Angiogenesis, Exudation, and Degeneration 2024, held February 3, was another triumphant virtual meeting, boasting nearly 14 hours of clinical education. Experts from around the globe discussed the latest in retina: advances in OCT imaging, wet and dry AMD diagnosis and treatment, diabetic eye disease, and the management of retinal vascular diseases. Here, we highlight some of the key presentations.
IMAGING
The meeting began with a session on OCT during which K. Bailey Freund, MD, shared clinical pearls gleaned from high-resolution OCT. While discussing cases, he noted that deep retinal age-related microvascular anomalies can often masquerade as type 3 macular neovascularization (MNV)—but are easily identified with high-resolution OCT.
Amitha Domalpally, MD, PhD, presented on the multimodal assessment of OCT biomarkers of geographic atrophy (GA). The goal of her work was to develop a translational map to better understand how advanced imaging biomarkers manifest in typical diagnostic imaging in GA. When complete retinal pigment epithelium (RPE) and outer retinal atrophy is identified on OCT, for example, it’s visible on color fundus photography just 50% of the time. The other 50% shows fading and calcified drusen.
DRY AMD
The next session opened with a lecture on early-onset AMD by Carel B. Hoyng, MD (Figure 1). Early-onset drusen maculopathy is an early manifestation of AMD with overlying phenotypic and genotypic characteristics, he said. Genetics are a significant risk factor, with earlier age of onset, the number of affected family members, and the presence of drusen and GA posing increased risk. Genetic testing would help raise awareness in at-risk families who could then make necessary lifestyle modifications, Dr. Hoyng stressed.

Figure 1. Dr. Hoyng discussed these early-onset drusen maculopathy patients. Note that their ages are displayed in the bottom right-hand corner of each image.
Christine A. Curcio, MD, discussed the prominence of lysolipids in subretinal drusenoid deposits, which are independent risk factors for AMD progression that are potentially linked to lipid transfers specific to rods and cones. Giovanni Staurenghi, MD, shared another early OCT biomarker of AMD, basal laminar deposits. High-resolution OCT increases axial resolution, with a 3 µm optical and 1.9 µm digital resolution, he explained, allowing for easy, timely identification of basal laminar deposits (Figure 2).
Emily Y. Chew, MD, led a talk on the classification and regression tree (CART) analysis of AMD. Since the Age-Related Eye Disease Study (AREDS) nine-step classification scale was published in 2006, researchers have identified the need for an updated version to incorporate an important risk factor for progression to late AMD, reticular pseudodrusen (RPD). Analyzing data from both AREDS and AREDS2 and taking a CART approach that considers drusen size and area, pigmentation and depigmentation, and RPD, the team found that the combination of pigment changes and RPD posed the greatest risk of progression.
Also in this session, Seung-Young Yu, MD, discussed the effect of risk factors on GA progression in a Korean patient cohort. The cohort had a comparable progression rate to Caucasian populations, she said. Higher rates were associated with larger baseline lesion size, thinner ganglion complex-inner plexiform layer thickness, multifocal pattern, and female gender.
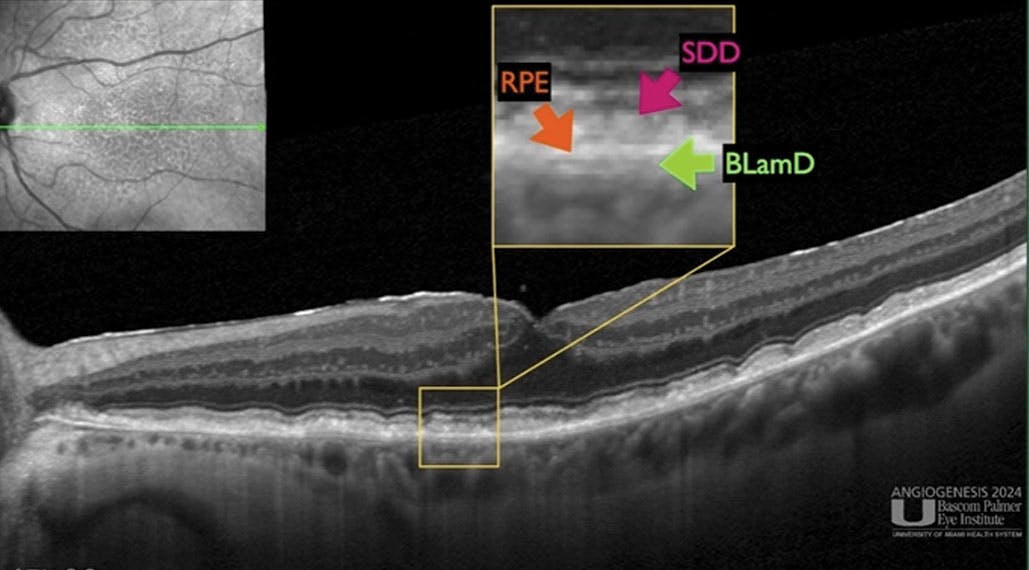
Figure 2. Dr. Staurenghi presented on the importance of basal laminar deposits in the early identification of AMD, showing attendees how to interpret the OCT to visualize the RPE, subretinal drusenoid deposits, and basal laminar deposits.
