AT A GLANCE
- To ensure a successful retinal detachment repair, act immediately, do as little harm as possible, properly evaluate the periphery, and treat with precision.
- Fundus autofluorescence can identify microscopic displacements of the retinal vessels after a successful retinal detachment repair operation.
- To avoid microscopic displacement of retinal vessels, leave some subretinal fluid to be aspirated naturally, allowing the retina to settle back to its original positioning.
At the end of the 20th century, during the era of 20-gauge pars plana vitrectomy (PPV) and scleral buckling to repair retinal detachment (RD), the final success rate ranged from 70% to 85%.1,2 Today, our surgical instruments—including gauge size, vitrectomy consoles, illumination systems, and visualization—have improved dramatically, and so have our single-surgery success rates, up to 95%.3 Now that we can confidently achieve anatomic success, the next goal is improving patients’ postoperative quality of vision. For example, we can now use OCT to identify complications such as cystoid macular edema (CME) and epiretinal membrane formation (ERM), both of which can compromise vision.
Regardless of the instruments we use, the principles for a successful operation remain the same: Act immediately, do as little harm as possible, properly evaluate the periphery, and treat with precision.
TREATMENT CONSIDERATIONS
We recommend draining through the existing break because it provides shorter operating time. We also avoid using diathermy, additional laser, and perfluorocarbon liquids (PFCL). Doing so provides better functional vision, reduces the risk of ERM formation or proliferative vitreoretinopathy, and lowers the cost.
In addition, surgeons can leave some subretinal fluid at the end of the operation. During scleral buckling surgery, we find the break, perform cryotherapy or laser retinopexy, and may or may not drain; sometimes, we allow the retinal pigment epithelium (RPE) pump to aspirate the fluid and flatten the retina naturally. Surgeons often feel the need to flatten the retina at the end of the operation to avoid creating retinal folds either outside of the macular area or, in rare cases, in the macula (Figures 1 and 2). However, during PPV to reattach the retina, we have found that if we do not force the retina flat at the end of the operation, we have fewer complications, such as residual PFCL bubbles under the retina, especially in the macula, which can be devastating. As another benefit, we have no additional inflammation from the diathermy and laser, which are applied to the drainage retinotomy.
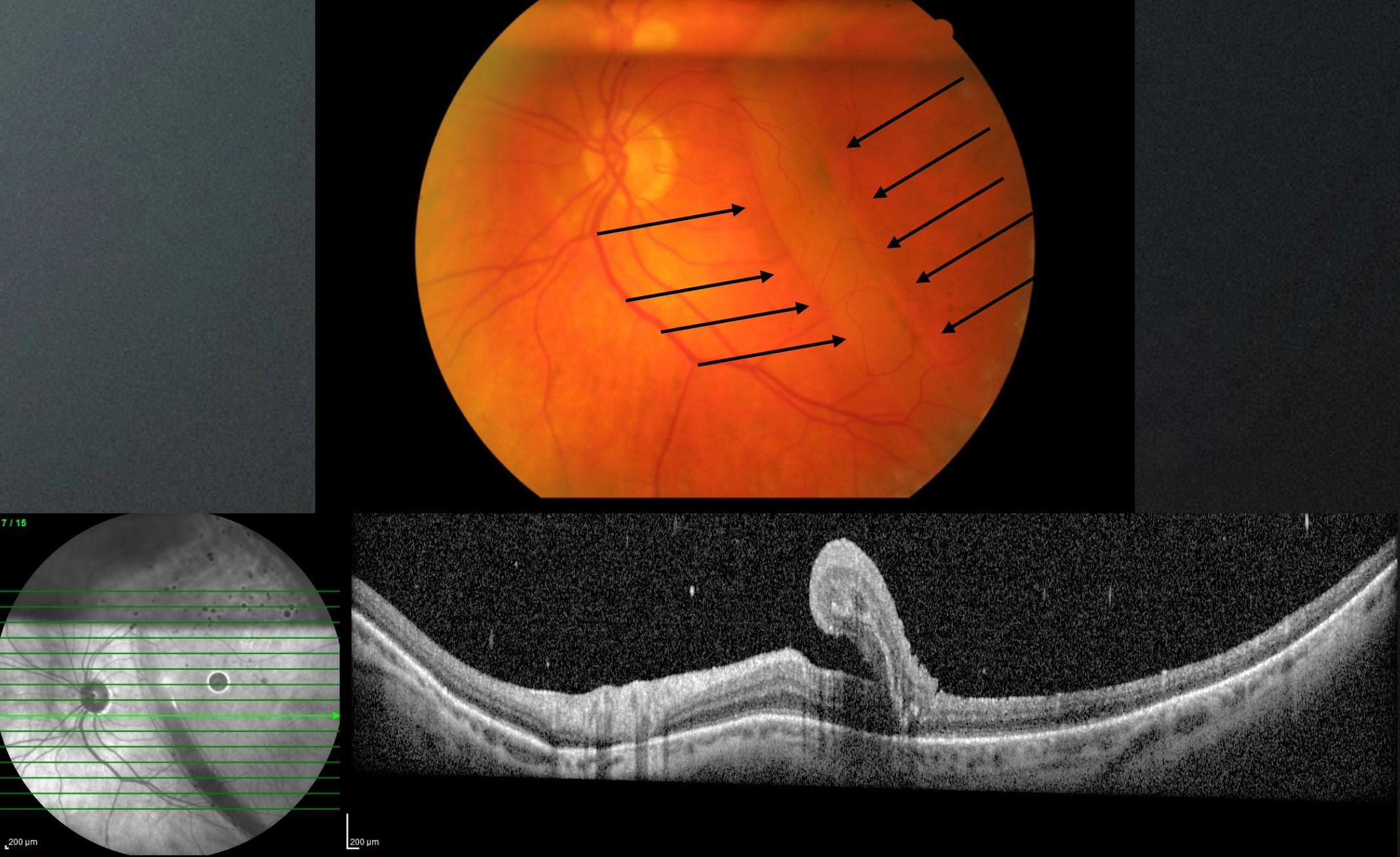
Figure 2. Fundus imaging captures a macular fold after RD repair (arrows), confirmed on OCT imaging.
OUR PREFERRED TECHNIQUE
