AT A GLANCE
- Several companies are investigating various therapies to improve visual acuity for patients with macular degeneration.
- Surgical implantation with a miniature telescope or subretinal implant could improve patients’ vision.
- Novel therapeutics are harnessing the power of retinal pigment epithelium cell therapy with human embryonic stem cells.
The approval of pegcetacoplan intravitreal injection (Syfovre, Apellis Pharmaceuticals) has—at long last—offered retina specialists and their patients a therapy to slow the progression of geographic atrophy (GA). Given that no prior treatment existed for GA, this new arrival has the potential to revolutionize care.
But what if we could do more than slow disease progression? Quality of life and vision are what patients value most in their day-to-day activities, and many have difficulty adhering to a treatment regimen that can only prevent an already advanced problem from getting worse. This article reviews several devices and therapies currently in the pipeline that are aimed at improving vision.
SURGICAL INTERVENTIONS
Subretinal Implant
The Prima System (Pixium Vision; Figure 1) is a subretinal implant designed to provide a photovoltaic substitute for photoreceptors. Once implanted, the device electrically stimulates the residual inner retinal neurons to elicit functional prosthetic (or bionic) central vision in combination with remaining natural peripheral vision.1
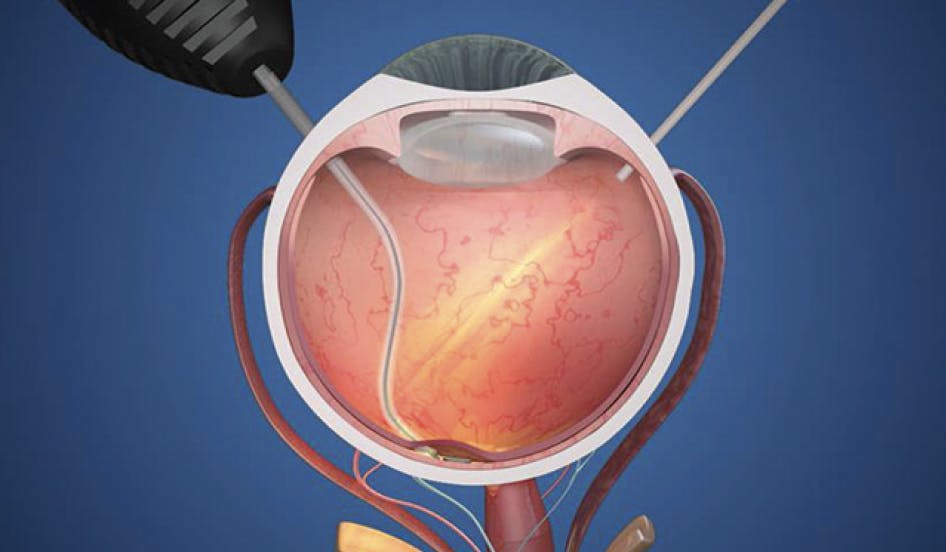
Figure 1. Diagram depicting the implantation of the Prima System into the eye.
Image courtesy of Samsara Vision
A team of investigators in France published a study in the Journal of Neural Engineering involving three patients to evaluate the effects of this device on neurosensory macular structures;2 they used spectral-domain OCT to measure changes in retinal thickness (RT) and the distance between the electrodes and the retinal inner nuclear layer (INL). Although the total RT above the implant decreased (average 39 µm ± 12 µm) during a 3-month postoperative period, no significant changes were observed beyond that point up to 36 months follow-up.2 Moreover, no structural or functional adverse events were observed, and the distance between the INL and the implant remained stable up to 36 months.
Following these preliminary findings, the European-based PRIMAvera clinical trial completed surgeries on 38 study participants as of December 2022.1 The primary endpoint is change in VA of 0.2 logMAR or more from baseline after 1 year. The study is also evaluating device- and procedure-related adverse events with up to 3 years of follow-up.
The FDA granted breakthrough device designation to the Prima System in March to expedite its development, assessment, and review.1 A readout of data from the PRIMAvera study to verify safety and efficacy is expected later this year.
SING-IMT
The smaller-incision new-generation implantable miniature telescope (SING-IMT, Samsara Vision; Figure 2) is designed to help patients regain a portion of the central vision that is lost due to late-stage AMD.3 It works by magnifying images viewed directly ahead of the patient by 2.7 times and projecting these images onto undamaged portions of the macula.3 The device is implanted during an outpatient cataract surgery that involves only a small corneal incision ranging in size from 6.5 mm to 7.5 mm.