Giuseppe Querques, MD, PhD, then took over with a module on predictors of GA development in eyes with type 3 MNV treated with anti-VEGF therapy. Type 3 MNV is the second most frequent subtype of MNV in AMD and presents treatment challenges that often result in worse functional outcomes due to atrophy development and progression, he explained. The number of anti-VEGF injections negatively correlates with atrophy development, and subretinal fluid serves as an independent, negative predictor of final BCVA in type 3 MNV, he concluded.
EMERGING THERAPIES
Data from a phase 1 trial of a novel gene therapy, LX102 (Innostellar Biotherapeutic) for the treatment of wet AMD, was presented by Xiandong Sun, MD. This gene therapy works by promoting the expression of VEGF trap proteins. Safety data at 1 year (n = 18) showed that LX102 was generally well tolerated; in addition, patients experienced a durable treatment effect up to 1.5 years after one subretinal injection of LX102. Phase 2 of the trial is underway, according to Dr. Sun.
Charles C. Wykoff, MD, PhD, presented topline data from the phase 2 DAVIO 2 trial of sustained-release vorolanib (Duravyu [formerly EYP-1901], Eyepoint Pharmaceuticals) compared with aflibercept (Eylea, Regeneron) in previously treated patients with wet AMD. The drug was administered using the company’s modified Durasert E implant. The trial met its primary endpoint of noninferiority in mean change in BCVA from baseline at weeks 28 and 32 compared with aflibercept. In addition, 64% of eyes did not require supplemental injections up to week 32, according to Dr. Wykoff.
W. Lloyd Clark, MD, and Diana V. Do, MD, presented 96-week data from the PULSAR and PHOTON trials, respectively. The results of each trial demonstrated comparable safety and efficacy between 8 mg aflibercept (Eylea HD, Regeneron) and 2 mg aflibercept with fewer injections required to maintain the treatment effect. In the PULSAR trial, 78% of patients who randomly received the 8 mg dose every 16 weeks achieved ≥ 16-week dosing intervals, and 53% achieved ≥ 20-week dosing intervals. In the PHOTON trial, 88% of patients dosed with 8 mg aflibercept every 12 weeks and 84% of those dosed every 16 weeks maintained those dosing intervals or longer through 96 weeks.
John A. Wells, MD, followed these presentations with a pooled analysis of safety data from the CANDELA, PULSAR, and PHOTON trials showing comparable rates of ocular inflammation between the low- and high-dose aflibercept treatment groups. No cases of endophthalmitis or ischemic optic neuropathy were reported in the high-dose group versus two cases and one case of these adverse events, respectively, in the low-dose group.
Rishi P. Singh, MD, presented data on treatment with faricimab (Vabysmo, Genentech/Roche). The study found a significantly higher reduction in macular leakage area in patients with diabetic macular edema and retinal vein occlusion who were treated with faricimab compared with aflibercept. Dr. Singh also discussed other advantages of the dual pathway inhibition observed in the study, including greater anatomical improvements and greater reduction of hyperreflective foci and epiretinal membrane.
Similarly, data from the TRUCKEE study of real-world outcomes with faricimab, presented by Arshad M. Khanani, MD, MA, FASRS, showed that 25% of difficult-to-treat patients who switched from anti-VEGF monotherapy experienced complete resolution of intraretinal/subretinal fluid after one injection of faricimab. In addition, an almost 50 µm reduction in central subfield thickness and 60 µm reduction in RPE detachment was achieved after six injections of faricimab, he concluded.
UNTIL NEXT YEAR
The meeting concluded after a full day of education, leaving attendees with much to mull over regarding the current treatment landscape—and what’s coming through the pipeline. We look forward to next year’s meeting!


















_1773249222.png?auto=compress,format&w=75)










