AT A GLANCE
- In eyes with rhegmatogenous retinal detachment (RRD) that are high-risk for the development of proliferative vitreoretinopathy (PVR), strongly consider adding a scleral buckle, particularly among patients younger than 65 years of age.
- Early surgical intervention for repairing PVR RRDs can optimize visual outcomes.
- Lens removal may not be necessary in all PVR RRDs, especially in cases with an adequate view.
Proliferative vitreoretinopathy (PVR) is the leading cause of surgical failure following primary rhegmatogenous retinal detachment (RRD) repair with a reported incidence of 5% to 10%.1 PVR can be characterized as an exaggerated and maladaptive wound healing process of the retina defined by cellular proliferation, membrane formation, and intraretinal fibrosis, resulting in contraction and foreshortening of the retina and, eventually, recurrent RRD.2
PREOPERATIVE PLANNING FOR HIGH-RISK RRD
Preoperatively, it is important to identify primary RRDs with high-risk features for developing PVR, as the surgical method of repair may influence outcomes, including single surgery anatomic success (SSAS). High-risk features that have been identified include preoperative PVR grade A or B (Figure 1), vitreous hemorrhage, more extensive RRDs (ie, involving at least 50% of the retinal area), multiple breaks (ie, presence of three or more retinal breaks), choroidal detachment, prior cryotherapy, and/or more chronic RRD (ie, duration of more than 2 weeks).3
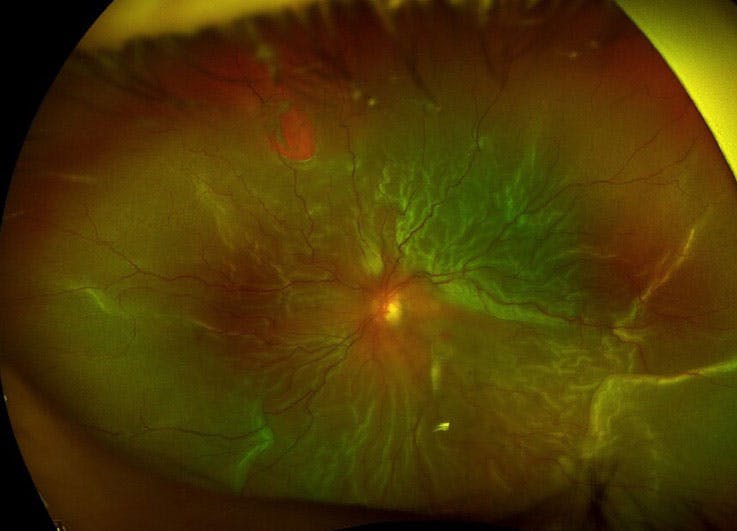
Figure 1. This phakic patient with an RRD and high-risk features would benefit from the addition of an SB.
Salabati et al evaluated 389 eyes with high-risk RRDs at their institution and found a much lower SSAS rate in these eyes at 71.5%.4 However, eyes that underwent pars plana vitrectomy (PPV) combined with scleral buckle (SB) in their study had a significantly higher SSAS rate compared with eyes that underwent PPV alone (80.8% vs 67%, respectively).4 This effect was driven primarily by patients younger than 65 years of age, where SSAS was 85.6% in the PPV/SB group versus 67.9% in the PPV group (P = .003). In patients 65 years of age or older, there was no significant difference in SSAS for PPV/SB and PPV (68.6% vs 66.1%, P = .99). Storey et al similarly found a higher surgical success rate in the PPV/SB cohort versus PPV alone among 65 eyes with high-risk features (75% vs 48.3%, respectively).5
SURGICAL TIMING
Eyes with PVR RDs are often treated differently compared with eyes with primary RRDs. Traditionally, there is less urgency to repair PVR RDs due to the perceived notion that the visual outcomes will be poor regardless, particularly in macula-off detachments. In fact, one school of thought suggests that it may be better to wait for further maturation of the membranes to help make membrane peeling easier and potentially lower the chance of future redetachments. There may also be less inflammation present if surgery is delayed, again lowering the risk of PVR recurrence.
However, in a retrospective study of 5,355 eyes with primary RRD, of which 345 developed PVR, time to surgery from the diagnosis of a recurrent RRD was the only modifiable factor that was associated with good visual acuity in eyes that had PVR RDs and underwent retinectomies.6 This was independent of whether the macula was on or off. There were significantly fewer days between recurrent RD diagnosis and time to repair in eyes achieving good VA (mean Snellen equivalent 20/42) compared with eyes with worse VA (mean Snellen equivalent 20/693), regardless of macula status (mean 2.9 days vs mean 5.8 days).6
APPROACHING PVR DETACHMENTS INTRAOPERATIVELY
The initial step when managing PVR detachments is determining whether to proceed with PPV alone or in combination with an SB. Placing an SB helps relieve anteroposterior and circumferential traction. Therefore, in most cases, an SB should be strongly considered, except in situations where a 360° retinectomy may be needed (eg, to repair a funnel-shaped RD with a pronounced anterior loop). Even if a 180° retinectomy is done, the SB still supports the remaining vitreous base and the anterior edges of the retinectomy.
