmol. 2016;16:127.
15. Lee WK, Iida T, Ogura Y, et al. Efficacy and safety of intravitreal aflibercept for polypoidal choroidal vasculopathy in the PLANET study: a randomized clinical trial. JAMA Ophthalmol. 2018;136(7):786-793.
16. Ogura Y, Jaffe GJ, Cheung CM, et al. Efficacy and safety of brolucizumab versus aflibercept in eyes with polypoidal choroidal vasculopathy in Japanese participants of HAWK study. Preprint. Published online July 22, 2021. Br J Ophthalmol.
17. Gomi F, Tano Y. Polypoidal choroidal vasculopathy and treatments. Curr Opin Ophthalmol. 2008;19(3):208-212.
18. Marcus DM, Singh H, Fechter CM, Chamberlain DP. High-dose ranibizumab monotherapy for neovascular polypoidal choroidal vasculopathy in a predominantly non-Asian population. Eye (Lond). 2015;29(11):1427-37.
19. Lafaut BA, Leys AM, Snyers B, Rasquin F, De Laey JJ. Polypoidal choroidal vasculopathy in Caucasians. Graefes Arch Clin Exp Ophthalmol. 2000;238(9):752-759.
20. Mettu PS, Allingham MJ, Nicholas PC, Cousins SW. Neovascular morphology by ICG angiography and response to loading-dose anti-VEGF therapy in patients with neovascular AMD: Results of the PERSIST study. Invest Ophthalmol Vis Sci. 2016;57(12).
21. Pereira FB, Veloso CE, Kokame GT, Nehemy MB. Characteristics of neovascular age-related macular degeneration in Brazilian patients. Ophthalmologica. 2015;234(4):233-242.
22. Cheung CM, Lai TY, Chen SJ, et al. Understanding indocyanine green angiography in polypoidal choroidal vasculopathy: the group experience with digital fundus photography and scanning laser ophthalmoscopy. Retina. 2014;34:2397-2406.






-1_1638377570.jpg?auto=compress,format&w=70)








_1773249222.png?auto=compress,format&w=75)










