AT A GLANCE
- Vitrectomy with scleral buckling in eyes with primary rhegmatogenous retinal detachment (RRD) and high-risk features may be advised.
- Extended internal limiting membrane peeling in eyes with proliferative vitreoretinopathy (PVR) grade C may be recommended for anatomic and visual acuity considerations.
- In the GUARD trial of methotrexate for the prevention of PVR after RRD repair, 44% more redetachments occurred in the historical control group compared with the intervention arm.
Proliferative vitreoretinopathy (PVR) remains an unmet clinical need in the practice of surgical retina, with ongoing research seeking to better describe, manage, and prevent this challenging disease process.1 Several such projects were recently published or presented at the 2023 American Society of Retinal Specialists (ASRS) Annual Meeting, adding practical considerations to the current body of literature. In this article, I summarize the recent work that may aid the retina specialist when encountering PVR during repair of rhegmatogenous retinal detachment (RRD).
HIGH-RISK RETINAL DETACHMENT
When encountering a primary RRD, several risk factors for PVR have been previously described, including vitreous hemorrhage, preoperative PVR, large or chronic detachments, large or multiple retinal breaks, choroidal detachment, intraocular inflammation, and extensive cryotherapy (Figure 1).2 However, limited studies exist that evaluate the role of these high-risk features on RRD repair outcomes and whether such risk factors truly influence single-surgery anatomic success (SSAS) in primary RRD.
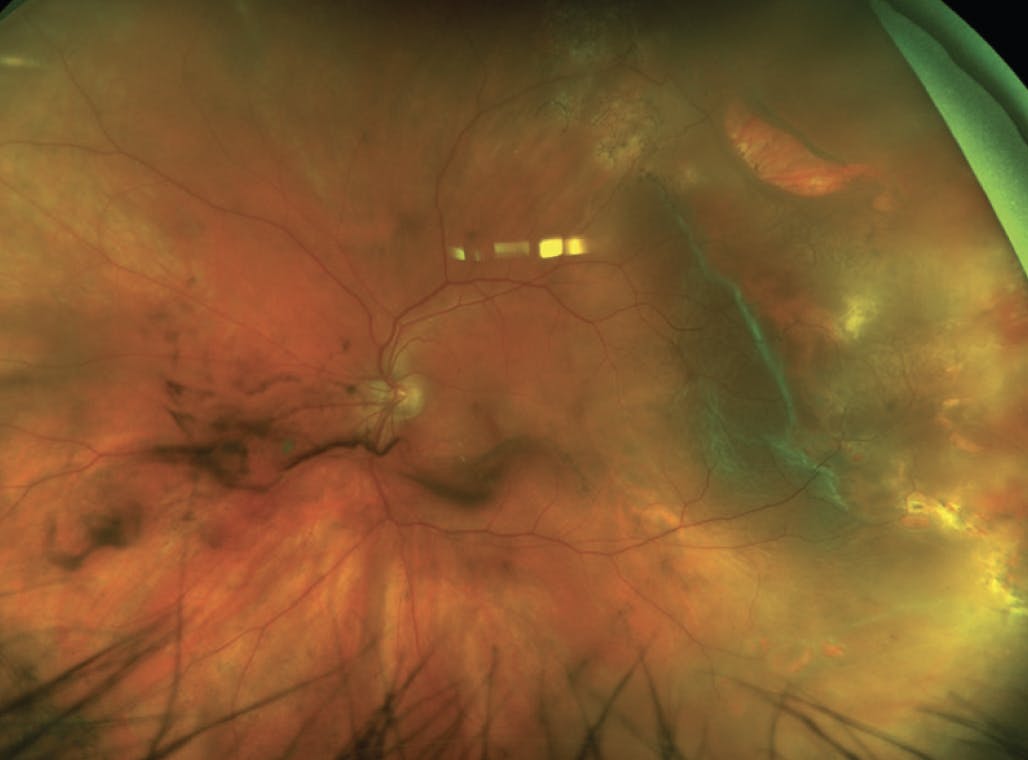
Figure 1. In this patient who presented with a high-risk RRD, vitreous hemorrhage and multiple breaks are present. The larger break superotemporally has rolled edges, consistent with PVR grade B.