Figure 2. The SING-IMT implant used in the feasibility trial was 2 mm x 2 mm in size and 30 µm thick.
Images courtesy of Samsara Vision
The CONCERTO trial to evaluate the safety and efficacy of the implant for patients with late-stage AMD completed its first procedures in the United States last year. The study will enroll a total of 100 patients 65 years of age or older with nonactive neovascularization and bilateral central scotomas and those with fovea-involving GA or disciform scar. Each patient will receive the implant in one eye. In addition, patients will be required to undergo postoperative rehabilitation and training to strengthen their vision.
The first surgeries of the study have gone smoothly,4 with plans to continue working with the FDA toward gaining approval to market SING-IMT in the United States.
RPE REGENERATION
CPCB-RPE1
A type of implant that uses retinal pigment epithelium (RPE) cell therapy, the California Project to Cure Blindness Retinal Pigment Epithelium 1 (CPCB-RPE1, Regenerative Patch Technologies; Figure 3), is also in development to attempt to restore in vision lost from macular degeneration.5 The implant is composed of an investigational human embryonic stem cell (hESC)-derived RPE monolayer that is seeded on a synthetic substrate.

Figure 3. This fundus photograph shows the CPCB-RPE1 implant in the eye.
Image courtesy of Regenerative Patch Technologies
The phase 1/2a feasibility study enrolled 16 participants, split between two cohorts (median age 78 years, range 69–85 years), who underwent implantation in their worse seeing eye (VA of 20/200) with CPCB-RPE1. The implant is 3.5 mm x 6.25 mm in size, and the study procedure was completed using 23-gauge vitrectomy, custom surgical forceps, and an operating microscope with or without intraoperative OCT. Low-dose tacrolimus was prescribed during the 68-day period leading up to the procedure to achieve immunosuppression.
A 1-year follow-up report showed positive preliminary safety and tolerability data, with no unexpected serious adverse events. Four participants in cohort 1 experienced serious ocular adverse events, including retinal hemorrhage, edema, focal retinal detachment, and RPE detachment.6
Noninvasive Low-Vision Tools
In addition to treatments for improving vision, certain devices can help to further optimize vision and enhance quality of life. Below are a few low vision products for patients with central vision loss.
Eyedaptic eye wear offers a line of glasses (the Eye3, Eye4, and Eye5)1 that uses built-in software to optimize peripheral vision for patients with AMD or diabetic retinopathy to help make everyday tasks, such as reading, easier to perform.
The eSight vision enhancement platform offers another set of low vision glasses. The latest generation, eSight 4, uses a small, high-speed, high-definition camera to capture what the wearer is viewing and presents these images on the monitors in the glasses in real-time so that the brain can synthesize the images and help wearers achieve enhanced visual acuity.2 The eSight 4 eye wear is registered with the FDA, and the company offers a telehealth program for new users to set up and try out the device at home.3 In partnership with Marathon Medical Corp, eSight is working to provide the United States Department of Veteran Affairs with streamlined purchasing and contract processes and enhanced product choices.4
1. Eyedaptic in your clinic & practice. Eyedaptic. Accessed April 10, 2023. eyedaptic.com/eyecare-professionals/
2. Life-changing device for people with low vision. eSight. Accessed April 11, 2023. www.esighteyewear.com
3. eSight assistive device for visually impaired available for demonstrations at AAO. Eyewire+. September 9, 2022. Accessed April 11, 2023. eyewire.news/news/esight-assistive-device-for-visually-impaired-available-for-demonstrations-at-aao
4. eSight and Marathon Medical Partner to provide sight medical devices to the US Department of Veterans Affairs. Eyewire+. August 23, 2022. Accessed April 11, 2023. eyewire.news/news/esight-and-marathon-medical-partner-to-provide-sight-medical-devices-to-the-us-department-of-veterans-affairs
The study was not designed to evaluate efficacy, but the researchers reported that four treated eyes experienced an increase of BCVA of > 5 letters (range 6–13 letters). Although not statistically significant, a larger proportion of treated eyes experience BVCA gains compared with untreated eyes.6
RG6501
RG6501 (OpRegen) is an RPE cell transplant therapy (Figure 4) being developed by Lineage Cell Therapeutics in collaboration with Genentech and Roche. RG6501 is administered via a single subretinal injection and works to slow, stop, and potentially reverse RPE cell loss in areas of the GA lesions. Like CPCB-RPE1, it is composed of RPE cells derived from hESC.7

Figure 4. Effects of treatment with RG6501 shown on OCT, including reappearnce of the outer nuclear layer and external limiting membrane, greater hyperreflectivity visible at the retinal pigment epithelium/Bruch membrane, and weaker choroidal hypertransmission. Presented at the Association for Research in Vision and Opthalmology.
