AT A GLANCE
- OCT can identify and quantify anatomic changes within the retina and abnormal retinal fluids.
- Intraretinal and subretinal fluid volumes on OCT at different points should be considered for retreatment decisions in wet AMD.
- Artificial intelligence-based software may one day reliably and accurately measure fluid activity on OCT volume scans in real-time to promote personalized treatment approaches.
Wet AMD is a major cause of severe vision loss in eyes with AMD, representing a significant burden to health care systems in developed countries.1 Given that wet AMD is a chronic condition, the assessment of disease activity for appropriate management is essential. Most therapeutic clinical trials define markers for wet AMD activity as reduced visual acuity (> 5 ETDR letters), new hemorrhages on fundus examination, and the presence of retinal fluid on OCT.2-4 The presence of these markers usually prompts treatment with monthly intravitreal anti-VEGF agents.5 Management strategies often include either PRN or treat and extend.5
OCT is the current standard of care for image-guided diagnosis and management of wet AMD. For example, central subfoveal thickness (CST) has been widely used as a surrogate OCT biomarker for wet AMD activity to assess the presence of fluid and inform retreatment decisions.6,7 However, evidence now suggests that CST is not an ideal marker of exudative disease activity and may not properly guide management decisions. Limitations include inaccurate fluid representation, inability to segment fluid compartments, and moderate to poor correlation with BCVA.8,9
Therefore, researchers are working to identify other parameters more relevant to visual outcomes, disease activity, and prognosis to improve patient care.9
NEW APPROACHES
Advanced multimodal imaging techniques and OCT devices have provided new perspectives on the characterization of qualitative and quantitative features in acute and degenerative stages of wet AMD. OCT allows the precise identification and quantification of small anatomic changes within the retina and abnormal retinal fluids, which highlight the presence of pathological macular neovascularization (MNV) in wet AMD. Using OCT, clinicians and researchers can generally distinguish intraretinal fluid (IRF), subretinal fluid (SRF), and pigment epithelium detachment (PED).9
Post-hoc analyses of clinical trials and real-life studies have investigated these new OCT findings and suggest that the amount and location of fluid have a distinct effect on functional outcomes.10-14 For example, eyes with IRF are likely to exhibit worse visual outcomes, regardless of location and severity, while eyes with SRF are associated with a comparably more benign disease course.9,15 PED is often considered unresponsive to therapy and is associated with visual decline during PRN regimens.9,15
Therefore, OCT-based analysis that includes macular volumes instead of thicknesses, characterizes fluid subtypes, and differentiates fluid from neural tissue volumes can provide novel insights into how fluid affects visual outcomes in wet AMD.9,16-18 Such an analysis can also reflect the fluid resolution during different treatment regimens and with varying drug molecules. IRF and SRF volumes at different time points should be considered for retreatment decisions in wet AMD, together with other OCT findings.
In the CATT and HARBOR trials, the OCT-based outcome measure was change in CST, while the fluid subtypes from MNV were included in binary terms of present or absent in the PRN retreatment regimen.18-20 The HAWK and HARRIER trials evaluated the presence of IRF, SRF, and subretinal pigment epithelium fluid as qualitative parameters.21
However, treatment decisions do not always match reading center fluid determinations based on OCT scans, and artificial intelligence (AI)-based studies show that retina specialists have imperfect accuracy and low sensitivity in detecting retinal fluid.20,22 Qualitative assessment of macular fluids by manual OCT interpretation—usually graded as present or absent and mild or severe—is time-consuming, difficult to standardize, and not feasible in the routine clinic. Considering the growing number of OCT scans necessary for optimal wet AMD management and the increasing OCT volume scan density, accurate fluid assessment may create a significant gap between optimal patient care and efficient workflow in routine clinical practice.23
Artificial Intelligence in the Real World
RetInSight’s Fluid Monitor is a first-in-class artificial intelligence-based class IIa medical device recently developed in Europe to help clinicians make treatment decisions and monitor patients with wet AMD. It is compatible with all leading OCT devices, starting from the Heidelberg Engineering ophthalmic imaging platforms and applications (Figure).