Salabati et al recently published a study of 389 eyes that evaluated SSAS rates for primary RRD repair in eyes with high-risk features for PVR.3 Eyes were deemed high-risk if they had at least one of the following risk factors: preoperative PVR grade A or B, vitreous hemorrhage, RRD involving 50% or more of the retinal area, presence of three or more retinal breaks, history of prior cryotherapy, presence of choroidal detachment, and/or duration of RRD greater than 2 weeks. Choice of surgical technique was pars plana vitrectomy (PPV) in 67.9% of eyes and combined PPV with scleral buckling (SB) in 32.1% of eyes. Overall, the SSAS rate was 71.5% at 3 months after surgery. When comparing surgical techniques, the SSAS rate was significantly higher in eyes treated with PPV/SB compared with PPV alone (80.8% vs 67%, P = .006). This higher SSAS was noted even in eyes treated with PPV/SB that were more likely to be macula-off and have a greater extent of RRD compared with eyes in the PPV only group (Figure 2).3
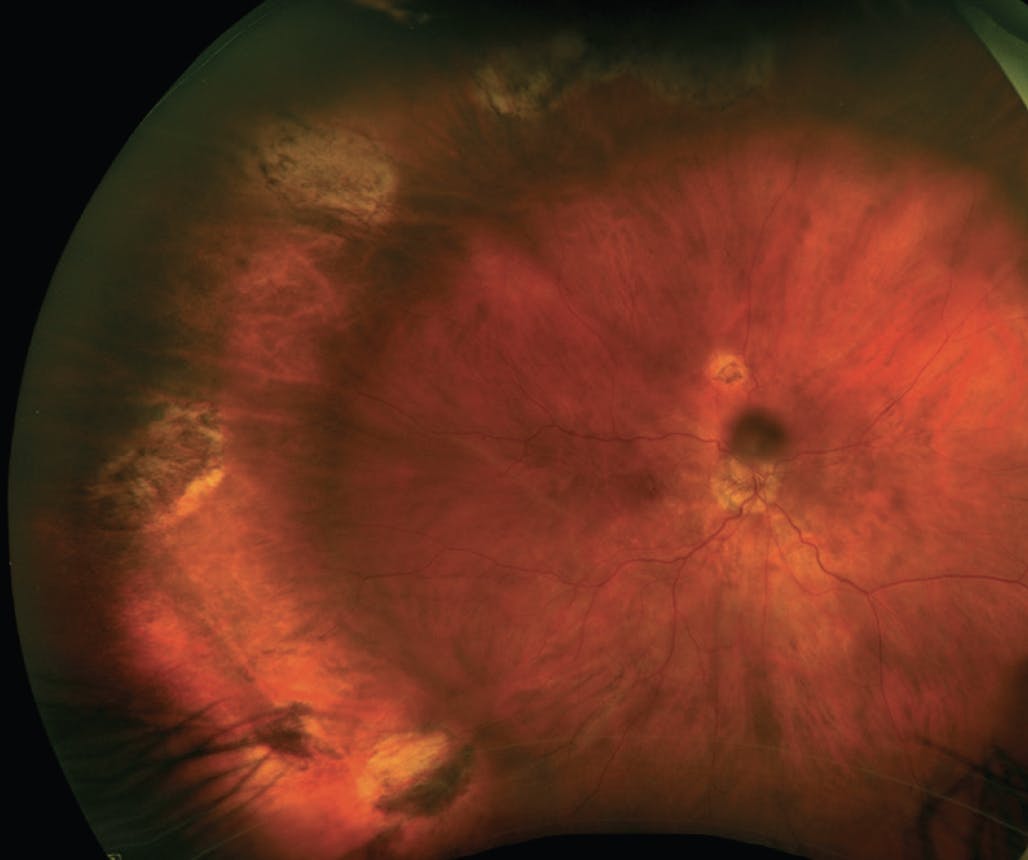
Figure 2. A high-risk RD was repaired with PPV and SB. The initial detachment was macula-off with 8 clock hours of detachment. Note the multiple breaks treated with laser retinopexy temporally.
The authors completed a multivariate analysis to assess the effect of the various risk factors on SSAS. However, no individual risk factor was significantly associated with the rate. Notably, use of PPV/SB was the only feature associated with SSAS (odds ratio [OR] = 2.04, P = .019).
The relatively lower SSAS rate of 71.5% observed in eyes with RRD and high-risk features, compared with the 88% to 90% rate reported in other large series of primary RRD,4-6 suggests these risk factors remain clinically relevant. Given the significantly higher SSAS rate with PPV/SB observed in this study, use of PPV/SB in eyes with primary RRD and high-risk features may be advised. Additional study of this high-risk cohort is of interest and may be a study population worth including in future clinical trials for PVR.
EXTENDED ILM PEELING
Internal limiting membrane (ILM) peeling has been suggested during repair of RD with or without PVR.7 Previous data yielded lower rates of epiretinal membrane formation and recurrent RD incidence with ILM peeling, but results have conflicted between studies and RD populations.
Recently, Yonekawa et al presented data regarding the anatomic and visual benefits of extended ILM peeling for patients with PVR grade C RDs.8 The authors describe extended ILM peeling as the peeling of ILM not only within the macula, but also beyond the vascular arcades and to the furthest reasonable extent that is surgically available, often under perfluorocarbon liquid.