The main goal of surgery in PVR cases is to reattach the retina and release all traction, both from the preretinal membranes and subretinal fibrosis. Triamcinolone can be helpful to ensure the hyaloid is up and highlight both posterior and more peripheral membranes for peeling.
During the surgery, look for star folds. Pinching with forceps while aiming for the center of the fold can often help initiate the peel because there is often a fibrous band. Alternatively, a Finesse Flex Loop (Alcon) can be used to gently massage the surface of the retina and identify additional membranes or edges from which to initiate a peel. Surgeons should consider using a chandelier light with dual Flex Loops, particularly in areas of contraction, to help identify preretinal membranes. The membranes can be very adherent in certain cases, making it difficult to peel, especially if the underlying retina is bullously detached. A lighted pick and forceps may be useful in this situation.
ICG or brilliant blue can also be used to stain the internal limiting membrane (ILM) and reveal negative staining of posterior membranes (Figure 2). In a recent multicenter international study of 370 eyes with grade C PVR, ILM peeling, and extended ILM peeling in particular, was associated with higher SSAS at 3 months (86.6% vs 73.2%, respectively) and 6 months (75.2% vs 65.3%, respectively) and significantly better visual acuity compared with eyes that did not undergo ILM peeling.7
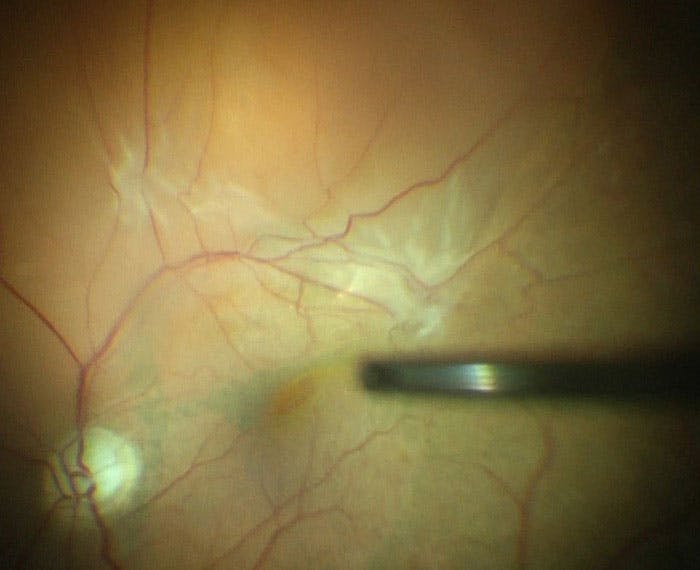
Figure 2. During surgery for a PVR RRD, surgeons can use ICG to stain the ILM and posterior PVR membranes.
All membranes should be peeled, if possible, prior to considering a retinectomy. If a retinectomy cannot be avoided, the more posterior membranes that have been removed the better so that the retinectomy can remain as anterior as possible while removing the intrinsic fibrosis (Figure 3). Diathermy should be used to demarcate the area of planned retinectomy. Careful diathermy during the retinectomy is important to achieve good hemostasis, as hemorrhage is a risk factor for PVR proliferation. A small initial PFO bubble can be used to stabilize the retina and protect the macula from any inadvertent subretinal hemorrhage prior to initiating the retinectomy (Figure 4). Surgeons should make sure this initial bubble is small enough such that tilting the eye during the retinectomy does not lead to small PFO bubbles going into the subretinal space.
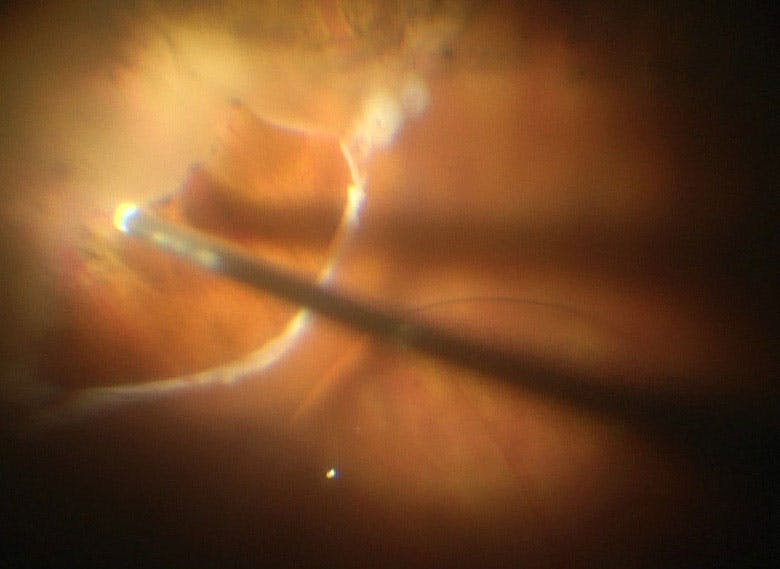
Figure 3. This ultra-widefield fundus photograph depicts the eye of a patient who underwent 360° retinectomy for RD repair.