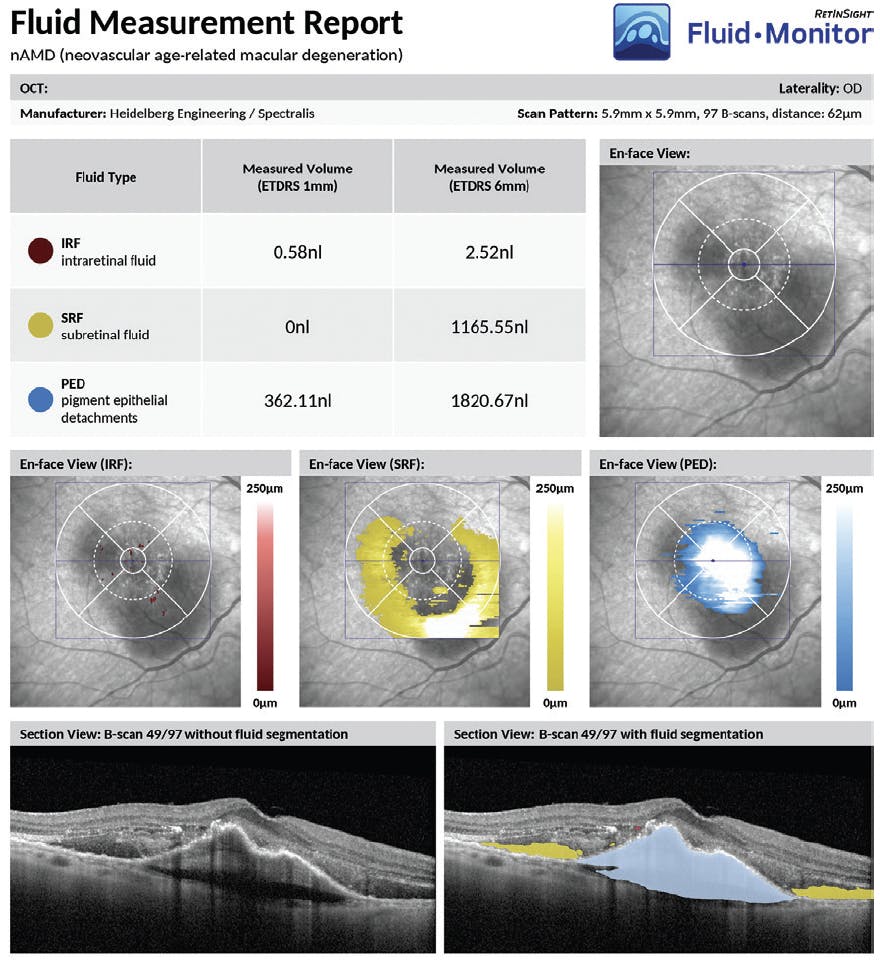
Figure. The RetInSight Fluid Monitor can provide real-time reports during routine OCT imaging and can identify intraretinal fluid, subretinal fluid, and pigment epithelial detachment, as well as the fluid’s distribution, quantity in nanoliters, and location within the central 1-mm and 6-mm macular area.
THE POWER OF AI
AI-based segmentation can automatically process OCT scans and accurately identify, localize, and quantify macular fluid.23-26 AI has proven superior to human experts in terms of the accuracy and speed of fluid analyses in both clinical trial and real-world settings.23,26-28 As such, quantitative metrics of IRF, SRF, and PED within OCT volume scans can be used as scalable biomarkers in wet AMD, providing substantial advantages to research and clinical practice.23,26
Automated segmentation of OCT volumes should greatly improve our understanding of the effect macular fluid has on visual outcomes. It may also offer the opportunity to extract other relevant biomarkers, such as macular atrophy, subretinal hyperreflective material, and fibrosis, to name a few.26,29 AI-based tools may unveil novel structural-functional correlations in wet AMD, affecting patients’ visual prognoses. In the clinic, it may provide a “fluid activity meter” to optimize and personalize wet AMD therapy.26
Thus, AI-based software may one day reliably and accurately measure fluid activity on OCT volume scans in real-time to promote personalized treatment approaches. This accurate and user-friendly approach may be able to improve clinical workflow at multiple levels. Patients could benefit from shorter wait times, lower risk of over- or under-treatment, and minimized travel costs and absence from work. For hospital and clinic administrators, it may offer improved resource allocation and enhanced patient access.