The retrospective study analyzed 307 eyes of 307 patients from five institutions. The minimum follow-up was 6 months, and 157 eyes treated with extended ILM peeling were compared with 150 eyes without ILM peeling. At 6 months, the reattachment rate under fluid was higher (61% vs 45%, P = .005), and the number of redetachments was fewer (0.39 vs 0.59, P = .010) in the extended peeling group. Visual acuity and visual acuity improvement were also better in the ILM peeling group (P < .001 and P = .043, respectively).8
Based on this study, extended ILM peeling during RRD repair in eyes with PVR grade C may be recommended for both anatomic and visual acuity considerations. Data from a prospective clinical trial would be helpful to confirm the benefits of extended ILM peeling in PVR-related surgery.
INTRAVITREAL METHOTREXATE
Results of the GUARD trial, a phase 3 prospective clinical trial investigating repeated intravitreal injections of 0.8% methotrexate (ADX-2191, Aldeyra Therapeutics) in PVR RRD, have been highly anticipated by the retina community. Per study protocol, 13 injections of methotrexate were administered over 4 months. The intervention cohort was compared with historical controls, with the primary endpoint being the incidence of recurrent RD requiring reoperation within 6 months.9
Results of the study were presented at the 2023 ASRS Annual Meeting.9 The intervention and historical control arms were similar regarding phakic status and history of open-globe repair. The rate of recurrent RD was 24% in the intervention arm and 39% in historical controls at 6 months (P = .024). The primary endpoint was achieved, with the intervention cohort having significantly fewer overall recurrent RDs compared with historical controls (18.8% vs 38.7%, OR = 0.49, P = .001) through week 24. The researchers noted that 44% more detachments occurred in the historical control group compared with the intervention arm. Additional outcomes, such as epiretinal membrane formation, hypotony, and adverse events, occurred less often in the intervention arm, although the study was not powered to detect differences for these secondary outcomes.9
Data from the GUARD trial were encouraging, with the primary endpoint met. Additional data may further establish intravitreal methotrexate in the treatment of PVR RRD.
IN THE OR
Renewed interest in the treatment and prevention of PVR has resulted in several ongoing and recently completed research studies. From these data, several insights and considerations may be immediately relevant, including the use of PPV/SB in primary RRDs with high-risk features for PVR and extended ILM peeling in eyes with established PVR grade C RRDs.3,8
Moreover, data from the GUARD trial may support the near-term use of intravitreal methotrexate for the prevention of PVR, with additional data regarding efficacy and guidance for case selection of special interest.
Ongoing and future studies will be helpful to further classify high-risk features and cohorts, identify optimal surgical interventions for this population, and, hopefully, identify and refine adjunct therapeutics for the treatment and prevention of PVR.
1. Khan MA, Brady CJ, Kaiser RS. Clinical management of proliferative vitreoretinopathy: an update. Retina. 2015;35(2):165-175.
2. Nagasaki H, Shinagawa K, Mochizuki M. Risk factors for proliferative vitreoretinopathy. Prog Retin Eye Res. 1998;17(1):77-98.
3. Salabati M, Massenzio E, Kim J, et al. Primary retinal detachment repair in eyes deemed high risk for proliferative vitreoretinopathy: surgical outcomes in 389 eyes [published online ahead of print July 13, 2023]. Ophthalmol Retina.
4. Rao RC, Blinder KJ, Smith BT, Shah GK. Internal limiting membrane peeling for primary rhegmatogenous retinal detachment repair. Ophthalmology. 2013;120(5):1102-1103.e1-2.
5. Joseph DP, Ryan EH, Ryan CM, et al. Primary retinal detachment outcomes study: pseudophakic retinal detachment outcomes: Primary Retinal Detachment Outcomes Study Report Number 3. Ophthalmology. 2020;127(11):1507-1514.
6. Ryan EH, Ryan CM, Forbes NJ, et al. Primary Retinal Detachment Outcomes Study Report Number 2: phakic retinal detachment outcomes. Ophthalmology. 2020;127(8):1077-1085.
7. Fallico M, Russo A, Longo A, et al. Internal limiting membrane peeling versus no peeling during primary vitrectomy for rhegmatogenous retinal detachment: a systematic review and meta-analysis. PloS One. 2018;13(7):e0201010.
8. Yonekawa Y. Extended internal limiting membrane peeling for grade C proliferative vitreoretinopathy: international multicenter study. Paper presented at ASRS Annual Meeting; July 31, 2023; Seattle, WA.
9. Flaxel C. Phase 3 GUARD trial of ADX-2191 (methrotrexate for intravitreal administration) to prevent proliferative vitreoretinopathy and other postoperative complications. Paper presented at ASRS Annual Meeting; July 21, 2023; Seattle, WA.











_1773249222.png?auto=compress,format&w=75)