The technique we are using in RD cases is 25-gauge PPV, after which we remove as much subretinal fluid as possible through the existing retinal break and apply laser or cryotherapy over the break. At the end, we inject 2.5 ml of pure SF6, which becomes diluted to 50% due to preexisting air in the eye. The SF6 bubble expands by 1.25 ml in 24 hours, effectively covering the space as the subretinal fluid is absorbed by the RPE pump or escapes into the vitreous cavity and, from there, the anterior chamber. Thus, in 24 hours, surgeons can expect a flat retina and a sufficient gas bubble volume that will fill the eye for the first 7 days. Postoperatively, we place the patient on the side of the break for up to 2 hours and then ask them to maintain face-down positioning for the following few days.
THE DATA
We have used this technique for more than 20 years and published the results in 2016.4 At that time, we had two groups of patients treated with either 20- or 25-gauge PPV. Both groups had equally good anatomic and functional results. Unfortunately, the number of patients limited our ability to create a quality analysis to prove our hypothesis that this approach led to fewer postoperative complications.
In 2022, other researchers published a prospective study that included three groups of 100 patients each with macula-off RD.5 In the first group of patients, the subretinal fluid was drained through the existing peripheral retinal break; in the second group, the retina was flattened with the use of PFCL; and the third group underwent a drainage retinotomy. Visual acuity testing and spectral-domain OCT were performed 3, 6, and 12 months postoperatively. Primary outcomes of the study included visual acuity and the discontinuity of the external limiting membrane, ellipsoid zone interdigitation zone, and RPE at 1 year.
The researchers found that the visual acuity was better in the group who underwent fluid drainage through the existing retinal break compared with the PFCL group.5 ERM formation was higher in the group who underwent a drainage retinotomy, and CME formation was higher in the group treated with PFCL, supporting the hypothesis that additional diathermy and laser application to the drainage retinotomy creates inflammatory factors that lead to ERM formation. In addition, the chemical component of the PFCL, although proven to be neutral, together with the turbulences of the fluid during the injection and aspiration, may promote inflammation that leads to CME formation.
These results suggest that draining the fluid through the existing break may reduce the risk of complications and provide better functional vision.
IMAGING PEARLS
Although OCT is useful for diagnosing and tracking CME and ERM formation in early stages, fundus autofluorescence (FAF) can be particularly helpful for identifying changes in the posterior pole after a successful RD repair surgery.
For example, FAF can identify microscopic displacements of the retinal vessels after a successful RD repair. The original position of the vessel before RD repair surgery appears as a hyperautofluorescent line and is known by several names in the literature, including retinal vessel printing,6 RPE vessel ghost lines, or simply ghost vessels (Figure 3).

Figure 3. The hyperautofluorescent lines on FAF imaging (arrows) are thought to represent the original position of retinal vessels prior to the RD repair, referred to as ghost vessels.