Image courtesy of OpRegen
Positive safety and tolerability data up to 1 year from the phase 1/2a clinical trial showed that the majority of adverse events associated with this therapy were mild (87% in cohorts one to three, 93% in cohort 4).8 The most common were related to the surgical procedure, including conjunctival hemorrhage/hyperemia (n = 17) and epiretinal membrane (n = 16). No cases of rejection, acute or delayed intraocular inflammation, or sustained IOP increases following subretinal delivery of RP6501 have been reported.8
Preliminary efficacy data showed that participants in cohort four (n = 12) experienced improvement in visual acuity from baseline, with an average gain of 7.6 letters at 1 year; three patients from cohort four experienced VA gains of 15 letters or greater at 1 year.8 Five patients for whom RG6501 was delivered to most or all of the GA area, including the fovea, experienced an average gain of 12.8 letters, with improvements in retinal structure evidenced on spectral-domain OCT.
Enrollment in a phase 2a clinical trial of RG6501 is ongoing (NCT05626114).7,9
THE FUTURE IS BRIGHT
At end of the day, improving vision and quality of life is a top priority for physicians and their patients. These therapies are still in early stages of development, but we have much to look forward to in the future of therapies for macular degeneration.
1. Pixium Vision announces FDA breakthrough device designation for the Prima System in dry AMD [press release]. Pixium Vision. March 31, 2023. Accessed April 6, 2023. www.pixium-vision.com/2023/03/pixium-vision-announces-fda-breakthrough-device-designation-for-the-prima-system-in-dry-amd
2. Muqit MMK, Mer YL, Holz FG, Sahel JA. Long-term observations of macular thickness after subretinal implantation of a photovoltaic prosthesis in patients with atrophic age-related macular degeneration. J Neural Eng. 2022;19(5):10.1088/1741-2552/ac9645.
3. Advantages of SING IMT. Samara Vision. Accessed April 5, 2023. singimt.samsaravision.com/en/sing-imt
4. Samsara Vision announces first US surgeries of the SING IMT (smaller-incision new-generation implantable miniature telescope), for age-related macular degeneration as part of the CONCERTO study [press release]. July 11, 2022. Accessed April 6, 2023. www.samsaravision.com/press-releases/detail/70/samsara-vision-announces-first-u-s-surgeries-of-the-sing
5. Kashani AH, Uang J, Mert M, et al. Surgical method for implantation of a biosynthetic retinal pigment epithelium monolayer for geographic atrophy: experience from a phase 1/2a study. Ophthalmol Retina. 2020;4(3):264-273.
6. Kashani A, Lebkowski JS, Rahhal FM, et al. One-year follow-up in a phase 1/2a clinical trial of an allogeneic RPE cell bioengineered implant for advanced dry age-related macular degeneration. Transl Vis Sci Technol. 2021;10(10):13.
7. Safety and efficacy study of OpRegen for treatment of advanced dry-form age-related macular degeneration. Clinicaltrials.gov. Updated March 6, 2023. Accessed April 10, 2023. clinicaltrials.gov/ct2/show/NCT02286089
8. RG6501 (OpRegen) phase 1/2a clinical results support the potential for OpRegen to slow, stop or reverse disease progression in geographic atrophy secondary to age-related macular degeneration [press release]. Lineage Cell Therapeutics. May 3, 2022. Accessed April 10, 2023. investor.lineagecell.com/news-releases/news-release-details/rg6501-opregenr-phase-12a-clinical-results-support-potential
9. RG6501 (OpRegen) phase 1/2a clinical results to be presented at 2023 retinal cell and gene therapy innovation summit. Eyewire+. March 20, 2023. Accessed April 10, 2023. eyewire.news/news/rg6501-opregen-phase-12a-clinical-results-to-be-presented-at-2023-retinal-cell-and-gene-therapy-innovation-summit












_1773249222.png?auto=compress,format&w=75)