FINAL PEARLS
The integrated analysis of functional and structural features allowed by AI-based approaches is a prerequisite to quantify disease progression, improve the therapeutic management of wet AMD, and refine treatment regimens in both real-world practice and clinical trials. Automated algorithms and computational data analysis help extract a wealth of quantitative data from high-resolution OCT imaging, while also reducing the burden of data analysis.
This could ultimately bridge the gap between clinical trial results and real-world outcomes and bring clinical trial quality disease management to routine clinical care.30 Further research is needed to assess the effect of automated quantification of fluid in wet AMD in the real-world setting. Continuous innovation, driven by advanced imaging devices, AI-based applications, and novel treatments, is shaping the field of medical retina to improve visual outcomes for patients and reduce the burden of care.
1. Jager RD, Mieler WF, Miller JW. Age-related macular degeneration. N Engl J Med. 2008;358(24):2606-2617.
2. Martin DF, Martin DF, Maguire MG, et al. Ranibizumab and bevacizumab for treatment of neovascular age-related macular degeneration: two-year results. Ophthalmology. 2012;119(7):1388-1398.
3. Kodjikian L, Souied EH, Mimoun G, et al. Ranibizumab versus bevacizumab for neovascular age-related macular degeneration: results from the GEFAL noninferiority randomized trial. Ophthalmology. 2013;120(11):2300-2309.
4. Chakravarthy U, Chakravarthy U, Harding SP, et al. Ranibizumab versus bevacizumab to treat neovascular age-related macular degeneration: one-year findings from the IVAN randomized trial. Ophthalmology. 2012;119(7):1399-1411.
5. Schmidt-Erfurth U, Chong V, Loewenstein A, et al. Guidelines for the management of neovascular age-related macular degeneration by the European Society of Retina Specialists (EURETINA). Br J Ophthalmol. 2014;98(9):1144-1167.
6. CATT Research Group, Martin DF, Maguire MG, et al. Ranibizumab and bevacizumab for neovascular age-related macular degeneration. N Engl J Med. 2011;364(20):1897-1908.
7. Martin DF, Maguire MG, Fine SL, et al. Ranibizumab and bevacizumab for treatment of neovascular age-related macular degeneration: two-year results. Ophthalmology. 2012;119(7):1388-1398.
8. Sharma S, Toth CA, Daniel E, et al. Macular morphology and visual acuity in the second year of the Comparison of Age-Related Macular Degeneration Treatments Trials. Ophthalmology. 2016;123(4):865-875.
9. Schmidt-Erfurth U, Waldstein SM. A paradigm shift in imaging biomarkers in neovascular age-related macular degeneration. Prog Retin Eye Res. 2016;50:1-24.
10. Simader C, Ritter M, Bolz M, et al. Morphologic parameters relevant for visual outcome during anti-angiogenic therapy of neovascular age-related macular degeneration. Ophthalmology. 2014;121(6):1237-1245.
11. Waldstein SM, Simader C, Staurenghi G, et al. Morphology and visual acuity in aflibercept and ranibizumab therapy for neovascular age-related macular degeneration in the VIEW Trials. Ophthalmology. 2016;123(7):1521-1529.
12. Waldstein SM, Wright J, Warburton J, et al. Predictive value of retinal morphology for visual acuity outcomes of different ranibizumab treatment regimens for neovascular AMD. Ophthalmology. 2016;123(1):60-69.
13. Willoughby AS, Ying G-S, Toth CA, et al. Subretinal hyperreflective material in the Comparison of Age-Related Macular Degeneration Treatments Trials. Ophthalmology. 2015;122(9):1846-1853.