These ghost vessels may explain some of the complaints patients may have after a successful RD repair, such as distortion of image size (dysmetropsia), metamorphopsia, binocular diplopia, asthenopia, and anisoiconia.6-11
In extreme cases, we have known some affected patients to partially occlude the operated eye despite good postoperative acuity.
FOCUS ON THE VESSELS
Many studies have documented the existence of ghost vessels, surgical approaches to avoid them, and postoperative visual disturbances if they happen.7-11 Most researchers have concluded that flattening the retina at the end of the operation produces microscopic displacement of the macular area. Thus, surgeons should instead leave some subretinal fluid to be aspirated by the RPE pump, allowing the retina to settle back to its original positioning. To help with this, surgeons can position the patient postoperatively either face down immediately or on the side of the break to help prevent microscopic displacement of the retinal vessels.8-14
1. Miller DM, Riemann CD, Foster RE, Petersen MR. Comparison of 20-, 23-, and 25-gauge pars plana vitrectomy in pseudophakic rhegmatogenous retinal detachment repair. Ophthalmic Surg Lasers Imaging. 2011;42(2):107-113.
2. Heimann H, Bartz-Schmidt KU, Bornfeld N, Weiss C, Hilgers R-D, Foerster MH; Scleral Buckling versus Primary Vitrectomy in Rhegmatogenous Retinal Detachment Study Group. Scleral buckling versus primary vitrectomy in rhegmatogenous retinal detachment: a prospective randomized multicenter clinical study. Ophthalmology. 2007;114(12):2142-2154.
3. Mura M, Tan SH, De Smet MD. Use of 25-gauge vitrectomy in the management of primary rhegmatogenous retinal detachment. Retina. 2009;29(9):1299-1304.
4. Gotzaridis S, Liazos E, Petrou P, Georgalas I. 25-Gauge vitrectomy and incomplete drainage of subretinal fluid for the treatment of primary rhegmatogenous retinal detachment. Ophthalmic Surg Lasers Imaging Retina. 2016;47(4):333-335.
5. McKay BR, Bansal A, Kryshtalskyj M, Wong DT, Berger A, Muni RH. Evaluation of subretinal fluid drainage techniques during pars plana vitrectomy for primary rhegmatogenous retinal detachment-ELLIPSOID study. Am J Ophthalmol. 2022;241:227-237.
6. Dell’Omo R, Mura M, Lesnik Oberstein SY, Bijl H, Tan HS. Early simultaneous fundus autofluorescence and optical coherence tomography features after pars plana vitrectomy for primary rhegmatogenous retinal detachment. Retina. 2012;32(4):719-728.
7. Shiragami C, Shiraga F, Yamaji H, et al. Unintentional displacement of the retina after standard vitrectomy for rhegmatogenous retinal detachment. Ophthalmology. 2010;117(1):86-92.e1.
8. Lee E, Williamson TH, Hysi P, et al. Macular displacement following rhegmatogenous retinal detachment repair. Br J Ophthalmol. 2013;97(10):1297-1302.
9. Shiragami C, Fukuda K, Yamaji H, Morita M, Shiraga F. A method to decrease the frequency of unintentional slippage after vitrectomy for rhegmatogenous retinal detachment. Retina. 2015;35(4):758-763.
10. Okuda T, Higashide T, Sugiyama K. Metamorphopsia and outer retinal morphologic changes after successful vitrectomy surgery for macula-off rhegmatogenous retinal detachment. Retina. 2018;38(1):148-154.
11. Okamoto F, Sugiura Y, Okamoto Y, Hiraoka T, Oshika T. Metamorphopsia and optical coherence tomography findings after rhegmatogenous retinal detachment surgery. Am J Ophthalmol. 2014;157(1):214-220.
12. Lina G, Xuemin Q, Qinmei W, Lijun S. Vision-related quality of life, metamorphopsia, and stereopsis after successful surgery for rhegmatogenous retinal detachment. Eye. 2016;30(1):40-45.
13. Casswell EJ, Yorston D, Lee E, Charteris DG. Distortion resolution after vitrectomy for macula-involving retinal detachment repair: post hoc analysis of the PostRD trial. Retina. 2022;42(12):2315-2320.
14. Casswell EJ, Yorston D, Lee E, et al. Effect of face-down positioning vs support-the-break positioning after macula-involving retinal detachment repair: The PostRD randomized clinical trial. JAMA Ophthalmol. 2020;138(6):634-642.












_1773249222.png?auto=compress,format&w=75)