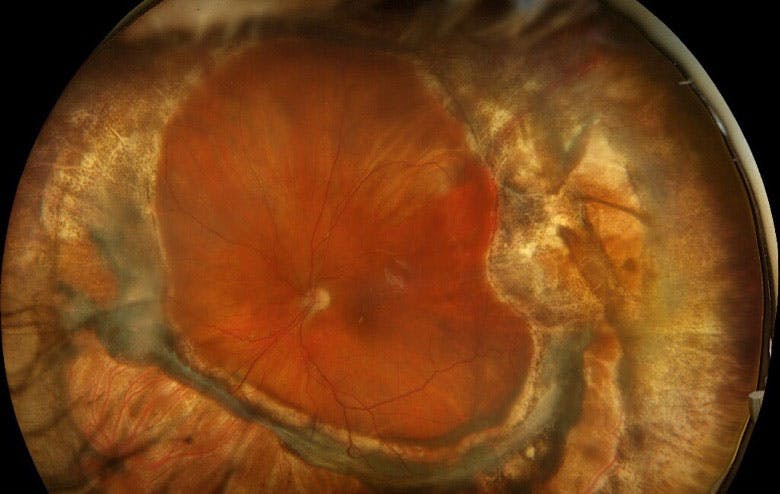
Figure 4. Before performing a retinectomy in eyes with PVR RRD, surgeons can instill a small PFO bubble to protect the macula.
LENSECTOMY OR NOT
For phakic eyes with PVR, surgery can be performed with or without lens removal. Lensectomy has the advantage of improved visualization during surgery, and it also allows for a closer vitreous base shave. Alternatively, lens removal increases surgery duration, can be proinflammatory, and may be associated with a greater incidence of postoperative complications such as cystoid macular edema and corneal edema. A retrospective study by Mahmoudzedah et al evaluated outcomes of vitrectomy with retinectomy without lensectomy for grade C PVR RRDs in phakic eyes.8 In their study, visual and anatomic outcomes were similar to prior studies where lensectomies had been performed.8
However, if a lensectomy is needed, it is essential to remove the entire capsule, as preservation of the anterior capsule may provide a scaffold for PVR over the ciliary body, increasing the risk of hypotony and compromising clinical outcomes. However, it is unclear whether this is an issue if the posterior capsule is preserved as in a standard phacoemulsification procedure with IOL implantation.
PLAN CAREFULLY
PVR detachments are complex, and their management begins even before the patient enters the OR. Take time to evaluate if the patient is at elevated risk for PVR and could benefit from SB placement unless there are plans for a retinectomy. Incorporate surgical adjuncts such as triamcinolone and dyes to improve intraoperative visualization and facilitate a smoother procedure. Find instruments that give you the most flexibility. Hemostasis is key to mitigate further PVR risk. Unless the cataract substantially limits intraoperative visualization, remember that a lensectomy may not be required in all PVR cases.
1. Pastor JC. Proliferative vitreoretinopathy: An overview. Surv Ophthalmol. 1998;43(1):3-18.
2. Pastor JC, De La Rúa ER, Martín F. Proliferative vitreoretinopathy: risk factors and pathobiology. Prog Retin Eye Res. 2002;21(1):127-44.
3. Nagasaki H, Shinagawa K, Mochizuki M. Risk factors for proliferative vitreoretinopathy. Prog Retin Eye Res. 1998;17(1):77-98.
4. Salabati M, Massenzio E, Kim J, et al. Primary retinal detachment repair in eyes deemed high risk for proliferative vitreoretinopathy: surgical outcomes in 389 eyes. Ophthalmol Retina. 2023;7(11):954-958.
5. Storey P, Alshareef R, Khuthaila M, et al. Pars plana vitrectomy and scleral buckle versus pars plana vitrectomy alone for patients with rhegmatogenous retinal detachment at high risk for proliferative vitreoretinopathy. Retina. 2014;34(10):1945-1951.
6. Israilevich RN, Starr MR, Mahmoudzadeh R, et al. Factors associated with good visual acuity outcomes after retinectomy in eyes with proliferative vitreoretinopathy: good visual acuity following retinectomy for RD. Am J Ophthalmol. 2022;240:143-148.
7. Wakabayashi T, Samuelson AG, Oshima Y, et al. Efficacy of internal limiting membrane peeling for grade c proliferative vitreoretinopathy: an international multicenter study [published online ahead of print July 9, 2025]. Ophthalmol Retina.
8. Mahmoudzadeh R, Mokhashi N, Anderson H, et al. Outcomes of retinectomy without lensectomy in rhegmatogenous retinal detachments with proliferative vitreoretinopathy. Ophthalmol Retina. 2023;7(1):52-58.











_1773249222.png?auto=compress,format&w=75)