14. Lai T-T, Hsieh Y-T, Yang C-M, et al. Biomarkers of optical coherence tomography in evaluating the treatment outcomes of neovascular age-related macular degeneration: a real-world study. Sci Rep. 2019;9(1):529.
15. Nguyen V, Puzo M, Sanchez-Monroy J, et al. Association between anatomical and clinical outcomes of neovascular age-related macular degeneration treated with antivascular endothelial growth factor. Retina. 2021;41(7):1446-1454.
16. Schmidt-Erfurth U, Bogunovic H, Sadeghipour A, et al. Machine learning to analyze the prognostic value of current imaging biomarkers in neovascular age-related macular degeneration. Ophthalmol Retina. 2018;2(1):24-30.
17. Guymer RH, Markey CM, McAllister IL, et al. Tolerating subretinal fluid in neovascular age-related macular degeneration treated with ranibizumab using a treat-and-extend regimen: FLUID study 24-month results. Ophthalmology. 2019;126(5):723-734.
18. Waldstein SM, Philip AM, Leitner R, et al. Correlation of 3-dimensionally quantified intraretinal and subretinal fluid with visual acuity in neovascular age-related macular degeneration. JAMA Ophthalmol. 2016;134(2):182-190.
19. Busbee BG, Ho AC, Brown DM, et al. Twelve-month efficacy and safety of 0.5 mg or 2.0 mg ranibizumab in patients with subfoveal neovascular age-related macular degeneration. Ophthalmology. 2013;120(5):1046-1056.
20. Toth CA, Decroos FC, Ying G-S, et al. Identification of fluid on optical coherence tomography by treating ophthalmologists versus a reading center in the comparison of age-related macular degeneration treatments trials. Retina. 2015;35(7):1303-1314.
21. Dugel PU, Koh A, Ogura Y, et al. HAWK and HARRIER: phase 3, multicenter, randomized, double-masked trials of brolucizumab for neovascular age-related macular degeneration. Ophthalmology. 2020;127(1):72-84.
22. Keenan TD, Clemons TE, Domalpally A, et al. Retinal specialist versus artificial intelligence detection of retinal fluid from OCT. Ophthalmology. 2021;128:100-109.
23. Keenan TDL, Chakravarthy U, Loewenstein A, Chew EY, Schmidt-Erfurth U. Automated quantitative assessment of retinal fluid volumes as important biomarkers in neovascular age-related macular degeneration. Am J Ophthalmol. 2021;224:267-281.
24. Schlegl T, Waldstein SM, Bogunovic H, et al. Fully automated detection and quantification of macular fluid in OCT using deep learning. Ophthalmology. 2018;125(4):549-558.
25. De Fauw J, Ledsam JR, Romera-Paredes B, et al. Clinically applicable deep learning for diagnosis and referral in retinal disease. Nat Med. 2018;24(9):1342-1350.
26. Schmidt-Erfurth U, Reiter GS, Riedl S, et al. AI-based monitoring of retinal fluid in disease activity and under therapy. Prog Retin Eye Res. 2022;86:100972.
27. Moraes G, Fu DJ, Wilson M, et al. Quantitative analysis of OCT for neovascular age-related macular degeneration using deep learning. Ophthalmology. 2020;128(5):693-705.
28. Chakravarthy U, Havilio M, Syntosi A, et al. Impact of macular fluid volume fluctuations on visual acuity during anti-VEGF therapy in eyes with nAMD. Eye. 2021;35(11):2983-2990.
29. Roberts PK, Vogl W-D, Gerendas BS, et al. Quantification of fluid resolution and visual acuity gain in patients with diabetic macular edema using deep learning: a post hoc analysis of a randomized clinical trial. JAMA Ophthalmol. 2020;138(9):945-953.
30. Mehta H, Tufail A, Daien V, et al. Real-world outcomes in patients with neovascular age-related macular degeneration treated with intravitreal vascular endothelial growth factor inhibitors. Prog Retin Eye Res. 2018;65:127-146.












_1773249222.png?auto=compress,format&w=75)










